Summary
The cytotoxic and cytokinetic effects of ICRF-159 and its d-enantiomer ICRF-187 have been examined in vitro. The effects of both agents were identical. Cytotoxicity is dependent on both the drug concentration and the duration of drug exposure. Drug exposure for twice the cell cycle time is necessary for maximum effect. Cytotoxicity is also dependent upon the rate of cell proliferation. A rapidly growing cell population is more sensitive to brief drug exposure than a slowly growing population.
The cytokinetic effects were studied using flow cytometry, determination of [3H]-thymidine incorporation and mitotic index. ICRF-159/187 appears to act only during the G2 phase of the cell cycle. There is no detectable delay in cell passage through the G1/S boundary or in transit through S phase. Inhibition of DNA synthesis occurs only after the G2 block prevents subsequent entry of cells in S phase. A fraction of the cells, depending upon drug concentration, undergo further DNA synthesis without cell division, resulting in a tetrapoid cell population.
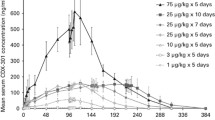
The cytokinetic effects were determined in the bone marrow of patients receiving ICRF-187. All dose-rates produced G2/M accumulation in the marrow with depletion of S phase cells. One patient was given a single injection of 1.0 gm/M2. G2/M accumulation was observed 24 h after treatment, with recovery to a pretreatment DNA cycle distribution 24 h later.
These studies suggest that a continuous drug infusion, or intermittent infusions timed to allow the normal cell population to recover, may produce superior clinical activity with this agent. A Phase I study of such an intermittent schedule is indicated.
Similar content being viewed by others
References
Bakowski MT: ICRF-159, (±) 1,2-di (3,5-dioxopiperazin-1-Y1) propane NSC-129943; Razoxane. Cancer Treat Rev 3:95–107, 1976
Bellet RE, Rozencweig M, Von Hoff DD, Penta JS, Masserman TH, Muggia FM: ICRF-159: Current status and clinical prospects. Eur J Cancer 13:1293–1298, 1977
Atherton HA, Janes SE, Hellmann K: Inhibition of growth of lung metastases with combined radiation and ICRF-159. (Abstract) Br J Cancer 34:315, 1976
Peters LJ: Modification of the radiocurability of a syngeneic murine squamous carcuinoma by its site of growth by electron-affinic drugs, and by ICRF-159. Br J Radiol 49:709–715, 1976
Wampler GL, Speckhart BJ, Regelson W: Phase I clinical study of adriamycin-ICRF-159 combination and other ICRF-159 drug combinations. (Abstract) Proc Am Assoc Cancer Res 15:189, 1974
Woodman BJ, Venditti JM, Schepartz SA, Kline I: ICRF-159 (1,2-Bis(3,5-Dioxopiperazin-1-YL)-Propane) (NSC-129943). Activity against intracerebrally inoculated L1210; therapeutic superiority against IP S1210 in combination with cis-platinum (II) Diamminodichloride (Cis-PT-II: NSC-119875). (Abstract) Proc Am Assoc Cancer Res 12:24, 1974
Hill HB, Hellman K: Razoxane and methotrexate resistance. Lancet 1:47, 1977
Hallowes RC, West DG, Hellman K: Cumulative cytostatic effect of ICRF-159. Nature 247:487–490, 1974
Hellman K, Field EO: Effect of ICRF-159 on the mammalian cell cycle: significance for its use in cancer chemotherapy. J Natl Cancer Inst 44:539–543, 1970
Sharpe HBA, Field EO, Hellman K: Mode of action of the cytostatic agent “ICRF-159”. Nature 226:524–526, 1970
Creighto AM, Birnie GD: Biochemical studies on growth-inhibitory Bisdioxopiperazines. I. Effect on DNA, RNA and protein synthesis in mouse-embryo fibro-blasts. Int J Cancer 5:47–54, 1970
DeCosse JJ, Gelfant S: Effects of nitrogen mustard during the cell cycle of the Ehrlich ascites tumor. Exp Cell Res 60:185–190, 1970
Drewinko BJ, Barlogie B: Age-dependent survival and cellcycle progression of cultured cells exposed to chemotherapeutic drugs. Cancer Treat Rep 60:1707–1717, 1976
Tobey RA: A simple, rapid technique for determination of the effects of chemotherapeutic agents on mammalian cellcycle traverse. Cancer Res 32:309–316, 1972
Barlogie G, Drewinko B, Johnston DA, Freireich EJ: The effect of adriamycin on the cell cycle traverse of a human lymphoid cell line. Cancer Res 36:1975–1979, 1976
