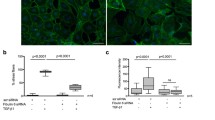
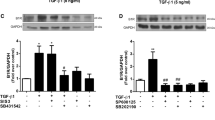
Abstract
Follistatins are extracellular inhibitors of the TGF-β family ligands including activin A, myostatin and bone morphogenetic proteins. Follistatin-like 3 (FSTL3) is a potent inhibitor of activin signalling and antagonises the cardioprotective role of activin A in the heart. FSTL3 expression is elevated in patients with heart failure and is upregulated in cardiomyocytes by hypertrophic stimuli, but its role in cardiac remodelling is largely unknown. Here, we show that the production of FSTL3 by cardiomyocytes contributes to the paracrine activation of cardiac fibroblasts, inducing changes in cell adhesion, promoting proliferation and increasing collagen production. We found that FSTL3 is necessary for this response and for the induction of cardiac fibrosis. However, full activation requires additional factors, and we identify connective tissue growth factor as a FSTL3 binding partner in this process. Together, our data unveil a novel mechanism of paracrine communication between cardiomyocytes and fibroblasts that may provide potential as a therapeutic target in heart remodelling.






Similar content being viewed by others
References
Spann, J. F., Bove, A. A., Natarajan, G., & Kreulen, T. (1980). Ventricular performance, pump function and compensatory mechanisms in patients with aortic stenosis. Circulation, 62, 576–582.
Grossman, W., Jones, D., & Mclaurin, L. P. (1975). Wall stress and patterns of hypertrophy in the human left ventricle. Journal of Clinical Investigation, 56, 56–64.
Sasayama, S., Ross, J., Jr., Franklin, D., Bloor, C. M., Bishop, S., & Dilley, R. B. (1976). Adaptations of the left ventricle to chronic pressure overload. Circulation Research, 38, 172–178.
Pluim, B. M., Zwinderman, A. H., Van Der Laarse, A., & Van Der Wall, E. E. (2000). The athlete’s heart: a meta-analysis of cardiac structure and function. Circulation, 101, 336–344.
Wikman-Coffelt, J., Parmley, W., & Mason, D. (1979). The cardiac hypertrophy process. Analyses of factors determining pathological vs. physiological development. Circulation Research, 45, 697–707.
Scott-Burden, T. (1994). Extracellular matrix: the cellular environment. Physiology, 9, 110–115.
Weber, K., Janicki, J., Shroff, S., Pick, R., Chen, R., & Bashey, R. (1988). Collagen remodeling of the pressure-overloaded, hypertrophied nonhuman primate myocardium. Circulation Research, 62, 757–765.
Norton, G. R., Woodiwiss, A. J., Gaasch, W. H., Mela, T., Chung, E. S., Aurigemma, G. P., & Meyer, T. E. (2002). Heart failure in pressure overload hypertrophy: the relative roles of ventricular remodeling and myocardial dysfunction. Journal of the American College of Cardiology, 39, 664–671.
Brower, G. L., Gardner, J. D., Forman, M. F., Murray, D. B., Voloshenyuk, T., Levick, S. P., & Janicki, J. S. (2006). The relationship between myocardial extracellular matrix remodeling and ventricular function. European Journal of Cardio-Thoracic Surgery, 30, 604–610.
Xia, Y., & Schneyer, A. L. (2009). The biology of activin: recent advances in structure, regulation and function. Journal of Endocrinology, 202, 1–12.
Yndestad, A., Ueland, T., Oie, E., Florholmen, G., Halvorsen, B., Attramadal, H., Simonsen, S., Froland, S. S., et al. (2004). Elevated levels of activin A in heart failure: potential role in myocardial remodeling. Circulation, 109, 1379–1385.
Mahmoudabady, M., Mathieu, M., Dewachter, L., Hadad, I., Ray, L., Jespers, P., Brimioulle, S., Naeije, R., et al. (2008). Activin-A, transforming growth factor-[beta], and myostatin signaling pathway in experimental dilated cardiomyopathy. Journal of Cardiac Failure, 14, 703–709.
Oshima, Y., Ouchi, N., Shimano, M., Pimentel, D. R., Papanicolaou, K. N., Panse, K. D., Tsuchida, K., Lara-Pezzi, E., et al. (2009). Activin A and follistatin-like 3 determine the susceptibility of heart to ischemic injury. Circulation, 120, 1606–1615.
Shimano, M., Ouchi, N., Nakamura, K., Oshima, Y., Higuchi, A., Pimentel, D. R., Panse, K. D., Lara-Pezzi, E., et al. (2011). Cardiac myocyte-specific ablation of follistatin-like 3 attenuates stress-induced myocardial hypertrophy. Journal of Biological Chemistry, 286, 9840–9848.
