Abstract
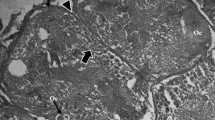
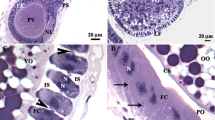
The route of egg yolk protein uptake into the oocytes of kuruma prawn, Penaeus japonicus, was studied using immunohistochemical and electron microscopical methods. Although a significant immunofluorescence with anti-vitellin-immunoglobulin was observed in the enlarged follicle cells surrounding oil globule stage oocytes of the early vitellogenic ovary, no fluorescence was detected in shrunken follicle cells surrounding oocytes in the yolk granule stage. Electron microscopically, yolk granule stage oocytes have an irregular surface with numerous well-developed microvilli. In contrast, the surface of follicle cells is relatively smooth. The irregular surface of yolk granule stage oocytes was covered with a layer of electron dense material. Similar dense material was found in the spaces between the neighboring follicle cells on the yolk granule stage oocytes. The outer surface of the follicle cells on yolk granule stage oocytes was covered by dense materials which were similar to those found on the irregular surface of oocytes. Micropinocytotic vesicles containing dense material were found in the ooplasm near the irregular surface with numerous well-developed microvilli. Dense material was concentrated in the peripheral part of the small forming yolk bodies of yolk granule stage oocytes. This suggests that the electron dense material, probably egg yolk protein, transferred to the surface of yolk granule stage oocytes from the spaces between the neighboring follicle cells may be incorporated into the ooplasm by pinocytosis through the microvilli and subsequently aggregate to form yolk bodies.
Similar content being viewed by others
References
Beams HW, Kessel RG (1963) Electron microscope studies on developing crayfish oocytes with special references to the origin of yolk. J Cell Biol 18:621–649
Eastman-Reks S, Fingerman M (1985) In vitro synthesis of vitellin by the ovary of the fiddler crab, Uca pugilator. J exp Zool 233: 111–116
Fielder DR, Rao KK, Fingerman M (1971) A female-limited lipoprotein and the diversity of hemocyanin components in the dimorphic variants of the fiddler crab, Uca pugilator, as revealed by disc electrophoresis. Comp Biochem Physiol 39B: 291–297
Fyffe WE, O'Connor JD (1974) Characterization and quantification of a crustacean lipovitellin. Comp Biochem Physiol 47B: 851–867
Ganion CR, Kessel RG (1972) Intracellular synthesis, transport, and packaging of proteinaceous yolk of Orconectes immunis. J Cell Biol 52:420–437
Hinsch GW, Cone MV (1969) Ultrastructure observations of vitellogenesis in the spider crab, Libinia emarginata L. J Cell Biol 40: 336–342
Kessel RG (1968) Mechanisms of protein yolk synthesis and deposition in crustacean oocytes. Z Zellforsch 89:17–38
Khayat M, Lubzens E, Tietz A, Funkenstein B (1994) Cell-free synthesis of vitellin in the shrimp Penaeus semisulcatus (de Haan). Gen comp Endocr 93:205–213
Krol M, Hawkins WE, Overstreet RM (1992) Reproductive components. In: Harrison FW (ed) Microscopic anatomy of invertebrates. Vol. 10. Wiley-Liss, New York, pp 295–343
Lui CW, Sage BA, O'Connor JD (1974) Biosynthesis of lipovitellin by the crustacean ovary. J exp Zool 188:289–296
Quackenbush LS (1989) Yolk protein production in the marine shrimp Penaeus vannamei. J Crustacean Biol 9:509–516
Quinitio ET, Hara A, Yamauchi K, Mizushima T, Fuji A (1989) Identification and characterization of vitellin in a hermaphrodite shrimp, Pandalus kessleri. Comp Biochem Physiol 94B:445–451
Rankin SM, Bradfield JY, Keeley LL (1989) Ovarian protein synthesis in the South American white shrimp, Penaeus vannamei, during the reproductive cycle. Int J Invert Reprod 15:27–33
Schade ML, Shivers RR (1980) Structural modulation of the surface and cytoplasm of oocytes during vitellogenesis in the lobster, Homarus americanus. An electron microscope-protein tracer study. J Morph 163:13–26
Wolin EH, Laufer H, Albertini DF (1973) Uptake of the yolk protein lipovitellin by developing crayfish oocytes. Devl Biol 35:160–170
Yano I (1987) Effect of 17α-hydroxy-progesterone on vitellogenin secretion in kuruma prawn, Penaeus japonicus. Aquaculture, Amsterdam 61:49–57
Yano I (1988) Oocyte development in the kuruma prawn, Penaeus japonicus. Mar Biol 99:547–553
Yano I (1993) Ultraintensive culture and maturation in captivity of penaeid shrimp. In: McVey JP (ed) Mariculture. Vol. 1. (2nd edn) CRC Press, Boca Raton, pp 289–313
Yano I, Chinzei Y (1987) Ovary is the site of Vg synthesis in kuruma prawn, Penaeus japonicus. Comp Biochem Physiol 86:213–218
Author information
Authors and Affiliations
Additional information
Communicated by T. Ikeda, Hakodate
Rights and permissions
About this article
Cite this article
Yano, I., Krol, R.M., Overstreet, R.M. et al. Route of egg yolk protein uptake in the oocytes of kuruma prawn, Penaeus japonicus . Marine Biology 125, 773–781 (1996). https://doi.org/10.1007/BF00349260
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00349260