Abstract
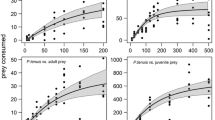
One strategy for predator avoidance involves the creation of a structural refuge. Onuphid polychaetes characteristically ornament above-sediment portions of their tubes (=tube-caps) with shell and algal debris. These species feed on the sediment surface through an opening in the tube-cap, and thus the ability to detect a surface predator while feeding would be advantageous. An investigation of the function of the ornamentation in Diopatra spp. suggests that ornamentation facilitates predator detection and avoidance. Three intensities of mechanical disturbance were applied directly to D. ornata tube-caps. When ornamented tube-caps were stimulated, the response of worms to the three intensities were significantly different, and increased in duration with greater intensities. In contrast, when no ornamentation was present, the responses were not significantly different, and were similar to the low-intensity response when ornamentation was present. This suggests that ornamentation should allow a worm to distinguish between harmful (high intensity due to mobile epifaunal predators) and profitable (low intensity due to drift algae) disturbances, and furthermore, worms with ornamented tube-caps should be more successful in escaping surface predators. Densities of intertidal populations of D. cuprea at Tom's Cove, Virginia, USA, correlated with the amount of tube ornamentation, consistent with this predator detection and avoidance hypothesis. Final tube-cap lengths of laboratory D. ornata and field D. cuprea were inversely related to the size of attached debris. When large debris was attached, cap formation ceased earlier and caps were shorter than when small debris or no debris was attached. Cryptic and food-catching functions would predict that highly ornamented tubes would be most advantageous, while only a few large debris would be required for disturbance transmission. Laboratory specimens showed no selectivity between 0.5 or 1.5 cm2 shell; or 1.0 cm2 or 3 to 8 cm2 algae, and utilized shell and algae when available and according to relative abundance. Tube-caps of field specimens also showed positive correlations between shell attached and shell abundance in the local sediment. While such lack of selectivity may enhance cryptic properties of the tube-cap, it is argued that conditions seldom exist which would permit selectivity of debris size or of specific material. These data are consistent with the hypothesis that ornamentation functions as a created refuge for predator detection and avoidance and further suggest that the availability of ornamental debris in the environment indirectly can affect these species' distributions and abundances.
Similar content being viewed by others
Literature Cited
Brenchley, G.A.: Competition, disturbance, and community structure: the importance of physical structure in a marine epifaunal community, 69 pp. M.S. thesis, University of Maryland, College Park 1975
Dayton, P.K., G.A. Robilliard and R.T. Paine: Benthic faunal zonation as a result of anchor ice at McMurdo Sound, Antarctica. In: Antarctic ecology, Vol. 1. pp 244–258. London: Academic Press 1970
Edmunds, M.: Defence in animals: a survey of anti-predator defences, 357 pp. Essex: Longman Group Ltd. 1974
Huffaker, C.B.: Experimental studies on predation: dispersion factors and predator-prey oscillations. Hilgardia 27, 343–383 (1958)
Mangum, C.P., S.L. Santos and W.R. Rhodes, Jr.: Distribution and feeding in the onuphid polychaete, Diopatra cuprea (Bosc). Mar. Biol. 2, 33–40 (1968)
Myers, A.C.: Some palaeoichnological observations on the tube of Diopatra cuprea (Bosc): Polychaeta, Onuphidae. In: Trace fossils, pp 331–334. Ed. by T.P. Crimes and J.C. Harper. Liverpool: Seel House Press 1970. (Geol. J. Spec. Iss. No. 3)
—: Tube-worm-sediment relationships of Diopatra cuprea (Polychaeta: Onuphidae). Mar. Biol. 17, 350–356 (1972)
Pillsbury, R.W.: Natural anchoring of the red alga, Gracilaria confervoides (L.) Grev. on unstable bottom by association with an annelid worm. Can. J. Res. (Ser. C) 28, 393–410 (1950)
Portmann, A.: Animal camouflage, 111 pp. Ann Arbor: University of Michigan Press 1959
Sokal, R.R. and F.J. Rohlf: Biometry, 776 pp. San Francisco: W.H. Freeman & Co. 1969
Verrill, A.E. and S.I. Smith: Report upon the invertebrate animals of Vineyard Sound and adjacent waters, with an account of the physical features of the region, 478 pp. Washington, D.C.: U.S. Government Printing Office 1874. [Reprint of Rep. U.S. Commnr Fish 1873, 295–852 (1871/1872)]
Woodin, S.A.: Structural heterogeneity and predation in an infaunal system. Am. Zool. 16, p. 195 (1976)
Author information
Authors and Affiliations
Additional information
Communicated by M.R. Tripp, Newark
Rights and permissions
About this article
Cite this article
Brenchley, G.A. Predator detection and avoidance: Ornamentation of tube-caps of Diopatra spp. (Polychaeta: Onuphidae). Mar. Biol. 38, 179–188 (1976). https://doi.org/10.1007/BF00390771
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00390771