Abstract
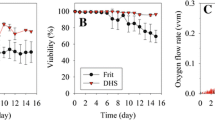
Serum-free perfusion cultures of hybridoma TO-405 cells were carried out in spinner flasks coupled with zeolite A-3 packed beads. Ammonia was selectively removed from the culture broth by passing cell free permeate from ceramic cross flow filtration, through the zeolite packed bed. Ammonia concentration in the culture broth was effectively maintained between 1 to 4 mmol/l which was below the inhibitory concentration for cell growth. Maximum cell density levels of 107 cells/ml as well as improved percentage cell viability higher than in serum-supplemented cultures were feasible in this system.
The possible effects of shear stress, generated by variation of the flow rates of the broth through the ceramic filter module, on the growth of the hybridoma cells were investigated. Backwashing, by reversing the direction of the permeate, was found necessary to prolong the life of the filter. Variation of the flow rates of the broth through the ceramic module between 0.29 m/s to 0.59 m/s did not cause immediate cell damage but growth was repressed at the higher flow rate.
This study also showed that glutamine appears to be one of the factors limiting the growth of the hybridoma cells.
Similar content being viewed by others
References
BelfortG (1989) Membrane and bioreactors: A technical challenge in biotechnology. Biotechnol. Bioeng. 33: 1047–1066.
ButlerM and JerkinsH (1989) Nutritional aspects of the growth of animal cells in culture. J. Biotechnol. 12: 97–110.
ButlerM (1985) Growth and limitation in high density microcarrier cultures. Dev. Biol. Stand. 60: 269–280.
ButlerM (1983) Growth limitations in microcarrier cultures. In: FiechterA (ed.), Advances in biochemical engineering/biotechnology, Vol. 43. (p. 7–84) Springer-Verlag, Berlin.
DoyleC and ButlerM (1990) The effect of pH on the toxicity of ammonia to a murine hybridoma. J. Biotechnol. 15: 91–100.
EagleH (1958) Amino acid metabolism in mammalian cell cultures. Science 130: 432–437.
EagleH (1955) Nutritional needs of mammalian cells in tissue culture. Science 122: 501–504.
GeaugeyV, DuvalD, GeahelA, MarcA and EngasserJM (1989) Influence of amino acids on hybridoma cell viability and antibody secretion. Cytotechnology 2: 119–129.
GlackenMW, FleischakerRJ and SinskeyAJ (1986) Reduction of waste product excretion via nutrient control: possible strategies for maximizing product and cell yields in cultures of mammalian cells. Biotechnol. Bioeng. 28: 1376–1389.
GriffithsJB (1973) The effects of adapting human diploid cells to grow in glutamic acid media on cell morphology, growth and metabolism. J. Cell Sci. 12: 617–629.
GriffithsJB (1970) The quantitative utilization of amino acids and glucose and contact inhibition of growth in cultures of the human diploid cell, WI-38. J. Cell Sci. 6: 739–749.
GriffithsJB and PirtSJ (1967) The uptake of amino acids by mouse cells (strain LS) during growth in batch culture and chemostat culture: the influence of cell growth rate. Proc. Roy. Soc. B. 168: 421–438.
HosoiS, MiohH, AnzaiC, SatoS, and FujiyoshiN (1988) Establishment of Navalva cell lines which grow continuously in glutamine-free medium. Cytotechnology 1: 151–158.
KuranoN, LeistC, MessiS, KuranoS, and FiecherA (1990) Growth behavior of chinese hamster ovary cells in a compact loop biorector. 2. Effects of medium components and waste products. J. Biotechnol. 15: 113–128.
LeMS and AtkinsonT (1985) Cross-flow microfiltration of extracellular products. Process Biochem. 26: 26–31.
McCartyK (1962) Selective utilization of amino acids by mammalian cell cultures. Exp. Cell Res. 27: 230–240.
MatsumuraM, MisatoT, KataokaH, and MayumiM (1989) Production of HBs-MAbs by perfusion culture in membrane reactor. In: MurakamiH (ed.) Trends in Animal Cell Technology (pp. 145–148) Kodansha Ltd., Tokyo.
MatsumuraM and MarklH (1986) Elimination of inhibition by perstraction. Biotechnol. Bioeng. 27: 534–541.
ShermanJP (1978) Ion exchange separation with molecular sieve zeolites. Am. Inst. Chem. Engr. Symp. Series 74: 98–116.
RobertsRS, HsuHW, LinKD and YangTJ (1976) Amino acid metabolism of myeloma cells in culture. J. Cell Sci. 21: 609–615.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Nayve, F.R.P., Motoki, M., Matsumura, M. et al. Selective removal of ammonia from animal cell culture broth. Cytotechnology 6, 121–130 (1991). https://doi.org/10.1007/BF00373029
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00373029