Abstract
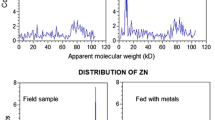
The zinc and copper associated with the soft tissues of the oyster Ostrea edulis Linnaeus have been separated into a soluble component and a tissue-residue, cell-debris bound component. In the case of zinc, the tissue-bound component was found to contain at least two species of complex; a firmly-bound species, exchangeable with 65Zn2+ and a less-firmly, reversibly-bound species, exchangeable with 65Zn2+. The soluble component, which constitutes some 40% of the total zinc and copper, was fractionated on Sephadex G-25 and the zinc and copper shown to be weakly-complexed to the small molecular weight compounds, taurine, lysine, ATP and possibly homarine (N-methyl-α-picolinic acid) and to be fully exchangeable with 65Zn2+. These soluble complexes can act as a freely available mobile reserve of metal to ensure a constant saturation of metal-dependent enzyme systems operating under adverse environments. Sephadex G-25 acts as a weak ion-exchange resin, which can cause a translocation of zinc and copper from its soluble weak complexes and result in the spurious association of the metals with other compounds.
Similar content being viewed by others
Literature Cited
Albert, A.: Quantitative studies of the avidity of naturally occurring substances for trace metals 1. Amino acids having only two ionizing groups. Biochem. J. 47, 531–538 (1950)
Awapara, J.: Free amino acids in invertebrates: a comparative study of their distribution and metabolism. In: Amino acid pools, pp 158–186. Ed. by J.T. Holden. Amsterdam: Elsevier 1962
Coombs, T.L.: The distribution of zinc in the oyster Ostrea edulis and its relation to enzymic activity and to other metals. Mar. Biol. 12, 170–178 (1972)
Feng, S.Y., E.A. Khairallah and W.J. Canzonier: Hemolymph-free-amino-acids and related nitrogenous compounds of Crassostrea virginica infected with Bucephalus sp. and Minchinia Nelsoni. Comp. Biochem. Physiol. 34, 547–556 (1970)
Gasteiger, E.L., P.C. Haake and J.A. Gergen: An investigation of the distribution and function of homarine (N-methyl picolinic acid). Ann. N. Y. Acad. Sci. 90, 622–636 (1960)
Hiltz, D.F.: Occurrence of trigonelline (N-methyl nicotinic acid) in the adductor muscle of a lamellibranch, the sea scallop (Placopecten magellanicus). J. Fish. Res. Bd Can. 27, 604–606 (1970)
— and W.J. Dyer: Principal acid-soluble nucleotides in adductor muscle of the scallop Placopecten magellanicus and their degradation during postmortem storage in ice. J. Fish. Res. Bd Can. 27, 83–92 (1970)
Nielsen, S.A. and E. Frieden: Some chemical and kinetic properties of oyster carbonic anhydrase. Comp. Biochem. Physiol. 41B, 875–889 (1972)
Pulido, P., J.H.R. Kagi and B.L. Vallee: Isolation and some properties of human metallothionein. Biochemistry, N.Y. 5, 1768–1777 (1966)
Romeril, M.G.: The uptake and distribution of 65Zn in oysters. Mar. Biol. 9, 347–354 (1971)
Saltman, P.: The role of chelation in metabolism. J. chem. Educ. 42, 682–687 (1965)
Salwin, H. and J.F. Bond: Quantitative determination of lactic acid and succinic acid in foods by gas chromatography. J. Ass. off. analyt. Chem. 52, 41–47 (1969)
Sillen, L.G. and A.E. Martell: Stability constants of metal-ion complexes. Spec. Publs chem. Soc. 17, 1–754 (1964)
Simpson, J.W. and J. Awapara: The pathway of glucose degradation in some invertebrates. Comp. Biochem. Physiol. 18, 537–548 (1966)
Virkar, R.A. and K.L. Webb: Free amino acid composition of the soft-shell clam Mya arenaria in relation to salinity of the medium. Comp. Biochem. Physiol. 32, 775–783 (1970)
Williams, R.J.P.: Coordination, chelation and catalysis. In. The enzymes, 2nd ed. Vol. 1. pp 391–441. Ed. by P.D. Boyer, H. Lardy and K. Myrback. New York: Academic Press 1959
Winzler, R.J.: Determination of serum glycoproteins. Meth. biochem. Analysis 2, 279–311 (1955)
Wolfe, D.A.: Zinc enzymes in Crassostrea virginica. J. Fish. Res. Bd Can. 27, 59–69 (1970)
Author information
Authors and Affiliations
Additional information
Communicated by J.H.S. Blaxter, Oban
Rights and permissions
About this article
Cite this article
Coombs, T.L. The nature of zinc and copper complexes in the oyster Ostrea edulis . Marine Biology 28, 1–10 (1974). https://doi.org/10.1007/BF00389111
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00389111