Abstract
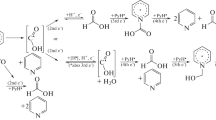
It is shown that the first step in the reduction of the investigated oxadiazoles on a dropping mercury cathode in dimethylformamide corresponds to two-electron cleavage of the N-O bond. The degree of reduction is determined by the nature and position of the substituents in the oxadiazole ring and by the rate of protonation of the intermediately formed particles.
Similar content being viewed by others
Literature cited
Z. V. Todres, Z. I. Fodiman, and É. S. Levin, Khim. Geterotsikl. Soedin., No. 5, 604 (1974).
I. G. Markova, M. K. Polievktov, and S. D. Sokolov, Zh. Obshch. Khim., 46, 398 (1976).
V. D. Bezuglyi, N. P. Shimanskaya, and E. M. Peresleni, Zh. Obshch. Khim., 34, 3540 (1964).
S. G. Mairanovskii,Catalytic and Kinetic Waves in Polarography [in Russian], Moscow (1966), p. 168.
C. Moussebois and J. F. M. Oth, Helv. Chim. Acta, 47, 942 (1964).
G. Del Re, J. Chem. Soc., 3324 (1962).
V. G. Mairanovskii, A. A. Engovatov, and G. I. Samokhvalov, Zh. Anal. Khim., 25, 2235 (1970).
Author information
Authors and Affiliations
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 1, pp. 38–40, January, 1979.
Rights and permissions
About this article
Cite this article
Markova, I.G., Polievktov, M.K. & Sokolov, S.D. 1,2,4- and 1,2,5-oxadiazoles. Chem Heterocycl Compd 15, 31–33 (1979). https://doi.org/10.1007/BF00471193
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00471193