Abstract
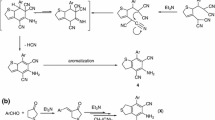
Under the influence of phosphorus oxychloride in an inert solvent, 1-aryl-2-acylhydrazines undergo intramolecular rearrangement to give 2-amino-3-alkylindoles. The latter are autooxidized in alkaline media to give 3-hydroxy-2-aminoindolenines or 3-hydroxy-2-iminoindolines. The structures of all of the compounds were proved by UV, IR, and PMR spectroscopy and also by mass-spectrometric data.
Similar content being viewed by others
Literature cited
L, G. Yudin, A. N. Kost, and E. Ya. Zinchenko, Khim. Geterotsikl. Soedin., 332 (1973).
Yu. N. Portnov, G. A. Golubeva, and A. N. Kost, Khim. Geterotsikl. Soedin., 61 (1972).
R. S. Sagitullin, V. I. Gorbunov, A. N. Kost, N.B. Kuplet-skaya, and V. V. Chistyakov, Khim. Geterotsikl. Soedin., 364 (1970).
W. E. Stewart and T. H. Siddall, Chem. Rev., 70, 517 (1970).
A. N. Kost, G. A. Golubeva, and Yu. N. Portnov, Dokl. Akad. Nauk SSSR, 200, 342 (1970).
L. G. Yudin, V. A. Budylin, A. N. Kost, and V. I. Minkin, Dokl. Akad. Nauk SSSR, 176, 1096 (1967).
M. Nakagawa, H. Tamaguchi, and T. Hino, Tetrahedron Lett., 4035 (1970).
Author information
Authors and Affiliations
Additional information
See [1] for communication XXXIV.
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 4, pp. 511–515, April, 1973.
Rights and permissions
About this article
Cite this article
Golubeva, G.A., Portnov, Y.N. & Kost, A.N. Chemistry of indole. Chem Heterocycl Compd 9, 471–475 (1973). https://doi.org/10.1007/BF00471528
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00471528