Abstract
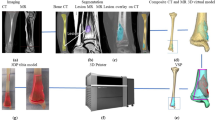
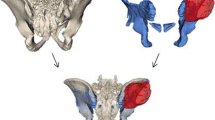
Oncological interventions in thoracic cavity have some important problems such as choice of correct operative approaches depending on the tumor, size, extension, and location. In sarcoma surgery, wide resection should be aimed for the curative surgery. Purpose of this study was to evaluate pre-operative planning of patient-specific thoracic cavity model made by multidisciplinary surgeon team for complex tumor mass for oncological procedures. Patient’s scans showed a large mass encroaching on the mediastinum and heart, with erosion of the adjacent ribs and vertebral column. Individual model of this case with thoracic tumor was reconstructed from the DICOM file of the CT data. Surgical team including six interdisciplinary surgeons explained their surgical experience of the use of 3D life-size individual model for guiding surgical treatment. Before patients consented to surgery, each surgeon explained the surgical procedure and perioperative risks to her. A questionnaire was applied to 10 surgical residents to evaluate the 3D model’s perception. 3D model scans were useful in determining the site of the lesion, the exact size, extension, attachment to the surrounding structures such as lung, aorta, vertebral column, or vascular involvement, the number of involved ribs, whether the diaphragm was involved also in which order surgeons in the team enter the surgery. 3D model’s perception was detected statistical significance as < 0.05. Viewing thoracic cavity with tumor model was more efficient than CT imaging. This case was surgically difficult as it included vital structures such as the mediastinal vessels, aorta, ribs, sternum, and vertebral bodies. A difficult pathology for which 3D model has already been explored to assist anatomic visualization was mediastinal osteosarcoma of the chest wall, diaphragm, and the vertebral column. The study helped to establish safe surgical line wherever the healthy tissue was retained and enabled osteotomy of the affected spinal corpus vertically with posterior-anterior direction by preserving the spinal cord and the spinal nerves above and distal the tumor. 3D tumor model helps to transfer complex anatomical information to surgeons, provide guidance in the pre-operative planning stage, for intra-operative navigation and for surgical collaboration purposes. Total radical excision of the bone tumor and reconstructions of remaining structures using life-size model was the key for successful treatment and better outcomes. The recent explosion in popularity of 3D printing is a testament to the promise of this technology and its profound utility in orthopedic oncological surgery.









Similar content being viewed by others
References
Arndt CA, Rose PS, Folpe AL, Laack NN (2012) Common musculoskeletal tumors of childhood and adolescence. Mayo Clin Proc 87(5):475–487
Gladish GW, Sabloff BM, Munden RF, Truong MT, Erasmus JJ, Chasen MH (2002) Primary thoracic sarcomas. Radiographics 22(3):621–637
Rocca M, Salone M, Galletti S, Balladelli A, Vanel D, Briccoli A (2013) The role of imaging for the surgeon in primary malignant bone tumors of the chest wall. Eur J Radiol 82(12):2070–2075
Athanassiadi K, Kalavrouziotis G, Rondogianni D, Loutsidis A, Hatzimichalis A, Bellenis I (2001) Primary chest wall tumors: early and long-term results of surgical treatment. Eur J Cardiothorac Surg 19(5):589–593
Hung JJ, Chou TY, Sun CH, Liu JS, Hsu WH (2008) Primary synovial sarcoma of the posterior chest wall. Ann Thorac Surg 85(6):2120–2122
Anderson BO, Burt ME (1994) Chest wall neoplasms and their management. Ann Thorac Surg 58(6):1774–1781
Krauel L, Fenollosa F, Riaza L, Pérez M, Tarrado X, Morale A, Gomà J, Mora J (2016) Use of 3D prototypes for complex surgical oncologic cases. World J Surg 40(4):889–894
Kurenov SN, Ionita C, Sammons D, Demmy TL (2015) Three dimensional printing to facilitate anatomic study, device development, simulation, and planning in thoracic surgery. J Thorac Cardiovasc Surg 149(4):973–979 e1
Braham E, Aloui S, Aouadi S, Drira I, Kilani T, El Mezni F (2013) Synovial sarcoma of the chest wall: a case report and literature review. Ann Transl Med 1(1):9
