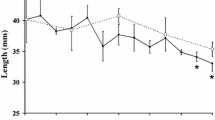
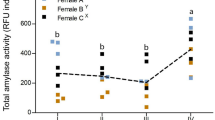
Abstract
Postlarval lobsters Homarus americanus Milne Edwards hatched from three females collected in 1989 from Block Island Sound, Rhode Island were reared individually in the laboratory under nine treatment combinations of temperature (15, 18 and 21°C) and feeding (starved, low ration and full ration). Total RNA, DNA (mgind.-1), RNA:DNA ratios and molt stage were determined for individuals sampled at daily intervals. Postlarval lobsters had high resistance to starvation. A majority of the lobsters survive 12 d of food deprivation, with some surviving up to 24–29 d. During starvation, cell biomass (estimated from protein:DNA) decreased to a minumum size, whereas cell number (based on total DNA) was generally conserved. The molt cycle was arrested at molt stage C in the starved postalrvae. Instar duration was inversely related to temperature. However, the duration of the postlarval instar did not differ between the low and full ration treatments. Uncoupling of cell growth and the molt cycle was evident in the full and low ration treatments. In the full ration treatments, the postmolt through early premolt was characterized by a rapid increase in total RNA and DNA. Maximum cell biomass was attained by molt stage C or D0. In the low ration treatments, total RNA and DNA were less than those in the full ration treatments and the maximum cell biomass was attained only at molt stage D2-3. Notably, different feeding regimes resulted in different patterns in RNA:DNA ratios over the molt cycle during the postlarval instar. RNA:DNA ratio was a sensitive indicator of recent (2 to 3 d) food deprivation. However, this ratio was positively related to the level of feeding only at molt stages C to D1 and was inversely related to temperature regime. These results indicate that the use of RNA:DNA ratios to estimate the relative nutritional state of postlarval tobsters must be qualified with respect to the period of the molt cycle and the temperature regime.
Similar content being viewed by others
Literature cited
Aiken, D. E., Waddy, S. L. (1986). Environmental influence on recruitment of the American lobster Homarus americanus: a perspective. Can J. Fish. aquat. Sciences 43: 2258–2270
Anger, K. (1984). Influence of starvation on moult cycle and morphogenesis of Hyas araneus larvae (Decapoda, Majidae). Helgoländer Meeresunters. 38: 21–33
Anger, K. (1987). The D0 threshold: a critical point in the development of decapod crustaceans. J. exp. mar. Biol. Ecol 108: 15–30
Anger, K., Dawirs, R. R. (1981). Influence of starvation on the larval development of Hyas araneus (Decapoda, Majidae). Helgoländer Meeresunters. 34: 287–311
Anger, K., Harms, J., Puschel, C., Seeger, B. (1989). Physiological and biochemical changes during the larval development of a brachyuran crab reared under constant conditions in the laboratory. Helgoländer Meersunters. 43: 225–244
Anger, K., Hirche, H.-J. (1990). Nucleic acids and growth of larval and early juvenile spider crab, Hyas araneus. Mar. Biol. 105: 403–411
Anger, K., Spindler, K.-D. (1987). Energetics, moult cycle and ecdysteroid titers in spider crab (Hyas araneus) larvae starved after the D0 threshold. Mar. Biol. 94: 367–375
Anger, K., Storch, V., Anger, V., Capuzzo, J. M. (1985). Effects of starvation on moult cycle and hepatopancreas of stage I lobster (Homarus americanus) larvae. Helgoländer Meeresunters. 39: 107–116
Buckley, L. J. (1979). Relationships between RNA-DNA ratio, prey density and growth rate in the Atlantic cod (Gadus morhua). J. Fish. Res. Bd Can. 36: 1497–1502
Buckley, L. J. (1984). RNA-DNA ratio: an index of larval fish growth in the sea. Mar. Biol. 80: 291–298
Buckley, L. J., Lough, R. G. (1987). Recent growth, biochemical composition and prey field of larval haddock (Melanogrammus aeglefinus) and Atlantic cod (Gadus morhua) on Georges Bank. Can. J. Fish. aquat. Sciences 44:14–25
Bulow, F. J. (1987). RNA-DNA ratios as indicators of growth in fish: a review. In: Summerfelt, R. C., Hall, G. E. (eds.) The age and growth of fish. Iowa State University Press, Iowa, p. 45–64
Capuzzo, J. M., Lancaster, B. A. (1979). Some physiological and biochemical considerations of larval development in the American lobster, Homarus americanus Milne Edwards. J. exp. mar. Biol. Ecol. 40: 53–62
Carlberg, J. M., Van Olst, J. C. (1976). Brine shrimp (Artemia salina) consumption by the larval stages of the American lobster (Homarus americanus) in relation to food density and water temperature. Proc. Wld. Maricult. Soc. 7: 379–389
Castell, J. D., Kean, J. C. (1986). Evaluation of the role of nutrition in lobster recruitment. Can. J. Fish. aquat. Sciences 43: 2320–2327
Cronin, T. W., Forward, R. B. (1980). The effects of starvation on phototaxis and swimming of larvae of the crab, Rhithropanopeus harrisii. Mar. Biol. 17: 283–294
Dagg, M. J., Littlepage, J. L. (1972). Relationships between growth rate and RNA, DNA, protein and dry weight in Artemia salina and Euchaeta elongatal. Mar. Biol. 17: 162–170
Dawirs, R. R. (1984). Influence of starvation on larval development of Carcinus maenas L. (Decapoda: Portunidae). J. exp. mar. Biol. Ecol. 80:47–66
Eagles, M. D., Aiken, D. E., Waddy, S. E. (1986). Influence of light and food on larval lobsters, Homarus americanus. Can. J. Fish. aquat. Sciences 43: 2303–2310
Fraser, A. J. (1989). Triacylglycerol content as a condition index for fish, bivalve and crustacean larvae. Can. J. Fish. aquat. Sciences 46: 1868–1873
Harding, G. C., Drinkwater, K. F., Vass, W. P. (1983). Factors influencing the size in American lobster (Homarus americanus) stocks along the Atlantic coast of Nova Scotia, Gulf of St. Lawrence, and Gulf of Maine: a new synthesis. Can. J. Fish. aquat. Sciences 39: 1117–1129
Hartnoll, R. C. (1982). Growth In: Abele, L. G. (ed.) The biology of crustacea-embryology, morphology and genetics. Academic. Press, New York, p. 111–196
Juinio, M. A. R., Cobb, J. S. (in preparation). RNA:DNA ratios and growth rates during the postlarval instar of Homarus americanus reared under different temperature and feeding regimes (manuscript a)
Juinio, M. A. R., Cobb, J. S. (in preparation) Development of a growth model and estimation of recent growth of field-caught postlarval lobsters from RNA:DNA ratios (manuscript b).
