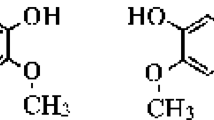Abstract
An essential step in the life cycle of human immunodeficiency virus type 1 (HIV-1) is integration of the double-stranded retroviral DNA into the genome of the host cell. HIV-1 integrase, the enzyme that inserts the vital DNA into the host chromosome, is an attractive and rational target for anti-AIDS drug design because it is essential for HIV replication and there are no known counterparts in the host cell. Inhibitors of this enzyme have the great potential to complement the therapeutic use of HIV protease and reverse transcriptase inhibitors. Natural products have provided a source of new drug candidates for anti-AIDS therapy. Baicalein and baicalin, identified components of a Chinese herbal medicine Scutellaria baicalensis Georgi, have been shown to inhibit infectivity and replication of HIV. They are therefore promising lead compounds for developing new anti-AIDS drugs. To understand how the inhibitors work and therefore design more potent and specific inhibitors, we have used molecular modeling techniques to investigate the binding modes of these inhibitors. The three-dimensional structures of these inhibitors were first built. Then, computational binding studies of these inhibitors, based on the crystal structure of the HIV-1 integrase catalytic domain, were performed to study the complex structure. The preliminary results of our computational modeling study demonstrated that Baicalein binds to the active site region of the HIV-1 integrase. Our study will be of help to identify the pharmacophores of inhibitors binding to HIV-1 integrase and design new pharmaceuticals for the treatment of AIDS.
Similar content being viewed by others
References
Ahn, H.C., Lee, S.Y., Kim, J.W., Son, W.S., Shin, C.G., Lee, B.J. 2001. Binding aspects of baicalein to HIV-1 integrase. Mol Cells 12, 127–130.
Bhattacharya, S., Osman, H. 2009. Novel targets for anti-retroviral therapy. J Infect 59, 377–386.
Cheng, C.W., Bian, Z.X., Wu, T.X. 2009. Systematic review of Chinese herbal medicine for functional constipation. World J Gastroenterol 15, 4886–4895.
Eron, J.J.J. 2009. Antiretroviral therapy: new drugs, formulations, ideas, and strategies. Top HIV Med 17, 146–150.
Ewing, T.J., Makino, S., Skillman, A.G., Kuntz, I.D. 2001. DOCK 4.0: search strategies for automated molecular docking of flexible molecule databases. J Comput Aided Mol Des 15, 411–428.
Farnet, C.M., Wang, B., Lipford, J.R., Bushman, F.D. 1996. Differential inhibition of HIV-1 preintegration complexes and purified integrase protein by small molecules. Proc Natl Acad Sci USA 93, 9742–9747.
Garrido, C., Geretti, A.M., Zahonero, N., Booth, C., Strang, A., Soriano, V., De Mendoza, C. 2010. Integrase variability and susceptibility to HIV integrase inhibitors: Impact of subtypes, antiretroviral experience and duration of HIV infection. J Antimicrob Chemother 65, 320–326.
Goldgur, Y., Craigie, R., Cohen, G.H., Fujiwara, T., Yoshinaga, T., Fujishita, T., Sugimoto, H., Endo, T., Murai, H., Davies, D.R. 1999. Structure of the HIV-1 integrase catalytic domain complexed with an inhibitor: A platform for antiviral drug design. Proc Natl Acad Sci USA 96, 13040–13043.
Gupta, P., Roy, N., Garg, P. 2009. Docking-based 3DQSAR study of HIV-1 integrase inhibitors. Eur J Med Chem 44, 4276–4287.
Hu, Z., Southerland, W. 2007. WinDock: Structure-based drug discovery on Windows-based PCs. J Comp Chem 28, 2347–2351.
Jenkins, T.M., Esposito, D., Engelman, A., Craigie, R. 1997. Critical contacts between HIV-1 integrase and viral DNA identified by structure-based analysis and photo-crosslinking. EMBO J 16, 6849–6859.
Kallings, L.O. 2008. The first postmodern pandemic: 25 years of HIV/AIDS. J Intern Med 263, 218–243.
Kiefer, D., Pitluk, J., Klunk, K. 2009. An overview of CAM: components and clinical uses. Nutr Clin Pract 24, 549–559.
Lang, P.T., Brozell, S.R., Mukherjee, S., Pettersen, E.F., Meng, E.C., Thomas, V., Rizzo, R.C., Case, D.A., James, T.L., Kuntz, I.D. 2009. DOCK 6: Combining techniques to model RNA-small molecule complexes. RNA 15, 1219–1230.
