Abstract
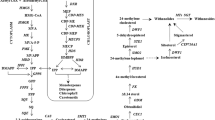
Coleus forskohlii is an herb, well-known for its medicinal compound forskolin present in its roots, with wide range of pharmaceutical applications. Here, we report, for the first time, the role of plant–probiotic bacterial endophytes of C. forskohlii, CFLB1 and CFRB1, isolated from leaf and root, which regulate plant growth and in plant forskolin content. Native bacterial endophyte, CFRB1 (Alcaligenes faecalis), significantly modulates primary plant productivity and forskolin content under pot and field conditions. Under field conditions, CFRB1 endophyte application significantly enhanced photosynthetic pigments and reduced the severity of root-knot and root rot diseases. Expression analyses of functional genes involved in the forskolin biosynthesis in C. forskohlii plants treated with CFRB1 endophyte under field conditions revealed differential upregulation of four C. forskohlii diterpene synthases (CfTPSs), CfTPS1, CfTPS2, CfTPS3 and CfTPS4, along with cytochrome P450 (CfCYP76AH15) and acyltransferase (CfACT1–8) genes. CFRB1 treatment reduced the severity of nematode infection and root rot in C. forskohlii plants by 81 and 78%, respectively. Overall, we demonstrate that cross-talk of plant–endophyte interaction in C. forskohlii is beneficial, leading to enhanced forskolin content through modulation of forskolin biosynthetic pathway genes along with increased plant yield and reduced disease incidence. Thus, endophytic isolate, A. faecalis (CFRB1), could be deployed as a novel bio-stimulant for enhancing in planta forskolin content during cultivation of C. forskohlii.







Similar content being viewed by others
References
Yan K, Gao LN, Cui YL, Zhang Y, Zhou X (2016) The cyclic AMP signaling pathway: exploring targets for successful drug discovery (review). Mol Med Rep 13:3715–3723
Raker VK, Becker C, Steinbrink K (2016) The cAMP pathway as therapeutic target in autoimmune and inflammatory diseases. Front Immunol 7:123
Ding X, Staudinger JL (2005) Induction of drug metabolism by forskolin: the role of the pregnane X receptor and the protein kinase a signal transduction pathway. J Pharmacol Exp Ther 312:849–856
Das A, Kamal S, Shakil NA, Sherameti I, Oelmüller R, Dua M, Tuteja N, Johri AK, Varma A (2012) The root endophyte fungus Piriformospora indica leads to early flowering, higher biomass and altered secondary metabolites of the medicinal plant, Coleus forskohlii. Plant Signal Behav 7:103–112
Kandel SL, Joubert PM, Doty SL (2017) Bacterial endophyte colonization and distribution within plants. Microorganisms 5:77
Gouda S, Das G, Sen SK, Shin HS, Patra JK (2016) Endophytes: a treasure house of bioactive compounds of medicinal importance. Front Microbiol 7:1538
Hardoim PR, van Overbeek LS, Berg G, Pirttilä AM, Compant S, Campisano A, Döring M, Sessitsch A (2015) The hidden world within plants: ecological and evolutionary considerations for defining functioning of microbial endophytes. Microbiol Mol Biol Rev 79:293–320
Pandey SS, Singh S, Babu CS, Shanker K, Srivastava NK, Kalra A (2016) Endophytes of Opium poppy differentially modulate host plant productivity and genes for the biosynthetic pathway of benzylisoquinoline alkaloids. Planta 243:1097–1114
van der Heijden MG, Bakker R, Verwaal J, Scheublin TR, Rutten M, van Logtestijn R, Staehelin C (2006) Symbiotic bacteria as a determinant of plant community structure and plant productivity in dune grassland. FEMS Microbiol Ecol 56:178–187
Win KT, Tanaka F, Okazaki K, Ohwaki Y (2018) The ACC deaminase expressing endophyte Pseudomonas spp. enhances NaCl stress tolerance by reducing stress-related ethylene production, resulting in improved growth, photosynthetic performance, and ionic balance in tomato plants. Plant Physiol Biochem 127:599–607
Hacquard S, Kracher B, Hiruma K, Münch PC, Garrido-Oter R, Thon MR, Weimann A, Damm U, Dallery JF, Hainaut M, Henrissat B, Lespinet O, Sacristán S, ver Loren van Themaat E, Kemen E, McHardy AC, Schulze-Lefert P, O’Connell RJ (2016) Survival trade-offs in plant roots during colonization by closely related beneficial and pathogenic fungi. Nat Commun 7:11362
