Abstract
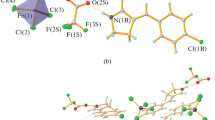
5,7-Dimethyl-3-phenylfuroxano[5,4-a]pyrimidiniumperchlorate (II) was obtained by the reaction of 4-amino-3-phenylfuroxan with acetylacetone in the presence of HClO4. The structures of II and its furazan analog I were proved by x-ray diffraction analysis, and their 13C NMR spectra were studied. The cations of I and II (which contain a conjugated condensed system with delocalization of the positive charge) and their monocyclic analogs — phenylfarazan, 3-phenylfuroxan, and 4,6-dimethylpyrimidine — were subjected to quantum-chemical calculation by the CNDO/2 method.
Similar content being viewed by others
Literature cited
L. I. Khmel'nitskii, S. S. Novikov, and T. I. Godovikov, The Chemistry of Furoxans (Structure and Synthesis) [in Russian], Nauka, Moscow (1981).
I. P. Bachkovskii, A. P. Mikhailovskii, and V. A. Chuiguk, Ukr. Khim. Zh., 46, 637 (1980).
A. S. Batsanov and Yu. T. Struchkov, Zh. Struk. Khim, 26, No. 1, 65 (1985).
A. Bondi, J. Chem. Phys., 68, 441 (1964).
R. Calvino, A. Gasco, A. Serafmo, and D. Viterbo, J. Chem. Soc., Perkin Trans. II, No. 9, 1240 (1981).
M. Mellini and S. Merlino, Ada Cryst., B32, 1079 (1976).
P. J. Wheatly, Acta Cryst., 13, 80 (1960).
A. R. Gagneux and R. Meier, Helv. Chim. Acta, 53, 1883 (1970).
R. G. Gerr, A. I. Yanovskii, and Yu. T. Struchkov, Kristallografiya, 28, 1029 (1983).
Author information
Authors and Affiliations
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 2, pp. 233–238, February, 1992.
Rights and permissions
About this article
Cite this article
Struchko, Y.T., Batsanov, A.S., Chuiguk, V.A. et al. 5,7-Dimethyl-3-phenylfurazano- and -furoxano[5,4-a]pyrimidinium perchlorates: New types of condensed systems. Chem Heterocycl Compd 28, 193–197 (1992). https://doi.org/10.1007/BF00473944
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00473944