Logan, D. T., Epifanio, C. E. (1978). A laboratory energy balance for the larvae and juveniles of the American lobster Homarus americanus. Mar. Biol. 47: 381–389
MacKenzie, B. R. (1988). Assessment of temperature effects on interrelationships between stage durations, mortality, and growth in laboratory-reared Homarus americanus Milne Edwards larvae. J. exp. mar Biol. Ecol. 116: 87–98
McConaugha, J. R. (1985). Nutrition and larval growth. In: Wenner, A. M. (ed.) Larval growth, crustacean issues. A.A. Balkema, Boston, p. 127–154
Olson, R. R., Olson, M. H. (1989). Food limitation of planktonic marine invertebrate larvae: does it control recruitment success? A. Rev. Ecol. Syst. 20: 225–247
Ota, A., Landry, M. R. (1984). Nucleic acids as growth rate indicators for early developmental stages of Calcous pacificus Brodsky. J. exp. mar. Biol. Ecol. 80: 147–160
Paul, A. J., Paul, J. M. (1980). The effect of early starvation on later feeding success of king erab zoea. J. exp. mar. Ecol. 44: 247–251
Phillips, B. F., Sastry A. N. (1980). Larval ecology. In: Phillips, B. F. Cobb, J. S. (eds.) The biology and management of lobsters. Academic Press, New York, p. 11–57
Regnault, M., Luquet, P. (1974). Study of evolution of nucleic acid content of prepubertal growth in the shrimp Crangon vulgaris. Mar Biol 25: 291–298
Rosenberg, R., Costlow, J. D. (1979). Delayed response to irreversible non-genetic adaptation to salinity in early development of the brachyuran crab Rhithropanopeus harrisii, and some notes on adaptation to temperature. Ophelia 18: 97–112
SAS Institute Inc. (1985). SAS user's guide: statistics, version 5th Edn. SAS Inst. Inc., Cary, North Carolina
Sasaki, G. C. (1984). Biochemical changes associated with embryonic and larval development in the American lobster Homarus americanus Milne Edwards. Ph. D. thesis, Massachusetts Institute of Technology, Woods Hole Oceanographic Institute, Woods Hole, Massachusetts, WHOI-84-8
Sasaki, G. C., Capuzzo, J. M., Biesiot, P. (1986). Nutritional and bioenergetic considerations in the development of the American lobster Homarus americanus. Can. J. Fish. aquat. Sciences 43: 2311–2319
Sastry, A. N. (1980). Effects of thermal pollution on pelagic larvae of crustacea. EPA-600/3-80-064. Nat. Tech. Inf. Service, Springfield, Virginia
Scarratt, D. J. (1964). Abundance and distribution of lobster larvae (Homarus americanus) in Northumberland Strait. J. Fish. Res. Bd Can. 21: 661–680
Skinner, D. M. (1966). Macromolecular changes associated with the growth of crustacean tissues. Am. Zool. 6: 235–242
Sulkin, S. D., Morgan, R. P., Minasian, L. L. (1975). Biochemical changes during larval development of the xanthid crab Rhithropanopeus harrisii. II. Nucleic Acids. Mar. Biol. 32: 113–117
Sutcliffe, W. H. (1970). Relationship between growth rate and ribonucleic acid concentration in some invertebrates. J. Fish. Res. Bd Can. 27: 606–609
Templeman, W. (1936). The influence of temperature, salinity, light and food conditions on the survival growth of the larvae of the lobster (Homarus americanus). J. Biol. Bd Can. 2: 485–497
Thorson, G. (1950). Reproductive and larval ecology of marine bottom invertebrates. Biol. Rev. Cambridge Philos. Soc. 25: 1–45
Wang, S. Y., Stickle, W. B. (1986). Changes in nucleic acid concentration with starvation in the blue crab Callinectes sapidus Rathbun. J. Crust. Biol. (Lawrence, Kansas). 6: 49–56
Wang, S. Y., Stickle, W. B. (1988). Biochemical composition of the blue crab Callinectes sapidus exposed to water-soluble fraction of crude oil. Mar. Biol. 98:23–30
Author information
Authors and Affiliations
Additional information
Communicated by J. Grassle, New Brunswick
Rights and permissions
About this article
Cite this article
Juinio, M.A.R., Cobb, J.S., Bengtson, D. et al. Changes in nucleic acids over the molt cycle in relation to food availability and temperature in Homarus americanus postlarvae. Marine Biology 114, 1–10 (1992). https://doi.org/10.1007/BF00350850
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00350850