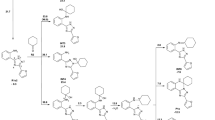
Abstract
Under the Influence of anhydrous aluminum chloride, cyclohexane converts 2,2,4-trimethyl-1,2-dihydroquinoline to the tetrahydro derivative and alkylates it at the 7-position with simultaneous reduction of the Δ3-bond.
Similar content being viewed by others
Literature cited
B. A. Lugovik, P. V. Borodin, L. G. Yudin, S. M. Vinogradova, and A. N. Kost, Khim. Geterotsikl. Soedin., 795 (1971).
B. A. Lugovik, A. N. Kost, and L. G. Yudin, Dokl. Akad. Nauk SSSR, 170, 340 (1966).
B. A. Lugovik, P. V. Borodin, L. G. Yudin, and A. N. Kost, Khim. Geterotsikl. Soedin., 1512 (1970).
A. Rosowsky and E. Modest, J. Org. Chem., 30, 1832 (1965).
N. D. Zelinskii, Collection of Works, Vol. 3 [in Russian], Izd. AN SSSR (1955), p. 28.
C. D. Nenitzescu and C. D. Ionescu, Ann., 491, 189 (1931).
C. D. Nenitzescu and A. Dragan, Ber., 66, 1892 (1933).
K. D. Nenitzescu, Usp. Khim., 26, 399 (1957).
W. H. Clitte, J. Chem. Soc., 1327 (1933).
J. W. Elliott and P. Yates, J. Org. Chem., 26, 1237 (1961).
N. D. Zelinskii, Ber., 44, 2781 (1911).
Author information
Authors and Affiliations
Additional information
See [1] for communication III.
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 7, pp. 977–980, July, 1971.
Rights and permissions
About this article
Cite this article
Lugovik, B.A., Borodin, P.V., Yudin, L.G. et al. Reactions of 1,2-dihydroquinolines. Chem Heterocycl Compd 7, 912–915 (1971). https://doi.org/10.1007/BF00475725
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00475725