Abstract
Background
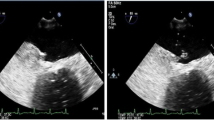
Therapeutic uncertainty is inherent in decisions in patients with patent foramen ovale (PFO) and cryptogenic stroke. We aimed to determine clinical implications of PFO identification in transesophageal echocardiography (TEE) after a cryptogenic ischemic stroke.
Methods
Consecutive TEE done between 2011 and 2015 in patients with previous cryptogenic stroke was evaluated. Clinical implications of PFO identification (closure and/or medical therapy) were retrieved from the medical records and discharge summaries. Adverse events related to therapy, stroke recurrence and death were analyzed during follow-up.
Results
Three-hundred one patients (mean age 59 ± 11 years; 61% male) underwent a TEE, of which 77 (26%) patients had a diagnosis of PFO. Patients with PFO were younger (56 ± 13 versus 60 ± 14, p = 0.03). Of those with PFO, 23 (30%) underwent percutaneous closure of PFO and these patients had more frequently complex or large PFO (p < 0.001 and p = 0.004, respectively). The remaining 54 (70%) were treated with medical therapy: 30 (39%) with antiplatelet therapy and 24 (31%) with oral anticoagulation. During follow-up (44 ± 17 months), only two patients had another stroke (both referred for PFO closure, while they were waiting for the procedure) and two patients, on whom PFO closure was not performed, died (not for cardiovascular causes).
Conclusion
PFO’s (size and complexity) and patients’ characteristics influenced clinical decision when PFO was detected on TEE. The risk for recurrent stroke was not increased in patients who did not undergo PFO closure; although two patients waiting for PFO closure had recurrent stroke, demonstrating its importance.



Similar content being viewed by others
Abbreviations
- AF:
-
Atrial fibrillation
- ASA:
-
Atrial septal aneurysm
- ECG:
-
Electrocardiogram
- h:
-
Hour
- LA:
-
Left atrium
- OAC:
-
Oral anticoagulation
- PFO:
-
Patent foramen ovale
- RA:
-
Right atrium
- TEE:
-
Transesophageal echocardiography
- TTE:
-
Transthoracic echocardiography
References
Calvet D, Mas JL. Closure of patent foramen ovale in cryptogenic stroke: a never ending story. Curr Opin Neurol. 2014;27:13–9.
Furlan AJ, Reisman M, Massaro J, et al. Closure or medical therapy for cryptogenic stroke with patent foramen ovale. N Engl J Med. 2012;366:991–9.
Carroll JD, Saver JL, Thaler DE, et al. Closure of patent foramen ovale versus medical therapy after cryptogenic stroke. N Engl J Med. 2013;368:1092–100.
Meier B, Kalesan B, Mattle HP, et al. Percutaneous closure of patent foramen ovale in cryptogenic embolism. N Engl J Med. 2013;368:1083–91.
Rengifo-Moreno P, Palacios IF, Junpaparp P, et al. Patent foramen ovale transcatheter closure vs. medical therapy on recurrent vascular events: a systematic review and meta-analysis of randomized controlled trials. Eur Heart J. 2013;34:3342–52.
Mas JL, Derumeaux G, Guillon B, et al. Patent foramen ovale closure or anticoagulation vs. antiplatelets after stroke. N Engl J Med. 2017;377:1011–21.
Søndergaard L, Kasner SE, Rhodes JF, et al. Patent foramen ovale closure or antiplatelet therapy for cryptogenic stroke. N Engl J Med. 2017;377:1033–42.
Ay H, Furie KL, Smith WS, et al. An evidence-based causative classification system for acute ischemic stroke. Ann Neurol. 2005;58(5):688–97.
Pinto FJ. When and how to diagnose patent foramen ovale. Should it always be repaired? Heart. 2005;91:438–40.
Vitarelli A, Mangieri E, Capotosto L, et al. Echocardiographic findings in simple and complex patent foramen ovale before and after transcatheter closure. Eur Heart J Cardiovasc Imaging. 2014;15(12):1377–85.
Pepi M, Evangelista A, Nihoyannopoulos P, et al. Recommendations for echocardiography use in the diagnosis and management of cardiac sources of embolism European Association of Echocardiography. Eur J Echocardiogr. 2010;11:461–76.
Kaatz S, Ahmad D, Spyropoulus C, et al. Definition of clinically relevant non-major bleeding in studies of anticoagulants in atrial fibrillation and venous thromboembolic disease in non-surgical patients: communication from the SSC of the ISTH. J Thromb Haemost. 2015;13:2119–26.
Saric M, Armour AC, Arnaout MS, et al. Guidelines for the use of echocardiography in the evaluation of a cardiac source of embolism. J Am Soc Echocardiogr. 2016;29(1):1–42.
McGrath ER, Paikin JS, Motlagh B, et al. Transesophageal echocardiography in patients with cryptogenic ischemic stroke: a systematic review. Am Heart J. 2014;168:706–12.
Handke M, Harloff A, Olschewski M, et al. Patent foramen ovale and cryptogenic stroke in older patients. N Engl J Med. 2007;357:2262–8.
Rodrigues AC, Picard MH, Carbone A, et al. Importance of adequately performed valsalva maneuver to detect patent foramen ovale during transesophageal echocardiography. J Am Soc Echocardiogr. 2013;26:1337–43.
Buttignoni SC, Khorsand A, Mundigler G, et al. Agitated saline versus polygelatine for the echocardiographic assessment of patent foramen ovale. J Am Soc Echocardiogr. 2004;17:1059–65.
Gladstone D, Spring M, Dorian P, et al. Atrial fibrillation in patients with cryptogenic stroke. N Engl J Med. 2014;370:2467–77.
Sanna T, Diener HC, Passman RS, et al. Cryptogenic stroke and underlying atrial fibrillation. N Engl J Med. 2014;370(26):2478–86.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Rita Marinheiro, Leonor Parreira, Pedro Amador, Isabel Silvestre, Carla Antunes and Rui Caria declare that they have no conflict of interest.
Human rights statements and informed consent
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1964 and later revisions. Informed consent was obtained from all patients for being included in the study.
Rights and permissions
About this article
Cite this article
Marinheiro, R., Parreira, L., Amador, P. et al. Implications of detection of foramen ovale patent after cryptogenic ischemic stroke. J Echocardiogr 17, 44–51 (2019). https://doi.org/10.1007/s12574-018-0388-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12574-018-0388-2