Barranco SC, Humphrey RM: The effects of bleomycin on survival and cell progression in Chinese hamster cells in vitro. Cancer Res 31:1218–1223, 1971
Barranco SC, Luce SK, Ramsdahl MW, Humphrey RM: Bleomycin as a possible synchronizing agent for human tumor cells in vivo. Cancer Res 33:882–887, 1973
Costanzi JJ, Loukas D, Gagliano RG, Griggiths C and Barranco S: Intravenous bleomycin infusion as a potential synchronizing agent in human disseminated malignancies. Cancer 38:1503–1506, 1976
Tobey RA: Different drugs arrest cells at a number of distinct stages in G2. Nature 254:245–247, 1975
Creaven PJ, Allen LM, Alford DA: The bioavailability in man of ICRF-159, a new oral antineoplastic agent. J Pharm Pharmacol 27:914–918, 1975
Repta AJ, Baltezor MJ, Bansal PC; Utilization of an enantiomer as a solution to a pharmaceutical problem: application to solubilization of 1,2-di(4-piperazine-2,6-dione)propane. J Pharm Sci 65:238–242, 1976
Burton KA: Study of the conditions and mechanism of the Diphenylamine reaction for the colorimetric estimation of Deoxyribonucleic acid. Biochem J 62:315–323, 1956
Krishan A: Rapid flow cytofluorometric analysis of mammalian cell cycle by propidium iodide staining. J Cell Biol 66:188–193, 1975
Dawson KM: Studies on the stability and cellular distribution of dioxopiperazines in cultured BHJ-21S cells. Biochem Pharmacol 24:2249–2253, 1975
Sadee W, Staroscik J, Finn C, Cohen J: Determination of (±)-1,2-Bis(3,5-dioxopiperazin-yl) Propane plasma levels in rats, rabbits and humans by GLC and mass fragmentography. J Pharm Sci 64:998–1001, 1975
Creaven PH, Cohen MH, Hansen HH, Selawry OS, Taylor SG, III: Phase I clinical trial of a single-dose and two weekly schedules of ICRF-159 (NSC-129943). Cancer Chemother Rep 58:393–400, 1974
Blackett NM, Adams K: Cell proliferation and the action of cytotoxic agents on haemopoietic tissue. Br J Haematol 32:751–758, 1972
Taylor IW, Bleehen NM: Changes in sensitivity to radiation and ICRF-159 during the life of monolayer cultures of EMT6 tumor line. Br J Cancer 35:587–594, 1977
Grieder A, Maurer R, Stahelin H: Comparative study of early effects of epipodophyllotoxin derivatives and other cytostatic agents on mastocytoma cultures. Cancer Res 37:2998–3005, 1977
Stephens TC, Creighton AM: Mechanism of action studies with ICRF-159: effects on the growth and morphology of BHK-21S cells. Br J Cancer 239:99–100, 1974
Taylor JW, Bleehen NM: Razoxane-induced polyploidy. Br J Cancer 38:143–147, 1978
Wheeler GP, Bowdon BJ, Adamson DJ, Vail MH: Effect of certain nitrogen mustards upon progression of cultured HEp-2 cells through the cell cycle. Cancer Res 30:100–111, 1970
Kim SH, Kim JH: Lethal effect of adriamycin on the division cycle of HeLa cells. Cancer Res 32:323–325, 1976
Sandbert-Wollheim M, Yarbro JW, Kennedy BJ: Effect of mithramycin on HeLa cells. Cancer 21:22–25, 1968
Tobey RA: Effects of cytosine arabinoside, daunomycin, mithramycin, azacytidine, adriamycin and camptothecin on mammalian cell cycle traverse. Cancer Res 32:2720–2725, 1972
Tobey RA, Crissman HA: Use of flow microfluorometry in detailed analysis of effects of chemical agents on cell cycle progression. Cancer Res 32:2726–2732, 1972
Tobey RA, Peterson DF, Anderson EC: Biochemiostry of G2 and mitosis. In: R Baserga (ed): The Cell Cycle and Cancer. Marcel Dekker, Inc., New York, pp 309–354
White K, Creighton AM: Studies of resistance to ICRF-159 in cell line BS/159–1. Br J Cancer 36:421, 1978
Gosalvey M, Pezzi L, Vivero C: Inhibition of capping of surface immunoglobulins at femtomolar concentrations of adriamycin, compound ICRF-159 and tetrodotoxin. Biochem Soc Trans 6:659–661, 1978
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Wheeler, R.H., Clauw, D.J., Natale, R.B. et al. The cytokinetic and cytotoxic effects of ICRF-159 and ICRF-187 in vitro and ICRF-187 in human bone marrow in vivo . Invest New Drugs 1, 283–295 (1983). https://doi.org/10.1007/BF00177411
Issue Date:
DOI: https://doi.org/10.1007/BF00177411