Abstract
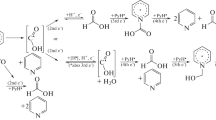
The electrochemical reduction of some chlorophthalazine derivatives that differ with respect to the nature of the substituent and the heteroring (phthalazines, phthalazones, and 1,2,4-triazolo[3,4-a]phthalazines) on a dropping mercury electrode in dimethylformamide was studied by polarography, coulometry, and preparative electrolysis with isolation and identification of the electrolysis products. It is shown that the carbon-chlorine bond is cleaved initially in the cathode reduction of the investigated compounds to give the corresponding dehalogenated heterocycles.
Similar content being viewed by others
Literature cited
K. Alwair and J. Grimshaw, J. Chem. Soc., Perkin II, 1811 (1973).
J. E. O'Reilly and P. J. Elving, J. Electroanal. Chem., 75, 507 (1977).
I. M. Sosonkin, G. N. Strogov, V. N. Novikov, and T. K. Ponomareva, Khim. Geterotsikl. Soedin., No. 1, 23 (1977).
H. Lund and E. T. Jensen, Acta Chem. Scand., 24, 1867 (1970).
B. I. Buzykin, N. N. Bystrykh, A. P. Stolyarov, S. A. Flegontov, V. V. Zverev, and Yu. P. Kitaev, Khim. Geterotsikl. Soedin., No. 3, 402 (1976).
K. T. Potts and C. Lovelette, J. Org. Chem., 34, 3221 (1969).
K. Ueno, R. Moroi, M. Kitagawa, K. Asano, and S. Miyazaki, Chem. Pharm. Bull., 24, 1068 (1976).
L. Nadjo and J. M. Saveant, J. Electroanal. Chem., 30, 41 (1971).
J. Druey and B. H. Ringier, Helv. Chim. Acta, 34, 197 (1951).
Author information
Authors and Affiliations
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 4, pp. 541–544, April, 1979.
Rights and permissions
About this article
Cite this article
Ivanova, V.K., Buzykin, B.I. & Bystrykh, N.N. Electrochemical dechlorination of some derivatives of 1-chlorophthalazine in dimethylformamide. Chem Heterocycl Compd 15, 443–446 (1979). https://doi.org/10.1007/BF00471784
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00471784