Summary
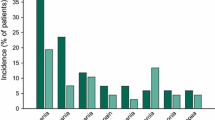
Mitoxantrone (Novantrone®), is an anthracenedione which in preclinical studies demonstrated a spectrum of antitumor activity similar to the anthracyclines, but with less cardiotoxicity. Novantrone is a cytotoxic agent that produces dose-dependent myelosuppression. When administered to patients intravenously every three weeks, white blood cell (WBC) and platelet nadirs occurred between days 8 and 15 with hematologic recovery by day 22. In multiple clinical trials in over 4450 patients, including 372 patients in randomized trials against Adriamycin, Novantrone was consistently associated with a reduced incidence of moderate and severe acute side-effects. In four randomized trials the adverse experience profile associated with Novantrone was superior to that of Adriamycin with statistically significant lower incidences of mucositis/stomatitis, nausea, vomiting and alopecia. Novantrone was less cardiotoxic than Adriamycin and cardiac events were rare in patients without predisposing risk factors. The high level of activity combined with improved patient tolerance and decreased toxicity make Novantrone a promising agent for patients requiring cytotoxic chemotherapy.
Similar content being viewed by others
References
Tormey DC: Adriamycin (NSC-123127) in breast cancer: an overview of studies. Cancer Chemother Rep (Part 3) 6(2):319–327, 1975
Carter SK: Integration of chemotherapy into combined modality treatment of solid tumors. Cancer Treat Rep 3:141–174, 1976
Bristow MR, Thomson PD, Martin RP, Mason JW, Billingham ME, Harrison DC: Early anthracycline cardiotoxicity. Am J Med 65:823–832, 1978
Smith IE, Stuart-Harris R, Pavlides N, Bozek T: Mitoxantrone (Novantrone) as a single agent and in combination chemotherapy in the treatment of advanced breast cancer. Cancer Treat Rep 10 (Suppl B):37–41, 1983
Coltman CA Jr, McDaniel TM, Balcerzak SP, Morrison FS, Von Hoff DD: Mitoxantrone hydrochloride in lymphoma, Cancer Treat Rep 10 (Suppl B):73–77, 1983
Prentice HG, Robbins G, Ma DDF, Ho AD: Sequential studies on the role of mitoxantrone in the treatment of acute leukemia. Cancer Treat Rep 10 (Suppl B):57–65, 1983
Henderson BM, Dougherty WJ, James VC, Tilley LP, Noble JF: Safety assessment of a new anticancer compound, mitoxantrone, in beagle dogs: Comparison with doxorubicin. I Clinical observations. Cancer Treat Rep 66:1139–1143, 1982
Adamson RH: Daunomycin (NSC-8215) and Adriamycin (NSC-123127) hypothesis concerning antitumor activity and cardiotoxicity. Cancer Chemother Rep 58 (1):293–294, 1974
Data on file, American Cyanamid Company, Lederle Laboratories Division, Pearl River, NY
MacCara ME: Extravasation: A hazard of intravenous therapy. Drug Intell and Clin Pharm 17:713–171, 1983
Cadman E: Toxicity of chemotherapy agents. In FE Becker (ed): Cancer, a Comprehensive Treatise. Plenum Press, 1977, chapter 3
Henney JE, Von Hoff DD, Rozencweig M, Muggia FM: Thrombophlebitic potential of intravenous cytotoxic agents. Drug Intell and Clin Pharm 11:266–267, 1977
Von Hoff DD, Rozencweig M, Slavik M, Muggia FM: Daunomycin induced cardiotoxicity in children and adults. Am J Med 62:200–206, 1967
Saltiel E, McGuire W: Doxorubicin (Adriamycin) cardiomyopathy: A critical review. Western J Med 139:332–341, 1983
Double JC, Brown JR: Evaluation of the binding of some anthraquinones and napthacenequinones to DNA. J Pharm Pharmacol 28:116–169, 1976
Goodman J, Hochstein P: Generation of free radicals and lipid peroxidation by redox cycling of Adriamycin and daunomycin. Biochem Biophys Res Commun 77(2):797–803, 1977
Kharasch ED, Novak RF: Anthracenedione activation by NADPH — cytochrome P-450 reductase; comparison with anthracyclines. Biochem Pharmacol 30(20):2881–2884, 1981
Sparano BM, Gordon G, Hall C, Iatropoulos MJ, Noble JF: Safety assessment of the new anticancer compound, mitoxantrone, in beagle dogs: a comparison with doxorubicin II. Histologic and ultrastructural pathology. Cancer Treat Rep 66(5):1145–1158, 1982
Von Hoff DD, Layard MW, Basa P, Davis HL Jr, Von Hoff AL, Rozencweig M, Muggia FM: Risk factors for drug induced congestive heart failure. Ann Int Med 91:710–717, 1979
Alexander J, Dainak N, Berger HJ, Goldman L, Johnstone D, Reduto L, Duffy T, Schwartz P, Gottschalk A, Faret BL: Serial assessment of doxorubicin cardiotoxicity with quantitative radionuclide angiography. N Eng J Med 300:278–283, 1979
Cohen LF, Glaubiger DL, Kann HE, Kohn KW: Protein associated DNA single strand breaks and cytotoxicity of dihydroxyanthracenedione (DHAD) NSC-301739 in mouse L1210 leukemia cells. Proc Am Assoc Cancer Res 21:277, 1980
Benjamin RS, Chawla SP, Ewer MS, Carrasco CH, Mackay B, Haynie III TP, Yap HY, Blumenschein GR, Holmes F, Ali MK, Bodey GP: Evaluation of mitoxantrone cardiac toxicity by nuclear angiography and endomyocardial biopsy. ASCO Abstracts C-156, May 1984
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Posner, L.E., Dukart, G., Goldberg, J. et al. Mitoxantrone: an overview of safety and toxicity. Invest New Drugs 3, 123–132 (1985). https://doi.org/10.1007/BF00174159
Issue Date:
DOI: https://doi.org/10.1007/BF00174159