Summary
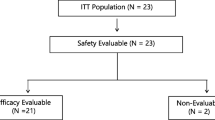
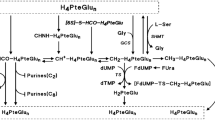
N-(phosphonacetyl)-disodium L-aspartic acid (PALA) demonstrates a synergistic antitumor effect when combined with 5-Fluorouracil (5-FU) in in vitro studies. In a Phase II trial, 23 eligible patients with unresectable or metastatic adenocarcinoma of the stomach were treated with weekly i.v. bolus PALA (250 mg/M2) followed 24 hours later by a 24-hour infusion of 5-FU (2600 mg/M2) for an initial period of 8 weeks. No objective responses were noted. PALA and 5-FU is inactive against gastric adenocarcinoma at the doses and schedule used in this trial.
Similar content being viewed by others
References
Grem JL, King SA, O'Dwyer PJ, Leyland-Jones B: Biochemistry and clinical activity of N-(phosphonacetyl)-L-aspartate: a review. Cancer Res 48:4441–4454, 1988
O'Dwyer PJ: Biochemical modulation of 5-Fluorouracil by PALA. Cancer Invest 8:261–262, 1990
Wils J: The treatment of advanced gastric cancer. Semin Oncol 23:397–406, 1996
Ardalan B, Singh G, Silberman H: A randomized phase I and II study of short-term infusion of high-dose fluorouracil with or without N-(phosphonacetyl)-L-aspartic acid in patients with advanced pancreatic and colorectal cancers. J Clin Oncol 6:1053–1058, 1988
Miller, AB, Hoogstraten B, Staquet M, Winkler A: Reporting results of cancer treatment. Cancer 47:207–214, 1981
O'Dwyer PJ, Paul AR, Walczak J, Weiner LM, Litwin S, Comis RL: Phase II study of biochemical modulation of fluorouracil by low dose PALA in patients with colorectal cancer. J Clin Oncol 8:1497–1503, 1990
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Martino, R.L., Fleming, T.R., Morrell, L.M. et al. Phase II trial of low-dose N-(phosphonacetyl)-disodium L-aspartic acid and high-dose 24-hour infusional 5-Fluorouracil in advanced gastric adenocarcinoma. Invest New Drugs 14, 419–421 (1996). https://doi.org/10.1007/BF00180821
Issue Date:
DOI: https://doi.org/10.1007/BF00180821