Abstract
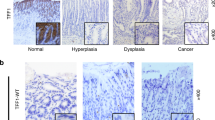
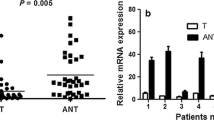
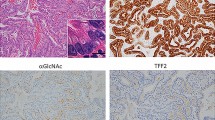
The trefoil factor family (TFF) of peptides, which are protease-resistant and have a strong affinity for mucins, play an important role in gastrointestinal mucosal protection and restitution. Prior studies have indicated that dysregulation of TFF2 was associated with cell migration, resistance to apoptosis, and gastric cancer invasion; however, the underlying mechanism associated with these actions remains unclear. Thus, to investigate the relationship between TFF2 and carcinogenesis in gastric cancer, TFF2 expression was analyzed by Western blot analysis in nine selected gastric cancer tissues and immunohistochemical staining was performed in paraffin-embedded samples from 157 gastric cancers. A reduced TFF2 expression was observed by Western blot analysis in the gastric cancer tissues. However, there was no significant difference in the TFF2 expression according to clinical and pathological parameters of the gastric cancers. To investigate the biological role of TFF2 in the development and progression of gastric cancer, a TFF2 expression plasmid was constructed for in vitro experiments of function. Introduction of TFF2 cDNA into a gastric cancer cell line did not affect tumor cell growth, cell migration or invasion. In conclusion, down-regulation of TFF2 in gastric cancer cells and restoration of TFF2 did not affect the tumorigenic potential of gastric cancer cells in vitro. The loss of TFF2 expression might be an early event of the multi-step process of gastrocarcinogenesis and may play a limited role in the mucosal protection of the normal gastric physiology.
Similar content being viewed by others
References
Bulitta, C. J. et al. Autoinduction of the trefoil factor 2 (TFF2) promoter requires an upstream cis-acting element. Biochem Biophys Res Commun 293:366–374 (2002).
Matsumoto, Y. et al. Helicobacter pylori infection triggers aberrant expression of activation-induced cytidine deaminase in gastric epithelium. Nat Med 13:470–476 (2007).
Maeda, K. et al. Vascular endothelial growth factor expression in preoperative biopsy specimens correlates with disease recurrence in patients with early gastric carcinoma. Cancer 86:566–571 (1999).
Alison, M. R. et al. Experimental ulceration leads to sequential expression of spasmolytic polypeptide, intestinal trefoil factor, epidermal growth factor and transforming growth factor alpha mRNAs in rat stomach. J Pathol 175:405–414 (1995).
Taupin, D. & Podolsky, D. K. Trefoil factors: initiators of mucosal healing. Nat Rev Mol Cell Biol 4:721–732 (2003).
Thim, L. Trefoil peptides: from structure to function. Cell Mol Life Sci 53:888–903 (1997).
Poulsom, R. Trefoil peptides. Baillieres Clin Gastroenterol 10:113–134 (1996).
Rio, M. C. et al. Breast cancer-associated pS2 protein: synthesis and secretion by normal stomach mucosa. Science 241:705–708 (1988).
Tomasetto, C. et al. hSP, the domain-duplicated homolog of pS2 protein, is co-expressed with pS2 in stomach but not in breast carcinoma. EMBO J 9:407–414 (1990).
Suemori, S., Lynch-Devaney, K. & Podolsky, D. K. Identification and characterization of rat intestinal trefoil factor: tissue- and cell-specific member of the trefoil protein family. Proc Natl Acad Sci USA 88: 11017–11021 (1991).
Chadwick, M. P., Westley, B. R. & May, F. E. Homodimerization and hetero-oligomerization of the singledomain trefoil protein pNR-2/pS2 through cysteine 58. Biochem J 327(Pt 1):117–123 (1997).
Kinoshita, K., Taupin, D. R., Itoh, H. & Podolsky, D. K. Distinct pathways of cell migration and antiapoptotic response to epithelial injury: structure-function analysis of human intestinal trefoil factor. Mol Cell Biol 20:4680–4690 (2000).
Marchbank, T. et al. Dimerization of human pS2 (TFF1) plays a key role in its protective/healing effects. J Pathol 185:153–158 (1998).
Otto, W. R. et al. Effects of pancreatic spasmolytic Polypeptide (PSP) on epithelial cell function. Eur J Biochem 235:64–72 (1996).
Hanby, A. M. et al. Spasmolytic polypeptide is a major antral peptide: distribution of the trefoil peptides human spasmolytic polypeptide and pS2 in the stomach. Gastroenterology 105:1110–1116 (1993).
Podolsky, D. K. et al. Identification of human intestinal trefoil factor. Goblet cell-specific expression of a peptide targeted for apical secretion. J Biol Chem 268:6694–702 (1993).
Oertel, M. et al. Trefoil factor family-peptides promote migration of human bronchial epithelial cells: synergistic effect with epidermal growth factor. Am J Respir Cell Mol Biol 25:418–424 (2001).
Playford, R. J. et al. Human spasmolytic polypeptide is a cytoprotective agent that stimulates cell migration. Gastroenterology 108:108–116 (1995).