Gillaspie EA, Matsumoto JS, Morris NE, Downey RJ, Shen KR, Allen MS, Blackmon SH (2016) From 3-dimensional printing to 5-dimensional printing: enhancing thoracic surgical planning and resection of complex tumors. Ann Thorac Surg 101(5):1958–1962
Govsa F, Karakas AB, Ozer MA, Eraslan C (2018) Development of life-size patient-specific 3D- printed dural venous models for preoperative planning. World Neurosurg 110:e141–e149
Cromeens BP, Ray WC, Hoehne B, Abayneh F, Adler B, Besner GE (2017) Facilitating surgeon understanding of complex anatomy using a three-dimensional printed model. J Surg Res 216:18–25
Hoang D, Perrault D, Stevanovic M, Ghiassi A (2016) Surgical applications of three-dimensional printing: a review of the current literature & how to get started. Ann Transl Med 4(23):456
Chowdhury SK, Subbarao KS, Nachiappan M, Agrawal K (2000) Primary neoplasm of the chest wall: surgical management. Asian Cardiovasc Thorac Ann 8:249–252
Cakir O, Topal U, Bayram AS, Tolunay S (2005) Sarcomas: rare primary malignant tumors of the thorax. Diagn Interv Radiol 11(1):23–27
Walsh GL, Davis BM, Swisher SG, Vaporciyan AA, Smythe WR, Willis-Merriman K, Roth JA Jr, Putnam JB (2001) A single-institutional, multidisciplinary approach to primary sarcomas involving the chest wall requiring full-thickness resections. J Thorac Cardiovasc Surg 121(1):48–60
Wang L, Cao T, Li X, Huang L (2016) Three-dimensional printing titanium ribs for complex reconstruction after extensive posterolateral chest wall resection in lung cancer. J Thorac Cardiovasc Surg 152(1):e5–e7
Staats K, Panotopoulos J, Tiefenboeck TM, Windhager R, Funovics PT (2017) Computer navigation-assisted surgery for musculoskeletal tumors: a closer look into the learning curve. Eur J Orthop Surg Traumatol 27(6):851–858
Novoa N, Benito P, Jime’nez MF, de Juan A, Luis Aranda J, Varela G (2005) Reconstruction of chest wall defects after resection of large neoplasms: ten-year experience. Interact Cardiovasc Thorac Surg 4(3):250–255
George E, Liacouras P, Rybicki FJ, Mitsouras D (2017) Measuring and establishing the accuracy and reproducibility of 3D-printed medical models. Radiographics 37(5):1424–1450
Matsumoto JS, Morris JM, Rose PS (2016) 3-dimensional printed anatomic models as planning aids in complex oncology surgery. JAMA Oncol 2(9):1121–1122
George E, Barile M, Tang A, Wiesel O, Coppolino A, Giannopoulos A, Mentzer S, Jaklitsch M, Hunsaker A, Mitsouras D (2017) Utility and reproducibility of 3-dimensional printed models in pre-operative planning of complex thoracic tumors. J Surg Oncol 116(3):407–415
Giannopoulos A, Steigner M, George E, Barile M, Hunsaker AR, Rybicki FJ, Mitsouras D (2016) Cardiothoracic applications of 3D printing. J Thorac Imaging 31(5):253–272
Kim MP, Ta AH, Ellsworth WA, Marco RA, Gaur P, Miller JS (2015) Three dimensional model for surgical planning in resection of thoracic tumors. Int J Surg Case Rep 16:127–129
Govsa F, Ozer MA, Biceroglu H, Karakas AB, Cagli S, Eraslan C, Alagoz AK (2018) Creation of 3D life size: patient specific C1 fracture models for screw fixation. World Neurosurg 114:e173–e181
Andolfi C, Plana A, Kania P, Banerjee PP, Small S (2017) Usefulness of three-dimensional modeling in surgical planning, resident training, and patient education. J Laparoendosc Adv Surg Tech A 27(5):512–515
Acknowledgments
Special thanks to Prof. Dr. Cemil Caliskan MD, Department of General Surgery and Dr. Serkan Ertugay MD, Department of Cardiovascular Surgery Faculty of Medicine, Ege University for sincere efforts and assistances.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The study was approved by the suitably constituted Ethical Committee at Researches Department of Ege University (17-6/19), within which the work was undertaken, and the study conforms to the Declaration of Helsinki. The patient provided written informed consent.
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ozturk, A.M., Sirinturk, S., Kucuk, L. et al. Multidisciplinary Assessment of Planning and Resection of Complex Bone Tumor Using Patient-Specific 3D Model. Indian J Surg Oncol 10, 115–124 (2019). https://doi.org/10.1007/s13193-018-0852-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13193-018-0852-5