Schneyer, A., Schoen, A., Quigg, A., & Sidis, Y. (2003). Differential binding and neutralization of activins A and B by follistatin and follistatin like-3 (FSTL-3/FSRP/FLRG). Endocrinology, 144, 1671–1674.
Bartholin, L., Maguer-Satta, V., Hayette, S., Martel, S., Gadoux, M., Bertrand, S., Corbo, L., Lamadon, C., et al. (2001). FLRG, an activin-binding protein, is a new target of TGFbeta transcription activation through Smad proteins. Oncogene, 20, 5409–5419.
Bartholin, L., Maguer-Satta, V., Hayette, S., Martel, S., Gadoux, M., Corbo, L., Magaud, J. P., & Rimokh, R. (2002). Transcription activation of FLRG and follistatin by activin A, through Smad proteins, participates in a negative feedback loop to modulate activin A function. Oncogene, 21, 2227–2235.
Lara-Pezzi, E., Felkin, L. E., Birks, E. J., Sarathchandra, P., Panse, K. D., George, R., Hall, J. L., Yacoub, M. H., et al. (2008). Expression of follistatin-related genes is altered in heart failure. Endocrinology, 149, 5822–5827.
Willoughby, D. S. (2004). Effects of heavy resistance training on myostatin mRNA and protein expression. Medicine & Science in Sports & Exercise, 36, 574–582.
Lara-Pezzi, E., Terracciano, C., Soppa, G., Smolenski, R., Felkin, L., Yacoub, M., & Barton, P. (2009). A gene expression profile of the myocardial response to clenbuterol. Journal of Cardiovascular Translational Research, 2, 191–197.
Felkin, L., Lara-Pezzi, E., Hall, J., Birks, E., & Barton, P. (2011). Reverse remodelling and recovery from heart failure are associated with complex patterns of gene expression. Journal of Cardiovascular Translational Research, 4, 321–331.
Birks, E., & George, R. (2010). Molecular changes occurring during reverse remodelling following left ventricular assist device support. Journal of Cardiovascular Translational Research, 3, 635–642.
Toraason, M., Luken, M. E., Breitenstein, M., Krueger, J. A., & Biagini, R. E. (1989). Comparative toxicity of allylamine and acrolein in cultured myocytes and fibroblasts from neonatal rat heart. Toxicology, 56, 107–117.
Lara-Pezzi, E., Winn, N., Paul, A., Mccullagh, K., Slominsky, E., Santini, M. P., Mourkioti, F., Sarathchandra, P., et al. (2007). A naturally occurring calcineurin variant inhibits FoxO activity and enhances skeletal muscle regeneration. The Journal of Cell Biology, 179, 1205–1218.
Lara-Pezzi, E., Majano, P. L., Yáñez-Mo, M., Gómez-Gonzalo, M., Carretero, M., Moreno-Otero, R., Sanchez-Madrid, F., & Lopez-Cabrera, M. (2001). Effect of the hepatitis B virus HBx protein on integrin-mediated adhesion to and migration on extracellular matrix. Journal of Hepatology, 34, 409–415.
Lara-Pezzi, E., Gomez-Gaviro, M. V., Galvez, B. G., Mira, E., Iniguez, M. A., Fresno, M., Martinez-A, C., Arroyo, A. G., et al. (2002). The hepatitis B virus X protein promotes tumor cell invasion by inducing membrane-type matrix metalloproteinase-1 and cyclooxygenase-2 expression. The Journal of Clinical Investigation, 110, 1831–1838.
Gómez-Gaviro, M. V., Scott, C. E., Sesay, A. K., Matheu, A., Booth, S., Galichet, C., & Lovell-Badge, R. (2012). Betacellulin promotes cell proliferation in the neural stem cell niche and stimulates neurogenesis. Proceedings of the National Academy of Sciences of the United States of America, 109, 1317–1322.
Felkin, L. E., Narita, T., Germack, R., Shintani, Y., Takahashi, K., Sarathchandra, P., López-Olañeta, M. M., Gómez-Salinero, J. M., et al. (2011). Calcineurin splicing variant CnAβ1 improves cardiac function after myocardial infarction without inducing hypertrophy. Circulation, 123, 2838–2847.
Bochmann, L., Sarathchandra, P., Mori, F., Lara-Pezzi, E., Lazzaro, D., & Rosenthal, N. (2010). Revealing new mouse epicardial cell markers through transcriptomics. PLoS One, 5, e11429.
