Abstract
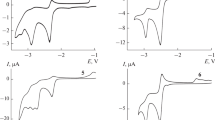
It was shown by cyclic voltammetry and electrolysis at controlled potential at a platinum electrode and also by TLC and UV spectroscopy that the electrochemical oxidation of substituted 1H-1,2-diazaphenalenes in anhydrous acetonitrile takes place in a single one-electron stage with the formation of a stable radicalation. The oxidation potentials of a series of derivatives of 1H-1,2-diazaphenalene and the parameters of the equation for the correlation of these potentials with the Taft substituent constants were determined.
Similar content being viewed by others
Literature Cited
A. F. Pozharskii, Khim. Geterotsikl. Soedin., 1977, No. 6, 723.
G. K. Budnikov, Principles and Application of Voltammetric Oscillographic Polarography [in Russian], Izd. Kazan. Univ., Kazan' (1975), p. 103.
B. Ya. Kaplan, R. G. Pats, and R. M-F. Salikhdzhanova, Alternating-Current Voltammetry [in Russian], Khimiya, Moscow (1985), p. 34.
A. M. Bond, Polarographic Methods in Analytical Chemistry [Russian translation], Khimiya, Moscow (1983), p. 27.
A. S. Morkovnik, V. B. Panov, D. M. Elisevich, and V. V. Mezheritskii, Zh. Org. Khim., 19, 1984 (1983).
V. V. Mezheritskii, V. V. Tkachenko, O. N. Zhukovskaya, D. M. Elisevich, and G. N. Dorofeenko, Zh. Org. Khim., 17, 627 (1981).
V. A. Palm, Fundamentals of Quantitative Theory of Organic Reactions [in Russian], Khimiya, Leningrad (1977), p. 324.
Author information
Authors and Affiliations
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 8, pp. 1098–1100, August, 1987.
Rights and permissions
About this article
Cite this article
Dmitriev, A.B., Okolelova, M.S., Bogdashev, N.N. et al. Electrochemical oxidation of substituted 1H-1,2-diazaphenalenes. Chem Heterocycl Compd 23, 878–880 (1987). https://doi.org/10.1007/BF00473465
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00473465