Abstract
Purpose
The purpose of this study was to evaluate the effect of transportation at prolonged low temperatures on the survival of pre-antral follicles.
Methods
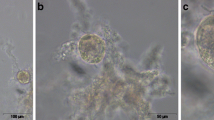
Ovarian tissue was removed from six women with gender identity disorder. Tissues were stored in an icebox at 4 °C for 6 or 18 h prior to vitrification. After warming, ovarian tissues were cultured for 24 h and follicle survival was assessed via a viability/cytotoxicity kit. Morphological features and oxygen consumption rate (OCR) were evaluated by scanning electrochemical microscopy (SECM).
Results
Survival rate of isolated primordial follicles was 95.7 and 100 %, and that of primary follicles was 91.7 and 81.8 % in the 6- and 18-h groups respectively. There was no difference in morphology between the 6- and 18-h storage groups. In comparison with OCR of vitrified-warmed follicles and OCR of 24-h culture after vitrified-warmed follicles, OCR of 24-h culture after vitrified-warmed primordial follicles was significantly higher in both 6-hour (0.02 ± 0.02 vs 0.07 ± 0.04, P < 0.05) and 18-h groups (0.02 ± 0.02 vs 0.11 ± 0.10, P < 0.05).
Conclusions
This strongly suggests that prolonged transportation of ovarian tissue at low temperatures is useful when there are no available local systems for fertility preservation.


Similar content being viewed by others
References
Jemal A, Clegg LX, Ward E, Ries LA, Wu X, Jamison PM, et al. Annual report to the nation on the status of cancer, 1975–2001, with a special feature regarding survival. Cancer. 2004;101:3–27.
Donnez J, Dolmans MM, Demylle D, Jadoul P, Pirard C, Squifflet J, et al. Livebirth after orthotopic transplantation of cryopreserved ovarian tissue. Lancet. 2004;364:1405–10.
Jeruss JS, Woodruff TK. Preservation of fertility in patients with cancer. N Engl J Med. 2009;360:902–11.
Kolp LA, Hubayter Z. Autotransplantation of cryopreserved ovarian tissue: a procedure with promise, risks, and a need for a registry. Fertil Steril. 2011;95:1879–86.
Donnez J, Dolmans MM. Preservation of fertility in females with haematological malignancy. Br J Haematol. 2011;154:75–184.
Donnez J, Jadoul P, Pirard C, Hutchings G, Demylle D, Squifflet J, Smitz J, Dolmans MM. Live birth after transplantation of frozen-thawed ovarian tissue after bilateral oophorectomy for benign disease. Fertil Steril. 2012;98:720–5.
Revel A, Laufer N, Ben Meir A, Lebovich M, Mitran E. Micro-organ ovarian transplantation enables pregnancy: a case report. Hum Reprod. 2011;26:1097–103.
Silbers S, Kagawa N, Kuwayama M, Gosden R. Duration of fertility after fresh and frozen ovary transplantation. Fertil Steril. 2010;94:2191–6.
Ferguson EM, Leese HJ. A potential role for triglyceride as an energy source during bovine oocyte maturation and early embryo development. Mol Reprod Dev. 2006;73:1195–201.
Magnusson C, Hillensjö T, Hamberger L, Nilsson L. Oxygen consumption by human oocytes and blastocysts grown in vitro. Hum Reprod. 1986;1:183–4.
Preis KA, Seidel GE Jr, Gardner DK. Reduced oxygen concentration improves the developmental competence of mouse oocytes following in vitro maturation. Mol Reprod Dev. 2007;74:893–903.
Tejera A, Herrero J, de Los Santos MJ, Garrido N, Ramsing N, Meseguer M. Oxygen consumption is a quality marker for human oocyte competence conditioned by ovarian stimulation regimens. Fertil Steril. 2011;96:618–23.
Kagawa N, Silber S, Kuwayama M. Successful vitrification of bovine and human ovarian tissue. RBM Online. 2009;18:568–77.
Shiku H, Shiraishi T, Aoyagi S, Utsumi Y, Matsudaira M, Abe H, et al. Respiration activity of single bovine embryos entrapped in a cone-shaped microwell monitored by scanning electrochemical microscopy. Anal Chim Acta. 2004;522:51–8.
