Abstract
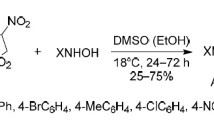
Proton magnetic resonance spectroscopy was used to establish that the addition of hydroxylamine to the exocyclic double bond of 4-benzamido-3-oxo-2-(δ-carbomethoxybutylidene)-thiophane occurs stereospecifically; the compound formed has the trans configuration with respect to the substituents in the 2 and 4 positions.
Similar content being viewed by others
Literature cited
S. A. Harris, D. E. Wolf, R. Mozingo, G. E. Arth, R. C. Anderson, N. R. Easton, and K. Folkers, J. Am. Chem. Soc., 67, 2096 (1945).
S. D. Mikhno, N. S. Kulachkina, and V. M. Berezovskii, Khim. Geterotsikl. Soedin., 179 (1966).
S. D. Mikhno, T. M. Filippova, N. S. Kulachkina, T. N. Polyanskaya, V. V. Mishchenko, I. K. Shmyrev, I. M. Kustanovich, and V. M. Berezovskii, Khim. Geterotsikl. Soedin., 760 (1972).
S. D. Mikhno, T. M. Filippova, N. S. Kulachkina, T. N. Polyanskaya, I. M. Kustanovich, and V. M. Berezovskii, Khim. Geterotsikl. Soedin., 897 (1972).
Author information
Authors and Affiliations
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 12, pp. 1600–1601, December, 1972.
Rights and permissions
About this article
Cite this article
Mikhno, S.D., Filippova, T.M., Polyanskaya, T.N. et al. Establishment of the configuration of 4-benzamido-3-oxo-2-(α-hydroxyamino-δ-carbomethoxybutyl)-thiophane. Chem Heterocycl Compd 8, 1449–1450 (1972). https://doi.org/10.1007/BF00471825
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00471825