Abstract
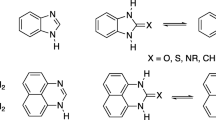
It is shown by means of IR, UV, and PMR spectroscopy that 5-amino-6-thio-1,6-dihydropyrimidines exist primarily in the thione form both in the crystalline state and in solution.
Similar content being viewed by others
Literature cited
M. P. Nemeryuk and T. S. Safonova, Khim. Geterotsikl. Soedin., 192 (1975).
T. S. Safonova and M. P. Nemeryuk, Khim. Geterotsikl. Soedin., 486 (1967).
T. S. Safonova, M. P. Nemeryuk, L. A. Myshkina, and N. I. Traven', khim. Geterotsikl. Soedin., No. 7, 944 (1972).
V. Inoue, N. Furutachi, and K. Nakanishi, J. Org. Chem., 31, 175 (1966).
D. I. Brown and T. Teitel, Austral. J. Chem., 17, 567 (1964).
E. C. Taylor, I. Barton, and W. Paudler, J. Org. Chem., 26, 4961 (1961).
L. Marchal, R. Promel, R. H. Martin, and A. Cardon, Bull. Soc. Chim., Belge, 69, 177 (1960).
Author information
Authors and Affiliations
Additional information
See [1] for communication XXXIII.
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 3, pp. 398–401, March, 1977.
Rights and permissions
About this article
Cite this article
Keremov, A.F., Peresleni, E.M., Vlasova, T.F. et al. Derivatives of condensed pyrimidine, pyrazine, and pyridine systems. Chem Heterocycl Compd 13, 322–325 (1977). https://doi.org/10.1007/BF00470320
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00470320