Meng X, Yan D, Long X, Wang C, Liu Z, Rengel Z (2014) Colonization by endophytic Ochrobactrum anthropi Mn1 promotes growth of Jerusalem artichoke. Microb Biotechnol 7:601–610
Saddique MAB, Ali Z, Khan AS, Rana IA, Shamsi IH (2018) Inoculation with the endophyte Piriformospora indica significantly affects mechanisms involved in osmotic stress in rice. Rice 11:34
Pandey SS, Singh S, Pandey H, Srivastava M, Ray T, Soni S, Pandey A, Shanker K, Babu CSV, Banerjee S, Gupta MM, Kalra A (2018) Endophytes of Withania somnifera modulate in planta content and the site of withanolide biosynthesis. Sci Rep 8:5450
Xia Y, DeBolt S, Dreyer J, Scott D, Williams MA (2015) Characterization of culturable bacterial endophytes and their capacity to promote plant growth from plants grown using organic or conventional practices. Front Plant Sci 6:490
Naqqash T, Hameed S, Imran A, Hafnif MK, Majeed A, van Elsas JD (2016) Differential response of potato toward inoculation with taxonomically diverse plant growth promoting rhizobacteria. Front Plant Sci 7:144
Hubbard M, Germida JJ, Vujanovic V (2014) Fungal endophytes enhance wheat heat and drought tolerance in terms of grain yield and second-generation seed viability. J Appl Microbiol 116:109–122
Sayyed RZ, Chincholkar SB (2009) Siderophore-producing Alcaligenes feacalis exhibited more biocontrol potential vis-à-vis chemical fungicide. Curr Microbiol 58:47–51
Ramakrishna A, Ravishankar GA (2011) Influence of abiotic stress signals on secondary metabolites in plants. Plant Signal Behav 6:1720–1731
Kumar A, Patil D, Rajamohanan PR, Ahmad A (2013) Isolation, purification and characterization of vinblastine and vincristine from endophytic fungus Fusarium oxysporum isolated from Catharanthus roseus. PLoS One 8:e71805
Palem PP, Kuriakose GC, Jayabaskaran C (2015) An endophytic fungus, Talaromyces radicus, isolated from Catharanthus roseus, produces vincristine and vinblastine, which induce apoptotic cell death. PLoS One 10:e0144476
Soliman SS, Tsao R, Raizada MN (2011) Chemical inhibitors suggest endophytic fungal paclitaxel is derived from both mevalonate and non-mevalonate-like pathways. J Nat Prod 74:2497–2504
Li J, Zhao GZ, Varma A, Qin S, Xiong Z, Huang HY, Zhu WY, Zhao LX, Xu LH, Zhang S, Li WJ (2012) An endophytic Pseudonocardia species induces the production of artemisinin in Artemisia annua. PLoS One 7:e51410
Pandey SS, Singh S, Babu CS, Shanker K, Srivastava NK, Shukla AK, Kalra A (2016) Fungal endophytes of Catharanthus roseus enhance vindoline content by modulating structural and regulatory genes related to terpenoid indole alkaloid biosynthesis. Sci Rep 6:26583
Sessitsch A, Hardoim P, Döring J, Weilharter A, Krause A, Woyke T, Mitter B, Hauberg-Lotte L, Friedrich F, Rahalkar M, Hurek T, Sarkar A, Bodrossy L, van Overbeek L, Brar D, van Elsas JD, Reinhold-Hurek B (2012) Functional characteristics of an endophyte community colonizing rice roots as revealed by metagenomic analysis. Mol Plant-Microbe Interact 1:28–36
Eevers N, Gielen M, Sánchez-López A, Jaspers S, White JC, Vangronsveld J, Weyens N (2015) Optimization of isolation and cultivation of bacterial endophytes through addition of plant extract to nutrient media. Microb Biotechnol 8:707–715
Wright MH, Adelskov J, Greene AC (2017) Bacterial DNA extraction using individual enzymes and phenol/chloroform separation. J Microbiol Biol Educ 18:18.2.48
Bharti N, Pandey SS, Barnawal D, Patel VK, Kalra A (2016) Plant growth promoting rhizobacteria Dietzia natronolimnaea modulates the expression of stress responsive genes providing protection of wheat from salinity stress. Sci Rep 6:34768
Aktas M, Köster S, Kizilirmak S, Casanova JC, Betz H, Fritz C, Moser R, Yildiz Ö, Narberhaus F (2014) Enzymatic properties and substrate specificity of a bacterial phosphatidylcholine synthase. FEBS J 281:3523–3541
Wilms I, Möller P, Stock AM, Gurski R, Lai EM, Narberhaus F (2012) Hfq influences multiple transport systems and virulence in the plant pathogen Agrobacterium tumefaciens. J Bacteriol 194:5209–5217
