Summary
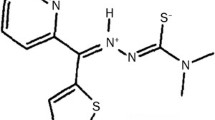
Batracylin (NSC 320846) is a water insoluble, solid tumor active compound discovered by the Developmental Therapeutics Program of the National Cancer Institute (NCI). In vivo, the NCI found this compound to be highly active [median treated tumor mass/median control tumor mass (T/C) = 0 to 20%] both orally and intraperitoneally against colon 38. In a disk diffusion, soft agar colony formation assay (500 ug/disk), we found solid tumor selectively (compared to leukemia L1210) against colon adenocarcinoma 38 (0–170 zu∶L1210 leukemia; > 950 zu∶C8), colon adenocarcinoma 9 (0–170 zu∶L1210; > 950 zu∶C9), colon adenocarcinoma 7/A (0–170 zu∶L1210; 250–400 zu∶C7), and pancreas ductal carcinoma 03 (0–170 zu∶L1210;>950 zu∶Panc 03 (200 zone units [zu] = 6.5 mm zone of inhibition of cultured tumor colonies from drug disk). In vivo we have tested batracylin against mammary adenocarcinoma 16/C, colon 9, colon 38, colon 51, Panc 03, and hepatoma 129. Upon oral administration, batracylin was effective against colon 9 (T/C = 2.4%) and marginally active against colon 38 (T/C = 39%). Batracylin was orally ineffective against Panc 03 (T/C > 100%), colon #51 (T/C = 77%) and hepatoma 129 (T/C > 100%). Upon subcutaneous administration, batracylin was effective against colon #9 (T/C = 0%), and Panc 03 (T/C = 15%) but ineffective against mammary 16/C (T/C > 100%). At efficacious doses, delayed neurotoxicity, hepatic toxicity and a significant host weight loss was noted (with slow recovery). Both our in vitro data and the NCI in vivo data confirm its scant activity against L1210 (%ILS = 8 to 16%). Although showing activity against selected murine solid tumors, it lacked curative potential with early stage disease [C38, C9, Panc 03] and has shown relative inactivity in vitro against human solid tumor cell lines (H-125, CX-1, HCT-8, HCT-116). Batracylin has entered large animal toxicology trials at the NCI, anticipating phase I clinical evaluation.
Similar content being viewed by others
References
Plowman J, Paull KD, Atassi G, Harrison Jr SD, Dykes DJ, Kabbe HJ, Narayanan VL, Yoder OC: Preclinical antitumor activity of Batracylin (NSC 320846). Invest New Drugs 6:147–153, 1988
Corbett TH, Roberts BJ, Leopold WR, Peckham JC, Wilkoff LJ, Griswold Jr DP, Schabel Jr FM: Induction and chemotherapeutic response of two transplantable ductal adenocarcinomas of the pancreas in C57B1/6 mice. Cancer Res 44:717–726, 1984
Corbett TH, Leopold WR, Dykes DJ, Roberts BJ, Griswold Jr DP, Schabel Jr FM: Toxicity and anticancer activity of a new triazine antifolate (NSC 127755). Cancer Res 42:1701–1715, 1982
Corbett TH, Griswold Jr DP, Roberts BJ, Peckham JC, Schabel Jr FM: Tumor induction relationships in development of transplantable cancers of the colon in mice for chemotherapy assays, with a note on carcinogen structure. Cancer Res 35:2434–2439, 1975
Corbett TH, Griswold Jr DP, Roberts BJ, Peckham JC, Schabel Jr FM: Evaluation of single agents and combinations of chemotherapeutic agents in mouse colon carcinomas. Cancer 40:2660–2680, 1977
Corbett TH, Roberts BJ, Trader MW, Laster Jr WR, Griswold Jr DP, Schabel Jr FM: Response of transplantable tumors of mice to anthracenedione derivatives alone and in combination with clinically useful agents. Cancer Treat Rep 66(5):1187–1200, 1982
Corbett TH, Griswold Jr DP, Roberts BJ, Peckham CH, Schabel Jr FM: Biology and therapeutic response of a mouse mammary adenocarcinoma (16/C) and its potential as a model for surgical adjuvant chemotherapy. Cancer Treat Rep 62:1471–1488, 1978