Bossenmeyer-Pourie, C. et al. The trefoil factor 1 participates in gastrointestinal cell differentiation by delaying G1-S phase transition and reducing apoptosis. J Cell Biol 157:761–770 (2002).
Calnan, D. P. et al. The trefoil peptide TFF1 inhibits the growth of the human gastric adenocarcinoma cell line AGS. J Pathol 188:312–317 (1999).
Marchbank, T. et al. Effect of ectopic expression of rat trefoil factor family 3 (intestinal trefoil factor) in the jejunum of transgenic mice. J Biol Chem 276:24088–24096 (2001).
Taupin, D. R., Kinoshita, K. & Podolsky, D. K. Intestinal trefoil factor confers colonic epithelial resistance to apoptosis. Proc Natl Acad Sci USA 97:799–804 (2000).
Lefebvre, O. et al. Gastric mucosa abnormalities and tumorigenesis in mice lacking the pS2 trefoil protein. Science 274:259–262 (1996).
Tebbutt, N. C. et al. Reciprocal regulation of gastrointestinal homeostasis by SHP2 and STAT-mediated trefoil gene activation in gp130 mutant mice. Nat Med 8:1089–1097 (2002).
Hippo, Y. et al. Differential gene expression profiles of scirrhous gastric cancer cells with high metastatic potential to peritoneum or lymph nodes. Cancer Res 61:889–895 (2001).
Missiaglia, E. et al. Analysis of gene expression in cancer cell lines identifies candidate markers for pancreatic tumorigenesis and metastasis. Int J Cancer 112:100–112 (2004).
Cook, G. A., Familari, M., Thim, L. & Giraud, A. S. The trefoil peptides TFF2 and TFF3 are expressed in rat lymphoid tissues and participate in the immune response. FEBS Lett 456:155–159 (1999).
Farrell, J. J. et al. TFF2/SP-deficient mice show decreased gastric proliferation, increased acid secretion, and increased susceptibility to NSAID injury. J Clin Invest 109:193–204 (2002).
Houghton, J. et al. Gastric cancer originating from bone marrow-derived cells. Science 306:1568–1571 (2004).
Dhar, D. K. et al. Expression of cytoplasmic TFF2 is a marker of tumor metastasis and negative prognostic factor in gastric cancer. Lab Invest 83:1343–1352 (2003).
Emami, S. et al. Trefoil factor family (TFF) peptides and cancer progression. Peptides 25:885–898 (2004).
Shi, S. Q., Cai, J. T. & Yang, J. M. Expression of trefoil factors 1 and 2 in precancerous condition and gastric cancer. World J Gastroenterol 12:3119–3122 (2006).
Chi, A. L., Lim, S. & Wang, T. C. Characterization of a CCAAT-enhancer element of trefoil factor family 2 (TFF2) promoter in MCF-7 cells. Peptides 25:839–847 (2004).
Vestergaard, E. M. et al. Immunoassays of human trefoil factors 1 and 2: measured on serum from patients with inflammatory bowel disease. Scand J Clin Lab Invest 64:146–156 (2004).
Dubeykovskaya, Z., Dubeykovskiy, A., Solal-Cohen, J. & Wang, T. C. Secreted trefoil factor 2 activates the CXCR4 receptor in epithelial and lymphocytic cancer cell lines. J Biol Chem 284:3650–3662 (2009).
Hoosein, N. M., Thim, L., Jorgensen, K. H. & Brattain, M. G. Growth stimulatory effect of pancreatic spasmolytic polypeptide on cultured colon and breast tumor cells. FEBS Lett 247:303–306 (1989).
Siu, L. S. et al. TFF2 (trefoil family factor2) inhibits apoptosis in breast and colorectal cancer cell lines. Peptides 25:855–863 (2004).
Otto, W. R. & Patel, K. Trefoil factor family (TFF)-domain peptides in the mouse: embryonic gastrointestinal expression and wounding response. Anat Embryol (Berl) 199:499–508 (1999).
Lalani, E. N. et al. Trefoil factor-2, human spasmolytic polypeptide, promotes branching morphogenesis in MCF-7 cells. Lab Invest 79:537–546 (1999).
Kononen, J. et al. Tissue microarrays for high-throughput molecular profiling of tumor specimens. Nat Med 4:844–847 (1998).
Nam, S. W. et al. A sensitive screening assay for secreted motility-stimulating factors. Cell Motil Cytoskeleton 46:279–842 (2000).
Song, J. et al. Autotaxin (lysoPLD/NPP2) protects fibroblasts from apoptosis through its enzymatic product, lysophosphatidic acid, utilizing albumin-bound substrate. Biochem Biophys Res Commun 337:967–975 (2005).
Author information
Authors and Affiliations
Corresponding author
Additional information
These authors contributed equally to this work
Rights and permissions
About this article
Cite this article
Jung, K.H., Kim, P.J., Kim, J.K. et al. Decreased expression of TFF2 and gastric carcinogenesis. Mol. Cell. Toxicol. 6, 261–269 (2010). https://doi.org/10.1007/s13273-010-0036-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13273-010-0036-z