Felkin, L. E., Lara-Pezzi, E., George, R., Yacoub, M. H., Birks, E. J., & Barton, P. J. (2009). Expression of extracellular matrix genes during myocardial recovery from heart failure following left ventricular assist device (LVAD) support. The Journal of Heart and Lung Transplantation, 28, 117–122.
Csiszar, K. (2001). Lysyl oxidases: a novel multifunctional amine oxidase family. Progress in Nucleic Acid Research and Molecular Biology, 70, 1–32.
Mcdonald, J. A., Kelley, D. G., & Broekelmann, T. J. (1982). Role of fibronectin in collagen deposition: Fab’ to the gelatin-binding domain of fibronectin inhibits both fibronectin and collagen organization in fibroblast extracellular matrix. The Journal of Cell Biology, 92, 485–492.
Spinale, F. G. (2002). Matrix metalloproteinases: regulation and dysregulation in the failing heart. Circulation Research, 90, 520–530.
Maguer-Satta, V., Forissier, S., Bartholin, L., Martel, S., Jeanpierre, S., Bachelard, E., & Rimokh, R. (2006). A novel role for fibronectin type I domain in the regulation of human hematopoietic cell adhesiveness through binding to follistatin domains of FLRG and follistatin. Experimental Cell Research, 312, 434–442.
Matsui, Y., & Sadoshima, J. (2004). Rapid upregulation of CTGF in cardiac myocytes by hypertrophic stimuli: implication for cardiac fibrosis and hypertrophy. Journal of Molecular and Cellular Cardiology, 37, 477–481.
Hayata, N., Fujio, Y., Yamamoto, Y., Iwakura, T., Obana, M., Takai, M., Mohri, T., Nonen, S., et al. (2008). Connective tissue growth factor induces cardiac hypertrophy through Akt signaling. Biochemical and Biophysical Research Communications, 370, 274–278.
Panek, A. N., Posch, M. G., Alenina, N., Ghadge, S. K., Erdmann, B., Popova, E., Perrot, A., Geier, C., et al. (2009). Connective tissue growth factor overexpression in cardiomyocytes promotes cardiac hypertrophy and protection against pressure overload. PLoS One, 4, e6743.
Koitabashi, N., Danner, T., Zaiman, A. L., Pinto, Y. M., Rowell, J., Mankowski, J., Zhang, D., Nakamura, T., et al. (2011). Pivotal role of cardiomyocyte TGF-β signaling in the murine pathological response to sustained pressure overload. Journal of Clinical Investigation, 121, 2301–2312.
Shioi, T., Kang, P. M., Douglas, P. S., Hampe, J., Yballe, C. M., Lawitts, J., Cantley, L. C., & Izumo, S. (2000). The conserved phosphoinositide 3-kinase pathway determines heart size in mice. EMBO Journal, 19, 2537–2548.
Sarkar, S., Vellaichamy, E., Young, D., & Sen, S. (2004). Influence of cytokines and growth factors in ANG II-mediated collagen upregulation by fibroblasts in rats: role of myocytes. American Journal of Physiology—Heart and Circulatory Physiology, 287, H107–H117.
Stawowy, P., Margeta, C., Blaschke, F., Lindschau, C., Spencer-Hänsch, C., Leitges, M., Biagini, G., Fleck, E., et al. (2005). Protein kinase C epsilon mediates angiotensin II-induced activation of β1-integrins in cardiac fibroblasts. Cardiovascular Research, 67, 50–59.
Sano, M., Fukuda, K., Kodama, H., Pan, J., Saito, M., Matsuzaki, J., Takahashi, T., Makino, S., et al. (2000). Interleukin-6 family of cytokines mediate angiotensin II-induced cardiac hypertrophy in rodent cardiomyocytes. Journal of Biological Chemistry, 275, 29717–29723.
Tsuruda, T., Jougasaki, M., Boerrigter, G., Huntley, B. K., Chen, H. H., D’assoro, A. B., Lee, S. C., Larsen, A. M., et al. (2002). Cardiotrophin-1 stimulation of cardiac fibroblast growth: roles for glycoprotein 130/leukemia inhibitory factor receptor and the endothelin type a receptor. Circulation Research, 90, 128–134.
Oshima, Y., Ouchi, N., Sato, K., Izumiya, Y., Pimentel, D. R., & Walsh, K. (2008). Follistatin-like 1 is an Akt-regulated cardioprotective factor that is secreted by the heart. Circulation, 117, 3099–3108.
Ouchi, N., Oshima, Y., Ohashi, K., Higuchi, A., Ikegami, C., Izumiya, Y., Walsh, K. (2008). Follistatin-like 1, a secreted muscle protein, promotes endothelial cell function and revascularization in ischemic tissue through a nitric oxide synthesis-dependent mechanism. Journal of Biological Chemistry, 283, 32802–32811.
