Summary
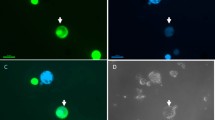
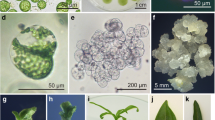
Defined cybridization was performed by one-to-one electrofusion (microfusion) of preselected protoplast-cytoplast pairs of male-fertile, streptomycin-resistant Nicotiana tabacum and cytoplasmic male-sterile, streptomycin-sensitive N. tabacum cms (N. bigelovii), followed by microculture of the fusion products until plant regeneration. Dominant selectable markers, namely, kanamycin resistance (nptII) and hygromycin B resistance (hpt) genes had been previously integrated in the nuclear genomes of the otherwise almost fully isogenic parental strains using direct gene transfer to protoplasts. In addition to chromosome counts indicating the expected allotetraploid tobacco count of 48, the absence of the nucleus from the cytoplast donor line was confirmed by Southern blot hybridization using nptII and hpt probes, as well as by an in vitro selection test with leaf expiants and the corresponding enzyme assays for 30 cybrids. The cytoplasmic composition of the cybrids obtained was analyzed for chloroplast type using the streptomycin resistance/sensitivity locus. The fate of mitochondria in cybrids was checked by species-specific patterns in Southern analysis of restriction endonuclease digests of total DNA with N. sylvestris mitochondrial DNA probes.
Similar content being viewed by others
References
Aviv D, Galun E (1987) Chondriome analysis in sexual progenies of Nicotiana cybrids. Theor Appl Genet 73:821–826
Aviv D, Arzee-Gonen P, Bleichman S, Galun E (1984) Novel alloplasmic Nicotiana plants by “donor-recipient” protoplast fusion: cybrids having N. tabacum or N. sylvestris nuclear genomes and either or both plastomes and chondriomes from alien species. Mol Gen Genet 196:244–253
Blochinger K, Diggelmann H (1984) Hygromycin B phosphotransferase as a selectable marker for DNA transfer experiments with higher eukaryotic cells. Mol Cell Biol 4:2929–2931
Böttcher UF, Aviv D, Galun E (1989) Complementation between protoplasts treated with either of two metabolic inhibitors results in somatic hybrid plants. Plant Sci 63:67–77
Bradford MM (1976) A rapid and sensitive method for the quantification of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Cabanes-Bastos E, Day AG, Lichtenstein CP (1989) A sensitive and simple assay for neomycin phosphotransferase II activity in transgenic tissue. Gene 77:169–177
Datta SK, Peterhans A, Karabi K, Potrykus I (1990) Genetically engineered fertile Indica rice recovered from protoplasts. Bio/Technol 8:736–740
Feinberg AP, Vogelstein B (1983) A technique for radiolabelling DNA restriction endonuclease fragments to high specific activity. Anal Biochem 132:6–13
Fluhr R, Aviv D, Edelman M, Galun E (1983) Cybrids containing mixed and sorted-out chloroplasts following interspecific somatic fusions in Nicotiana. Theor Appl Genet 65:289–294
Galun E, Arzee-Gonen P, Fluhr R, Edelman M, Aviv D (1982) Cytoplasmic hybridization in Nicotiana: mitochondrial DNA analysis in progenies resulting from fusion between protoplasts having different organelle constitutions. Mol Gen Genet 186:50–56
Gerstel DU (1980) Cytoplasmic male sterility in Nicotiana (a review). NC Agric Exp Stn Tech Bull 263:1–31
Gleba YY, Sytnik KM (1984) Protoplast fusion: genetic engineering in higher plants. In: Monographs on theoretical and applied genetics 8. Springer, Berlin Heidelberg New York, pp 1–220
Gleba YY, Kolesnik NN, Meshkene I, Cherep NN, Parokonny AS (1984) Transmission genetics of the somatic hybridization process in Nicotiana. 1. Hybrids and cybrids among the regenerants from cloned protoplast fusion products. Theor Appl Genet 69:121–128
Gleba YY, Komarnitsky, Kolesnik NN, Meshkene I, Martyn GI (1985) Transmission genetics of the somatic hybridization process in Nicotiana. 2. Plastome heterozygotes. Mol Gen Genet 198:476–481
Koop HU, Schweiger HG (1985a) Regeneration of plants from individually cultivated protoplasts using an improved microculture system. J Plant Physiol 121:245–257