Van den Hurk R, Spek ER, Hage WJ, Fair T, Ralph JH, Schotanus K. Ultrastructure and viability of isolated bovine preantral follicles. Hum Reprod Update. 1998;4:833–41.
Benos DJ, Baladan RS. Energy metabolism of preimplantation mammalian blastocysts. Am J Physiol. 1983;245:40–5.
Leese HJ. What does an embryo need? Hum Fertil. 2003;6:180–5.
Abe H, Shiku H, Yokoo M, et al. Evaluating the quality of individual embryos with a non-invasive and highly sensitive measurement of oxygen consumption by scanning electrochemical microscopy. J Reprod Dev. 2006;52(Suppl):55–64.
Lopes AS, Madsen SE, Ramsing NB, Løvendahl P, Greve T, Callesen H. Investigation of respiration of individual bovine embryos produced in vivo and in vitro and correlation with viability following transfer. Hum Reprod. 2007;22:558–66.
Sakagami N, Yamamoto T, Akiyama K, Nakazawa Y, Kojima N, Nishida K, Yokomizo S, Takagi Y, Abe H, Suzuki C, et al. Viability of porcine embryos after vitrification using water-soluble pullulan films. J Reprod Dev. 2010;56:279–84.
Yamanaka M, Hashimoto S, Amo A, Ito-Sasaki T, Abe H, Morimoto Y. Developmental assessment of human vitrified-warmed blastocysts based on oxygen consumption. Hum Reprod. 2011;26:3366–71.
Meirow D, Levron J, Eldar-Geva T, Hardan I, Fridman E, Zalel Y, Schiff E, Dor J. Pregnancy after transplantation of cryopreserved ovarian tissue in a patient with ovarian failure after chemotherapy. N Engl J Med. 2005;353:318–21.
Andersen CY, Rosendahl M, Byskov AG, Loft A, Ottosen C, Dueholm M, Schmidt KL, Andersen AN, Ernst E. Two successful pregnancies following autotransplantation of frozen/thawed ovarian tissue. Hum Reprod. 2008;23:2266–72.
Silber SJ, DeRosa M, Pineda J, et al. A series of monozygotic twins discordant for ovarian failure: ovary transplantation (cortical versus microvascular) and cryopreservation. Hum Reprod. 2008;23:1531–7.
Kuleshova LL, Lopata A. Vitrification can be more favourable than slow cooling. Fertil Steril. 2002;78:449–54.
Courbiere B, Odagescu V, Baudot A, Massardier J, Mazoyer C, Salle B, Lornage J. Cryopreservation of the ovary by vitrification as an alternative to slow-cooling protocols. Fertil Steril. 2006;86((Suppl 3)):1243–51.
Schmidt KL, Ernst E, Byskov AG, Andersen AN, Andersen CY. Survival of primordial follicles following prolonged transportation of ovarian tissue prior to cryopreservation. Hum Reprod. 2003;18:2654–9.
Schmidt KT, Rosendahl M, Ernst E, Loft A, Andersen AN, Dueholm M, Ottosen C, Andersen CY. Autotransplantation of cryopreserved ovarian tissue in 12 women with chemotherapy-induced premature ovarian failure: the Danish experience. Fertil Steril. 2011;95:695–701.
Dittrich R, Lotz L, Kech G, Hoffmann I, Mueller A, Bechmann MW, Van der Ven H, Montag M. Live birth after ovarian tissue autotransplantation following overnight transportation before cryopreservation. Fertil Steril. 2012;97:387–90.
Rosendahl M, Schmidt KT, Ernst E, Rasmussen PE, Loft A, Byskov AG, et al. Cryopreservation of ovarian tissue for a decade in Denmark: a view of the technique. RBM Online. 2011;22:162–71.
Isachenko E, Isachenko V, Nawroth F, Rahimi G, Weiss JM. Effect of long-term exposure at suprazero temperatures on activity and viability of human ovarian cortex. Fertil Steril. 2009;91:1556–9.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Kyoya, T., Nakamura, Y., Miyatani, S. et al. Evaluation of oxygen consumption in human vitrified and warmed pre-antral follicles after prolonged low temperatures. Reprod Med Biol 13, 47–52 (2014). https://doi.org/10.1007/s12522-013-0166-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12522-013-0166-9