Abstract
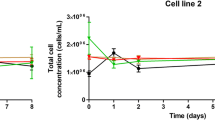
Cell cycle, cell size and rhodamine 123 fluorescence in cell populations of two batch cultures were analysed and quantified with a fluorescence-activated cell sorter (FACS). Two cultures derived from either exponential or stationary phase innocula were investigated in order to demonstrate the dependency of the subsequent cell growth on innoculum condition. The results demonstrated that the level of activity of cells in the innoculum culture could have a significant effect on cellular activity during the initial phase of the inoculated culture, as it advances through its growth cycle. Positive correlation was found between the cell size and mitochondrial activity (as measured by rhodamine 123 uptake) with S and G2 fractions as the cell progressed through the cell cycle. The enumeration of the fractions of cell cycle phases has helped in prediction of the changes in cell numbers following perturbation of the culture condition.
Similar content being viewed by others
References
Al-Rubeai M, Chalder S, Emery AN and Bird R (1991a) Monitoring of animal cell culture by flow cytometry. Chimica Oggi 9: 36–40.
Al-Rubeai M and Emery AN (1990) Mechanisms and Kinetics of monoclonal antibody synthesis and secretion in synchronous and asynchronous hybridoma cell cultures. J. Biotechnol. 16: 67–86.
Al-Rubeai M, Kloppinger M, Fertig G, Emery AN and Miltenburger HG (1991b) Monitoring of biosynthetic and metabolic activity in animal cell culture using flow cytometric methods. Proceedings of ESACT meeting, 2–6 September, Brighton.
Baserga R (1985) The biology of cell reproduction. Harvard University Press, Massachusetts.
Benel L, Ronot X, Kornprobst M, Adolphe M and Mounolou JC (1986) Mitochondrial uptake of rhodamine 123 by rabbit articular chondrocytes. Cytometry 7: 281–285.
Darzynkiewicz Z, Crissman H, Traganos F and Steinkamp J (1982) Cell heterogeneity during the cell cycle. J. Cell. Physiol. 113: 465–474.
Ernster L and Schatz G (1981) Mitochondria: A historical review. J. Cell Biol. 91: 2275–2555.
Johnson LV, Walsh ML and Chen LB (1980) Localization of mitochondria in living cells. Proc. Natl. Acad. Sci. USA 77: 990–994.
Luan YT, Mutharasan R and Magee WE (1987) Strategies to extend longevity of hybridomas in culture and promote yield of monoclonal antibodies. Biotechnology Letters 9: 691–696.
Kloppinger M, Fertig G, Fraune E and Miltenburger HG (1989) Flow cytometric process monitoring of hybridoma cells in batch and perfusion culture. In: Spier RE, Griffiths JB, Stephenne J, and Crooy PJ (eds.) Advances in Animal Cell Biology and Technology for Bioprocesses (pp. 125–128). Butterworths, London.
Oh SKW, Nienow AW, Al-Rubeai M and Emery AN (1989). The Effects of agitation intensity with and without continuous sparging on the growth and antibody production of hybridoma cells. J. Biotechnol. 12: 45–62.
Ormerod MG (1990) Flow Cytometry, a practical approach. IRL Press, Oxford University press.
Ozturk S and Palsson B (1990) Effect of initial cell density on hybridoma growth, metabolism, and monoclonal antibody production. J. Biotechnol. 16: 259–278.
Ramirez OT and Mutharasan R (1990) Cell cycle and growth phase-dependent variations in size distribution, antibody productivity and oxygen demand in hybridoma cultures. Biotechnol. Bioeng. 36: 839–848.
Ronot X, Benel L, Adolphe M and Mounolou J-C (1986) Mitochondrial analysis in living cells: The use of rhodamine 123 and flow cytometry. Bio. Cell 57: 1–8.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Al-Rubeai, M., Chalder, S., Bird, R. et al. Cell cycle, cell size and mitochondrial activity of hybridoma cells during batch cultivation. Cytotechnology 7, 179–186 (1991). https://doi.org/10.1007/BF00365929
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00365929