Abstract
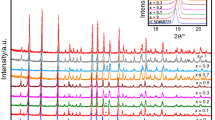
A series of samples in the BaxLa1−xFeO3−ysystem (x=0.00, 0.25, 0.50, 0.75, and 1.00) have been prepared at 1200°C under an atmospheric air pressure. The solid solutions of the system were analysed from the X-ray diffraction spectra and thermal analyses. X-ray diffraction studies assigned the compositions of the x=0.00 and 1.00 to the orthorhombic system and the compositions of the x=0.25, 0.50, and 0.75 to the cubic system. The reduced lattice volume increased with the x value in the system. The mole ratios of the Fe4+ ion in the solid solutions, or τ values, were determined by Mohr salt analyses and the non-stoichiometric chemical formulae of the system were formulated from the x, τ, and y values. From the results of the Mössbauer spectroscopy, the coordination and the magnetic property of the iron ion have been discussed. The electrical conductivities were measured as a function of temperature under atmospheric air pressure. The activation energy was minimum at the composition of x=0.50. The conduction mechanism can be explained by the hopping model between the mixed valences of the Fe3+ and Fe4+ ions.
Similar content being viewed by others
References
C. H. Yo, K. S. Roh, S. J. Lee, K. H. Kim and E. J. Oh J. Kor Chem. Soc. 35 (1991) 211.
C. H. Yo, E. S. Lee and M. S. Pyun, J. Solid State Chem. 73 (1988) 411.
C. H. Yo, S. J. Lee and S. H. Chsng, J. Nat. Soc. Res. Inst. 12 (1983) 65.
B. Buffat, G. Demazeau, M. Pouchard, J. M. Dance and P. Hagenmuller, J. Solid State Chem. 50 (1983) 33.
J. B. Goodenough, J. Phys. Chem. Solids 6 (1958) 287.
V. G. Bhide, D. S. Rajoria, G. Rama Rao and C. N. R. Rao, Phys. Rev. B 6 (1972) 1021.
M. Eibschütz, S. Shtrikman and D. Treves, Phys. Rev. 156 (1967) 562.
C. Bockema, F. Vander Woude and G. A. Sawatzky, Phys. Rev. B 11 (1975) 2705.
J. Mizusaki, T. Sasamoto, W. R. Cannon and H. K. Bowen, J. Am. Ceram. Soc. 65 (1982) 363.
Idem, ibid. 66 (1983) 247.
M. Parras, M. Vallet-Regi, J. M. Gonzalez-Calbet and J. C. Grenier, J. Solid State Chem. 83 (1989) 121.
P. K. Gallagher, J. B. MacChesney and D. N. E. Buchanamn, J. Chem. Phys. 43 (1965) 516.
H. J. Richtler and K. A. Hempel, J. Appl. Phys. 64 (1988) 5980.
J. M. Gonzalez-Calbet, M. Parras, M. Vallet-Regi and J. C. Grenier, J. Solid State Chem. 86 (1990) 149.
A. J. Jacobson, Acta. Crystallogr. B32 (1976) 1087.
P. D. Battle, T. C. Gibb, P. Lightfoot and M. Matsuo, J. Solid State Chem. 85 (1990) 38.
M. Parras, M. Vallet-Regi, J. M. Gonzalez-Calbet, M. Alzrio-Franco and J. C. Grenier, ibid. 74 (1988) 110.
I. Shaplygin, Ya Shubrt and A. Zakharov, Russ. J. Inorg. Chem. (English Translation) 31 (1986) 1074.
A. Wattiaux, J. C. Grenier, M. Pouchard and P. Hagenmuller, J. Electrochem. Soc. 134 (1987) 1718.
T. C. Gibb and M. Matsuo, J. Solid State Chem. 81 (1989) 83.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Roh, K.S., Ryu, K.H. & Yo, C.H. Nonstoichiometry and physical properties of the perovskite BaxLa1−xFeO3−y system. JOURNAL OF MATERIALS SCIENCE 30, 1245–1250 (1995). https://doi.org/10.1007/BF00356126
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF00356126