Sadoshima, J.-I., Xu, Y., Slayter, H. S., & Izumo, S. (1993). Autocrine release of angiotensin II mediates stretch-induced hypertrophy of cardiac myocytes in vitro. Cell, 75, 977–984.
Yamazaki, T., Komuro, I., Kudoh, S., Zou, Y., Shiojima, I., Hiroi, Y., Mizuno, T., Maemura, K., et al. (1996). Endothelin-1 is involved in mechanical stress-induced cardiomyocyte hypertrophy. Journal of Biological Chemistry, 271, 3221–3228.
Butt, R. P., & Bishop, J. E. (1997). Mechanical load enhances the stimulatory effect of serum growth factors on cardiac fibroblast procollagen synthesis. Journal of Molecular and Cellular Cardiology, 29, 1141–1151.
Van Wamel, A. J. E. T., Ruwhof, C., Van Der Valk-Kokshoorn, L. E. J. M., Schriern, P. I., & Van Der Laarse, A. (2001). The role of angiotensin II, endothelin-1 and transforming growth factor-β as autocrine/paracrine mediators of stretch-induced cardiomyocyte hypertrophy. Molecular and Cellular Biochemistry, 218, 113–124.
Van Wamel, A. J. E. T., Ruwhof, C., Van Der Valk-Kokshoorn, L. J. M., Schrier, P. I., & Van Der Laarse, A. (2002). Stretch-induced paracrine hypertrophic stimuli increase TGF-β expression in cardiomyocytes. Molecular and Cellular Biochemistry, 236, 147–153.
Yokoyama, T., Sekiguchi, K., Tanaka, T., Tomaru, K., Arai, M., Suzuki, T., & Nagai, R. (1999). Angiotensin II and mechanical stretch induce production of tumor necrosis factor in cardiac fibroblasts. American Journal of Physiology—Heart and Circulatory Physiology, 276, H1968–H1976.
Lindahl, G. E., Chambers, R. C., Papakrivopoulou, J., Dawson, S. J., Jacobsen, M. C., Bishop, J. E., & Laurent, G. J. (2002). Activation of fibroblast procollagen α1(I) transcription by mechanical strain is transforming growth factor-β-dependent and involves increased binding of CCAAT-binding factor (CBF/NF-Y) at the proximal promoter. Journal of Biological Chemistry, 277, 6153–6161.
Chiquet, M., Gelman, L., Lutz, R., & Maier, S. (2009). From mechanotransduction to extracellular matrix gene expression in fibroblasts. Biochimica et Biophysica Acta, 1793, 911–920.
Ahmed, M. S., Gravning, J., Martinov, V. N., Von Lueder, T. G., Edvardsen, T., Czibik, G., Moe, I. T., Vinge, L. E., et al. (2011). Mechanisms of novel cardioprotective functions of CCN2/CTGF in myocardial ischemia–reperfusion injury. American Journal of Physiology—Heart and Circulatory Physiology, 300, H1291–H1302.
Kadokami, T., Frye, C., Lemster, B., Wagner, C. L., Feldman, A. M., & Mctiernan, C. F. (2001). Anti-tumor necrosis factor-{alpha} antibody limits heart failure in a transgenic model. Circulation, 104, 1094–1097.
Kuwahara, F., Kai, H., Tokuda, K., Kai, M., Takeshita, A., Egashira, K., & Imaizumi, T. (2002). Transforming growth factor-{beta} function blocking prevents myocardial fibrosis and diastolic dysfunction in pressure-overloaded rats. Circulation, 106, 130–135.
Acknowledgments
This work was supported by a British Heart Foundation grant (PG/08/084/25827) to P.B., N.R. and E.L.P. In addition, PB was supported by Heart Research UK and by the National Institute for Health Research Cardiovascular Biomedical Research Unit at the Royal Brompton and Harefield NHS Foundation Trust and Imperial College. E.L.P. was supported by grants from the European Union (ERG-239158, ITN-289600), the Spanish Ministry of Science and Innovation (BFU2009-10016, CP08/00144) and the Regional Government of Madrid (S2010/BMD-2321 ‘Fibroteam’).
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 828 kb)
Rights and permissions
About this article
Cite this article
Panse, K.D., Felkin, L.E., López-Olañeta, M.M. et al. Follistatin-Like 3 Mediates Paracrine Fibroblast Activation by Cardiomyocytes. J. of Cardiovasc. Trans. Res. 5, 814–826 (2012). https://doi.org/10.1007/s12265-012-9400-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12265-012-9400-9