Pateraki I, Andersen-Ranberg J, Hamberger B, Heskes AM, Martens HJ, Zerbe P, Bach SS, Møller BL, Bohlmann J, Hamberger B (2014) Manoyl oxide (13R), the biosynthetic precursor of forskolin, is synthesized in specialized root cork cells in Coleus forskohlii. Plant Physiol 164:1222–1236
Pateraki I, Andersen-Ranberg, J, Jensen NB, Wubshet SG, Heskes AM, Forman V, Hallström B, Hamberger B, Motawia MS, Olsen CE, Staerk D, Hansen J, Møller BL, Hamberger B (2017) Total biosynthesis of the cyclic AMP booster forskolin from Coleus forskohlii. eLife 6: e23001. https://doi.org/10.7554/eLife.23001
Schmittgen TD, Livak KJ (2008) Analyzing real-time PCR data by the comparative CT method. Nat Protoc 3:1101–1108
Lichtenthaler HK, Wellburn AR (1971) Determinations of total carotenoids and chlorophylls a and b of leaf extracts in different solvents. Biochem Soc Trans 11:591–592
Singh R, Kalra A, Paramaeswarn TN, Ravish BS, Divya S, Srinivas KVNS, Bagyaraj DJ (2012) Effect of potential bioinoculants and organic manures on root-rot and wilt, growth, yield and quality of organically grown Coleus forskohlii in a semiarid tropical region of Bangalore (India). Plant Pathol 61:700–708
Dong W, Holbrook CC, Timper P, Brenneman TB, Mullinix BG (2007) Comparison of methods for assessing resistance to Meloidogyne arenaria in peanut. J Nematol 39:169–175
Ghosh SK, Banerjee S, Pal S, Chakraborty N (2018) Encountering epidemic effects of leaf spot disease (Alternaria brassicae) on Aloe vera by fungal biocontrol agents in agrifields—an ecofriendly approach. PLoS One 13:e0193720
Englund E, Andersen-Ranberg J, Miao R, Hamberger B, Lindberg P (2015) Metabolic engineering of Synechocystis sp. PCC 6803 for production of the plant diterpenoid manoyl oxide. ACS Synth Biol 4:1270–1278
Loftus HL, Astell KJ, Mathai ML, Su XQ (2015) Coleus forskohlii extract supplementation in conjunction with a hypocaloric diet reduces the risk factors of metabolic syndrome in overweight and obese subjects: a randomized controlled trial. Nutrients 7:9508–9522
Kanne H, Burte NP, Prasanna V, Gujjula R (2015) Extraction and elemental analysis of Coleus forskohlii extract. Pharm Res 7:237–241
Shan Y, Xu L, Lu Y, Wang X, Zheng Q, Kong L, Niwa M (2008) Diterpenes from Coleus forskohlii (WILLD.) Briq. (Labiatae). Chem Pharm Bull 56:52–56
Boby BU, Bagyaraj DJ (2003) Biological control of root-rot of Coleus forskohlii Briq. using microbial inoculants. World J Microbiol Biotechnol 19:175–180
Das A, Tripathi S, Varma A (2014) In vitro plant development and root colonization of Coleus forskohlii by Piriformospora indica. World J Microbiol Biotechnol 30:1075–1084
Ray S, Singh S, Sarma BK, Singh HB (2016) Endophytic Alcaligenes isolated from horticultural and medicinal crops promotes growth in okra (Abelmoschus esculentus). J Plant Growth Regul 35:401
Beckers B, Op De Beeck M, Weyens N, Boerjan W, Vangronsveld J (2017) Structural variability and niche differentiation in the rhizosphere and endosphere bacterial microbiome of field-grown poplar trees. Microbiome 5:25
López-Arredondo D, González-Morales SI, Bello-Bello E, Alejo-Jacuinde G, Herrera L (2015) Engineering food crops to grow in harsh environments. F1000Res 4:651
Alam M, Abdul K, Abdul S, Shukla RS, Anwar M, Dharani S (2011) Synergistic effect of arbuscular mycorrhizal fungi and Bacillus subtilis on the biomass and essential oil yield of rose-scented geranium (Pelargonium graveolens). Arch Agron Soil Sci 57:889–898
Dharni S, Srivastava AK, Samad A, Patra DD (2014) Impact of plant growth promoting Pseudomonas monteilii PsF84 and Pseudomonas plecoglossicida PsF610 on metal uptake and production of secondary metabolite (monoterpenes) by rose-scented geranium (Pelargonium graveolens cv. bourbon) grown on tannery sludge amended soil. Chemosphere 117:433–439
Jayakumar A, Krishna A, Mohan M, Nair IC, Radhakrishnan EK (2018) Plant growth enhancement, disease resistance, and elemental modulatory effects of plant probiotic endophytic Bacillus sp. Fcl1. Probiotics Antimicrob Proteins 11:526–534
Basheer J, Ravi A, Mathew J, Krishnankutty RE (2018) Assessment of plant-probiotic performance of novel endophytic Bacillus sp. in talc-based formulation. Probiotics Antimicrob Proteins 11:256–263
