Abstract
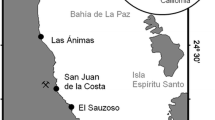
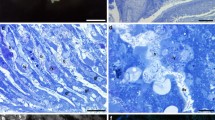
Sulfur content and fine structure were studied for tissues of three species of clams, Lucinoma annulata, Calyptogena elongata and Lucina floridana, which inhabit sulfide-rich environments and whose gills harbor symbiotic sulfur bacteria. Lucinoma annulata and C. elongata were dredged from the Santa Barbara basin, California, USA, at a depth of 480 to 490 m, and Lucina floridana were dug from below the roots of seagrasses in Saint Joseph Bay, Florida, at a depth of 0.25 to 2m. Foot tissue of Lucinoma annulata, without symbionts, had a total sulfur content of 1.4±0.1 (SD) mg 100 mg-1 dry weight of tissue (%DW). The symbiont-containing gill tissue of different individuals of L. annulata varied in color from dark red to pale yellow, and the total sulfur content was 2.5±0.4% DW in red gills and was 5.6±3.3 % DW in the yellowest gills. Maintenance of L. annulata in the laboratory for 21 d in the absence of sulfide resulted in the loss from the gill of yellow deposits which were elemental sulfur in the form of liquid-crystalline sulfur globules rather than solid orthorhombic sulfur crystals. The foot tissue did not contain elemental sulfur. When examined by freeze-etch microscopy, sulfur globules were found only within bacteria and not in the animal host cytoplasm. Sulfur globules were confined to the periplasmic space of the bacteria. C. elongata and Lucina floridana resembled Lucinoma annulata in the physical form and distribution of elemental sulfur. The absence of elemental sulfur in the animal cytoplasm suggests that its formation from sulfide is not a detoxification scheme to protect animal tissue from sulfide toxicity. The sulfur deposits probably represent inorganic energy reserves that permit the symbiotic bacteria to function even during the temporary absence of external sulfide.
Similar content being viewed by others
Literature cited
Cavanaugh, C. M., S. L. Gardner, M. L., Jones, H. J. Jannasch and J. B. Waterbury: Procaryotic cells in the hydrothermal vent tube worm Riftia pachyptila Jones: possible chemoautotrophic symbionts. Science, N.Y. 213, 340–342 (1981)
Chen, K. Y. and J. C. Morris: Kinetics of oxidation of aqueous sulfide by O2. Envir. Sci. Technol. 6, 529–537 (1972)
Childress, J. J., A. J. Arp and C. R. Fisher, Jr.: Metabolic and blood characteristics in the hydrothermal vent tube-worm Riftia pachyptila. Mar. Biol. 83, 109–124 (1984)
Felbeck, H.: Sulfide oxidation and carbon fixation by the gutless clam Solemya reidi: an animal-bacteria symbiosis. J. comp. Physiol. 152, 3–11 (1983)
Felbeck, H., J. J. Childress and G. N. Somero: Calvin-Benson ycle and sulphide oxidation enzymes in animals from sulphide rich habitats. Nature, Lond. 293, 291–293 (1981)
Fisher, M. R. and S. J., Hand: Chemoautotrophic symbionts in the bivalve Lucina floridana from seagrass beds. Biol. Bull. mar. biol. Lab., Woods Hole 167, 445–459 (1984)
Hageage, G. J., E. D. Eanes and R. L. Gherna: X-ray diffraction studies of the sulfur globules accumulated by Chromatium species. J. Bact. 101, 464–469 (1970)
Hand, S. C., and G. N. Somero: Energy metabolism pathways of hydrothermal vent animals: adaptations to a food-rich and sulfide-rich deep-sea environment. Biol. Bull. mar. biol. Lab., Woods Hole 165, 167–181 (1983)
Holt, S. C., J. M. Schively and J. W. Greenawalt: Fine structure of selected species of the genus Thiobacillus as revealed by chemical fixation and freeze-etching. Can. J. Microbiol. 20, 1347–1351 (1974)
Jannasch, H. W.: Chemosynthesis: the nutritional basis for life at deep-sea vents. Oceanus 27, 73–78 (1984)
Jones, M. L.: Riftia pachyptila Jones: Observations on the vestimentiferan worm from the Galápagos Rift. Science, N.Y. 213, 333–336 (1981)
Jørgensen, B. B.: Ecology of the bacteria of the sulfur cycle with special reference to anoxic-oxic interface environments. Phil. Trans. R. Soc. (Ser. B) 298, 543–561 (1983)
Jørgensen, B. B. and N. P. Revsbech: Colorless sulfur bacteria, Beggiatoa spp. and Thiovulum spp., on O2 and H2S microgradients. Appl. envirl. Microbiol 45, 1261–1270 (1983)
Kaplan, I. R., K. O. Emery and S. C. Rittenberg: The distribution and isotopic abundance of sulphur in recent marine sediments off southern California. Geochim. cosmochim. Acta 27, 297–331 (1963)
Lawrey, N. H., V. Jani and T. E. Jensen: Identification of the sulfur inclusion body in Beggiatoa alba B18LD by energy-dispersive X-ray microanalysis. Curr. Microbiol. 6, 71–74 (1981)
Moriarty, D. J. W. and D. J. D. Nicholas: Products of sulphide oxidation in extracts of Thiobacillus concretivorus. Biochim. biophys. Acta 197, 143–151 (1970)
National Research Council: Hydrogen sulfide, 183 pp. Baltimore: University Park Press 1979. (Committee on Medican and Biological Effects of Environmental Pollutants, Subcommittee on Hydrogen Sulfide; Division of Medical Sciences, Assembly of Life Sciences)
Nicolson, G. L. and G. L. Schmidt: Structure of the Chromatium sulfur particle and its protein membrance. J. Bact. 105, 1142–1148 (1971)
Powell, M. and G. N. Somero: Blood components prevent sulfide poisoning of respiration of the hydrothermal vent tube worm Riftia pachyptila. Science, N.Y. 219, 297–299 (1983)
Reid, R. G.: Aspects of the biology of a gutless species of Solemya (Bivalvia: Protobranchia). Can. J. Zool. 58, 386–393 (1980)
Remsen, C. C.: Comparative subcellular architecture of photosynthetic bacteria. In: The photosynthetic bacteria, pp 31–60. Ed. by R. K. Clayton and W. R. Sistrom. New York: Plenum Press 1978
Schedel, M. and H. Trüper: Anaerobic oxidation of thiosulfate and elemental sulfur in Thiobacillus denitrificans. Archs, Microbiol. 124, 205–210 (1980)
Schmidt, G. L., G. L. Nicolson and M. D. Kamen: Composition of the sulfur particle of Chromatium vinosum strain D. J. Bact. 105, 1137–1141 (1971)
Strohl, W. R., I. Geffers and J. M. Larkin: Structure of the sulfur inclusion envelopes from four Beggiatoas. Curr. Microbiol. 6, 75–79 (1981)
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Vetter, R.D. Elemental sulfur in the gills of three species of clams containing chemoautotrophic symbiotic bacteria: a possible inorganic energy storage compound. Mar. Biol. 88, 33–42 (1985). https://doi.org/10.1007/BF00393041
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00393041