Carney DN, Gazdar AF, Bepler G, Guccion JG, Marangos PJ, Moody TW, Zweig MH, Minna JD: Establishment and identification of small cell lung cancer cell lines having classic and variant features. Cancer Res 45:2913–2923, 1985
Ovejara AA, Houchens DP: Human tumor xenografts in athymic nude mice as a preclinical screen for anticancer agents. Seminars in Oncology, 8(4):386–393, 1981
Tompkins WAF, Watrach AM, Schmale JD, Schultz RM, Harris JA: Cultural and antigenic properties of newly established cell strains derived from adenocarcinomas of the human colon and rectum. J Natl Cancer Inst 52:1101–1110, 1974
Brattain MG, Fine WD, Khaled FM, Thompson J, Brattain DE: Heterogeneity of malignant cells from a human colonic adenocarcinoma. Cancer Res 41:1751–1756, 1981
Corbett TH, Valeriote FA: Rodent models in experimental chemotherapy. In: Kallman RF (ed), The Use of Rodent Tumors in Experimental Cancer Therapy: Conclusions and Recommendations. Pergamon Press, 1987, Chapt 50, pp 233–247
Corbett TH: A selective two-tumor soft agar assay for drug discovery (Abstr). Proc Am Assoc Cancer Res 25:325, 1984
Corbett TH, Wozniak A, Gerpheide S, Hanka L: A selective two-tumor soft agar assay for drug discovery. In: White E (ed), In Vitro and In Vivo Models for Detection of New Antitumor Drugs. 14th International Congress of Chemotherapy. University of Tokyo Press, Japan, 1987, pp. 5–14
Corbett TH, Bissery M-C, Wozniak A, Plowman J, Polin L, Tapazoglou E, Dieckman J, Valeriote F: Activity of Flavone acetic acid (NSC 347512) against solid tumors of mice. Invest New Drugs 4:207–220, 1986
LoRusso PM, Bissery MC, Polin L, White K, Corbett TH: NSC 32846 (Bayer): A novel agent with solid tumor selectivity. (Abstr) Amer Assoc Cancer Res 1332, 1987–88
Pazdur R, Redman BG, Corbett T, Phillips M, Baker L: Phase I trial of spiromustine (NSC 172112) and evaluation of toxicity and schedule in a murine model. Cancer Res 47:4213–4217, 1987
Rouesse JG, LeChevalier T, Caille P, Mondesir JM, Sancho-Garneir H, May-Levin F, Spielman M, DeJager R, Amiel JL: Phase II study of elliptinium in advanced breast cancer. Cancer Treat Rep 69(6):707–708, 1985
Caille P, Mondeser JM, Droz JP, Kerbrat P, Goodman A, Ducret JP, Theodore C, Speilman M, Rouesse J, Amiel JL: Phase II trial of elliptinium in advanced renal cell cancer. Cancer Treat Rep 69(7–8):901–902, 1985
Somers R, Rouesse J, Van Oosterom A, Thomas D: Phase II study of elliptinium in metastatic soft tissue sarcoma. Eur J Cancer Clin Oncol 21(5):591–593, 1985
Clarysse A, Brugaroles A, Siegenthaler P, Abele R, Cavalli F, DeJager R, Renard G, Rozeneweig M, Hansen HH: Phase II study of 9-hydroxy-2-N-methylellipticinium Acetate. Eur J Cancer Clin Oncol 20(2):243–247, 1984
Einzig AI, Gralla RJ, Leyland-Jones BR, Kelsen DP, Cibas I, Lewis E, Greenberg E: Phase I study of elliptinium (2-N-methyl-9-hydroxy-ellipticinium). Cancer Invest 3(3):235–241, 1985
Paoletti C, LePecq J-B, Dat-Xuong N, Juret P, Garnier H, Amiel J-L, Rouesse J: Antitumor activity, pharmacology, and toxicity of ellipticines, ellipticinium, and 9-hydrox derivatives: Preliminary clinical trials of 2-methyl-9-hydroxellipicinium (NSC 264–137). Recent results. Cancer Res 74:107–123, 1980
Paoletti C, Lesca C, Cros S, Malvy C, Auclair C: Ellipticine and derivatives induce breakage of L1210 cells DNA in vitro. Biochem Pharmacol 28:345–350, 1979.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Mucci-LoRusso, P., Polin, L., Bissery, MC. et al. Activity of batracylin (NSC-320846) against solid tumors of mice. Invest New Drugs 7, 295–306 (1989). https://doi.org/10.1007/BF00173759
Issue Date:
DOI: https://doi.org/10.1007/BF00173759