Mastan A, Bharadwaj R, Kushwaha RK, Vivek Babu CS (2019) Functional fungal endophytes in Coleus forskohlii regulate labdane diterpene biosynthesis for elevated forskolin accumulation in roots. Microb Ecol. https://doi.org/10.1007/s00248-019-01376-w
Wani ZA, Kumar A, Sultan P, Bindu K, Riyaz-Ul-Hassa S, Ashraf N (2017) Mortierella alpina CS10E4, an oleaginous fungal endophyte of Crocus sativus L. enhances apocarotenoid biosynthesis and stress tolerance in the host plant. Sci Rep 7:8598
Gupta R, Singh A, Srivastava M, Singh V, Gupta MM, Pandey R (2016) Microbial modulation of bacoside A biosynthetic pathway and systemic defense mechanism in Bacopamonnieri under Meloidogyne incognita stress. Sci Rep 7:41867
Tiwari R, Awasthi A, Mall M, Shukla AK, Srinivas KVNS, Syamasundar KV, Kalra A (2013) Bacterial endophyte-mediated enhancement of in planta content of key terpenoid indole alkaloids and growth parameters of Catharanthus roseus. Ind Crop Prod 43:306–310
Ray S, Singh V, Singh S, Sarma BK, Singh HB (2016) Biochemical and histochemical analyses revealing endophytic Alcaligenes faecalis mediated suppression of oxidative stress in Abelmoschus esculentus challenged with Sclerotium rolfsii. Plant Physiol Biochem 109:430–441
Battini F, Cristani C, Giovannetti M, Agnolucci M (2016) Multifunctionality and diversity of culturable bacterial communities strictly associated with spores of the plant beneficial symbiont Rhizophagus intraradices. Microbiol Res 183:68–79
Wu CH, Bernard SM, Andersen GL, Chen W (2009) Developing microbe-plant interactions for applications in plant-growth promotion and disease control, production of useful compounds, remediation and carbon sequestration. Microb Biotechnol 2:428–440
Mishra A, Singh SP, Mahfooz S, Bhattacharya A, Mishra N, Shirke PA, Nautiyal CS (2018) Bacterial endophytes modulates the withanolide biosynthetic pathway and physiological performance in Withania somnifera under biotic stress. Microbiol Res 212-213:17–28
Su L, Shen Z, Ruan Y, Tao C, Chao Y, Li R, Shen Q (2017) Isolation of antagonistic endophytes from banana roots against Meloidogyne javanica and their effects on soil nematode community. Front Microbiol 8:2070
Egamberdieva D, Wirth S, Shurigin VV, Hashem A, Abd Allah EF (2017) Endophytic bacteria improve plant growth, symbiotic performance of chickpea (Cicer arietinum L.) and induce suppression of root rot caused by Fusarium solani under salt stress. Front Microbiol 8:1887
Chen B, Luo S, Wu Y, Ye J, Wang Q, Xu X, Pan F, Khan KY, Feng Y, Yang X (2017) The effects of the endophytic bacterium Pseudomonas fluorescens Sasm05 and IAA on the plant growth and cadmium uptake of Sedum alfredii Hance. Front Microbiol 8:2538
Acknowledgements
The authors express their sincere thanks to the Director, CSIR—Central Institute of Medicinal and Aromatic Plants, Lucknow, India, for providing his support, encouragement and necessary facilities during the course of the investigation.
Funding
This work was supported by BSC0117 and BSC203 (XII Five Year Plan Project) from the Council of Scientific and Industrial Research (CSIR), India. Department of Science and Technology (DST –INSPIRE) New Delhi, India. AM greatly acknowledges the Department of Science and Technology (DST—INSPIRE), New Delhi, India, for providing research fellowship.
Author information
Authors and Affiliations
Contributions
Research concept was conceived and designed by CSV. AM and DR performed the bench work, and AM, CSV, DR and SGD analyzed the data. AM, SGD and CSV wrote the manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
CIMAP Publication Communication Number: CIMAP/PUB/2018/74
Electronic supplementary material
ESM 1
(DOCX 5389 kb)
Rights and permissions
About this article
Cite this article
Mastan, A., Rane, D., Dastager, S.G. et al. Plant Probiotic Bacterial Endophyte, Alcaligenes faecalis, Modulates Plant Growth and Forskolin Biosynthesis in Coleus forskohlii. Probiotics & Antimicro. Prot. 12, 481–493 (2020). https://doi.org/10.1007/s12602-019-09582-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12602-019-09582-1