Koop HU, Schweiger HG (1985b) Regeneration of plants after electrofusion of selected pairs of protoplasts. Eur J Cell Biol 39:46–49
Koop HU, Spangenberg G (1989) Electric field-induced fusion and cell reconstitution with preselected single protoplasts and subprotoplasts of higher plants. In: Neumann E, Sowers A, Jordan C (eds) Electroporation and electrofusion in cell biology. Plenum Press, New York London, pp 355–366
Koop HU, Dirk J, Wolff D, Schweiger HG (1983) Somatic hybridization of two selected single cells. Cell Biol Int Rep 7:1123–1128
Lichtenstein C, Draper J (1985) Genetic engineering of plants. In: Glover DM (ed) DNA cloning, vol II. IRL Press, Oxford Washington, pp 67–119
Maliga P, Breznovits A, Marton L (1973) Streptomycin-resistant plants from callus cultures of haploid tobacco. Nature 244:29–30
Maliga P, Lörz H, Lazar G, Nagy F (1982) Cytoplast-protoplast fusion for interspecific chloroplast transfer in Nicotiana. Mol Gen Genet 185:211–215
Maniatis T, Fritsch EF, Sambrook J (1982) Molecular cloning: a laboratory manual. Cold Spring Harbor Laboratory Press, Cold Spring Harbor/NY
McDonnell RE, Clark RD, Smith WA, Hinchee MA (1987) A simplified method for the detection of neomycin phosphotransferase II activity in transformed plant tissues. Plant Mol Biol Rep 5:380–386
Negrutiu I, Shillito R, Potrykus I, Biasini G, Sala F (1987) Hybrid genes in the analysis of transformation condition. Plant Mol Biol 8:363–373
Paszkowski J, Shillito RD, Saul MW, Mandak V, Hohn T, Hohn B, Potrykus I (1984) Direct gene transfer to plants. EMBO J 3:2717–2722
Paszkowski J, Baur M, Bogucki A, Potrykus I (1988) Gene targeting in plants. EMBO J 7:4021–4026
Pietrzak M, Shillito RD, Hohn T, Potrykus I (1986) Expression in plants of two bacterial antibiotic resistance genes after protoplast transformation with a new plant expression vector. Nucleic Acids Res 14:5857–5868
Potrykus I, Shillito RD (1986) Protoplasts: isolation, culture, plant regeneration. In: Weissbach E, Weissbach H (eds) Methods in Enzymology 118, Academic Press, Orlando, pp 549–578
Potrykus I, Paszkowski J, Saul MW, Petruska J, Shillito RD (1985) Molecular and general genetics of a hybrid foreign gene introduced into tobacco by direct gene transfer. Mol Gen Genet 199:167–177
Reiss B, Sprengel G, Will H, Schaller H (1984) A new sensitive method for qualitative and quantitative assay of neomycin phosphotransferase in crude cell extracts. Gene 30:217–223
Schreier PH, Seftor EA, Schell J, Bohnert HJ (1985) The use of nuclear-encoded sequences to direct the light-regulated synthesis and transport of a foreign protein into plant chloroplasts. EMBO J 4:25–32
Schweiger HG, Dirk J, Koop HU, Kranz E, Neuhaus G, Spangenberg G, Wolff D (1987) Individual selection, culture, and manipulation of higher plants cells. Theor Appl Genet 73:769–783
Sears BB (1980) Elimination of plastids during spermatogenesis and fertilization in the plant kingdom. Plasmid 4:233–255
Shillito RD, Paszkowski J, Müller M, Potrykus I (1983) Agarose plating and a bead-type culture technique enable and stimulate development of protoplast-derived colonies in a number of plant species. Plant Cell Rep 2:244–247
Spangenberg G, Schweiger HG (1986) Controlled electrofusion of different types of protoplasts including cell reconstruction in Brassica napus. Eur J Cell Biol 41:51–56
Spangenberg G, Koop HU, Lichter R, Schweiger HG (1986) Microculture of single protoplasts of Brassica napus. Physiol Plant 66:1–8
Spangenberg G, Osusky M, Oliveira MM, Freydl E, Nagel J, Pais MS; Potrykus I (1990) Somatic hybridization by microfusion of defined protoplast pairs in Nicotiana: morphological, genetic, and molecular characterization. Theor Appl Genet 80: 577–587
Author information
Authors and Affiliations
Additional information
Communicated by P. Maliga
Rights and permissions
About this article
Cite this article
Spangenberg, G., Freydl, E., Osusky, M. et al. Organelle transfer by microfusion of defined protoplast-cytoplast pairs. Theoret. Appl. Genetics 81, 477–486 (1991). https://doi.org/10.1007/BF00219437
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00219437