Lawn, S.D. 2004. AIDS in Africa: the impact of coinfections on the pathogenesis of HIV-1 infection. J Infect 48, 1–12.
Li, B.Q., Fu, T., Dongyan, Y., Mikovits, J.A., Ruscetti, F.W., Wang, J.M. 2000. Flavonoid baicalin inhibits HIV-1 infection at the level of viral entry. Biochem Biophys Res Commun 276, 534–538.
Lubkowski, J., Yang, F., Alexandratos, J., Wlodawer, A., Zhao, H., Burke, T.R.J., Neamati, N., Pommier, Y., Merkel, G., Skalka, A.M. 1998. Structure of the catalytic domain of avian sarcoma virus integrase with a bound HIV-1 integrase-targeted inhibitor. Proc Natl Acad Sci USA 95, 4831–4836.
Marchand, C., Maddali, K., Métifiot, M., Pommier, Y. 2009. HIV-1 IN inhibitors: 2010 update and perspectives. Curr Top Med Chem 9, 1016–1037.
Matsushita, S. 2000. Current status and future issues in the treatment of HIV-1 infection. Int J Hematol 72, 20–27.
McColl, D.J., Chen, X. 2010. Strand transfer inhibitors of HIV-1 integrase: Bringing IN a new era of antiretroviral therapy. Antiviral Res 85, 101–118.
Morgan, D., Mahe, C., Mayanja, B., Okongo, J.M., Lubega, R., Whitworth, J.A. 2002. HIV-1 infection in rural Africa: Is there a difference in median time to AIDS and survival compared with that in industrialized countries? AIDS 16, 597–603.
Moszynski, P. 2009. New HIV drug patent pool “offers hope to millions”. BMJ 339, b5557.
Moustakas, D.T., Lang, P.T., Pegg, S., Pettersen, E., Kuntz, I.D., Brooijmans, N., Rizzo, R.C. 2006. Development and validation of a modular, extensible docking program: DOCK 5. J Comput Aided Mol Des 20, 601–619.
Nakanjako, D., Colebunders, R., Coutinho, A.G., Kamya, M.R. 2009. Strategies to optimize HIV treatment outcomes in resource-limited settings. AIDS Rev 11, 179–189.
Robinson, W.E.J., Cordeiro, M., Abdel-Malek, S., Jia, Q., Chow, S.A., Reinecke, M.G., Mitchell, W.M. 1996. Dicaffeoylquinic acid inhibitors of human immunodeficiency virus integrase: Inhibition of the core catalytic domain of human immunodeficiency virus integrase. Mol Pharmacol 50, 846–855.
Schafer, J.J., Squires, K.E. 2010. Integrase inhibitors: A novel class of antiretroviral agents. Ann Pharmacother 44, 145–156.
Schneider, M.F., Gange, S.J., Williams, C.M., Anastos, K., Greenblatt, R.M., Kingsley, L., Detels, R., Muñoz, A. 2005. Patterns of the hazard of death after AIDS through the evolution of antiretroviral therapy: 1984–2004. AIDS 19, 2009–2018.
Tanaka, R., Tsujii, H., Yamada, T., Kajimoto, T., Amano, F., Hasegawa, J., Hamashima, Y., Node, M., Katoh, K., Takebe, Y. 2009. Novel 3alphamethoxyserrat-14-en-21beta-ol (PJ-1) and 3betamethoxyserrat-14-en-21beta-ol (PJ-2)-curcumin, kojic acid, quercetin, and baicalein conjugates as HIV agents. Bioorg Med Chem 17, 5238–5246.
Thomsen, R., Christensen, M.H. 2006. MolDock: A new technique for high-accuracy molecular docking. J Med Chem 49, 3315–3321.
UNAIDS, WHO (December 2007). “2007 AIDS epidemic update” (PDF). http://data.unaids.org/pub/EPISlides/2007/2007_epiupdate_en.pdf. Retrieved on 12 February 2009.
Wang, J.F., Wei, D.Q., Chou, K.C. 2008. Drug candidates from traditional Chinese medicines. Curr Top Med Chem 8, 1656–1665.
Weiss, R.A. 1993. How does HIV cause AIDS? Science 260, 1273–1279.
Wu, J.A., Attele, A.S., Zhang, L., Yuan, C.S. 2001. Anti-HIV activity of medicinal herbs: usage and potential development. Am J Chin Med 29, 69–81.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hu, J.Z., Bai, L., Chen, DG. et al. Computational investigation of the Anti-HIV activity of Chinese medicinal formula Three-Huang Powder. Interdiscip Sci Comput Life Sci 2, 151–156 (2010). https://doi.org/10.1007/s12539-010-0074-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12539-010-0074-1