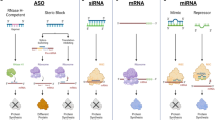
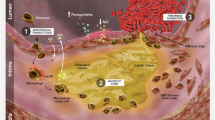
Abstract
Globally, one of the major causes of death is the cardiovascular disease (CVD), and platelets play an important role in thrombosis and atherosclerosis that led to death. Platelet activation can be done by different molecules, genes, pathways, and chemokines. Lipids activate platelets by inflammatory factors, and platelets are activated by receptors of peptide hormones, signaling and secreted proteins, microRNAs (miRNAs), and oxidative stress which also affect the platelet activation in older age. In addition, surface molecules on platelets can interact with other cells and chemokines in activated platelets and cause inflammation thrombosis events and CVD. However, these molecules activating platelets or being activated by platelets can be suggested as the markers to predict the clinical outcome of CVD and can be targeted to reduce thrombosis and atherosclerosis. However, hindering these molecules by other factors such as genes and receptors can reduce platelet activation and aggregation and targeting these molecules can control platelet interactions, thrombosis, and CVD. In addition, dual therapy with the receptor blockers and novel drugs results in better management of CVD patients. Overall, our review will emphasize on the molecules involved in the activation of platelets and on the molecules that are activated by platelets in CVD and discuss the molecules that can be blocked or targeted to reduce the thrombosis events and control CVD.

Similar content being viewed by others
References
Fuentes, F., Palomo, I., & Fuentes, E. (2017). Platelet oxidative stress as a novel target of cardiovascular risk in frail older people. Vascular Pharmacology,93, 14–19.
Haybar, H., Khodadi, E., Zibara, K., & Saki, N. (2018). Platelet activation polymorphisms in ischemia. Cardiovascular & Hematological Disorders: Drug Targets,18(2), 153–161.
Montenont, E., Echagarruga, C., Allen, N., Araldi, E., Suarez, Y., & Berger, J. S. (2016). Platelet WDR1 suppresses platelet activity and associates with cardiovascular disease. Blood,128(16), 2033–2042.
Chatterjee, M., Rath, D., Schlotterbeck, J., Rheinlaender, J., Walker-Allgaier, B., Alnaggar, N., et al. (2017). Regulation of oxidized platelet lipidome: Implications for coronary artery disease. European Heart Journal,38(25), 1993–2005.
Elbatarny, H. S., Netherton, S. J., Ovens, J. D., Ferguson, A. V., & Maurice, D. H. (2007). Adiponectin, ghrelin, and leptin differentially influence human platelet and human vascular endothelial cell functions: Implication in obesity-associated cardiovascular diseases. European Journal of Pharmacology,558(1–3), 7–13.
Lepropre, S., Kautbally, S., Octave, M., Ginion, A., Onselaer, M.-B., Steinberg, G. R., et al. (2018). AMPK-ACC signaling modulates platelet phospholipids and potentiates thrombus formation. Blood,132(11), 1180–1192.
A Gleissner, C. (2012). Platelet-derived chemokines in atherogenesis: What’s new? Current Vascular Pharmacology,10(5), 563–569.
Brandt, E., Ludwig, A., Petersen, F., & Flad, H. D. (2000). Platelet-derived CXC chemokines: Old players in new games. Immunological Reviews,177(1), 204–216.
Masselli, E., Carubbi, C., Pozzi, G., Martini, S., Aversa, F., Galli, D., et al. (2017). Platelet expression of PKCepsilon oncoprotein in myelofibrosis is associated with disease severity and thrombotic risk. Annals of Translational Medicine,5(13), 273.
Parguina, A. F., Grigorian-Shamajian, L., Agra, R. M., Teijeira-Fernandez, E., Rosa, I., Alonso, J., et al. (2010). Proteins involved in platelet signaling are differentially regulated in acute coronary syndrome: A proteomic study. PLoS ONE,5(10), e13404.
Gurbel, P. A., Fox, K. A., Tantry, U. S., ten Cate, H., & Weitz, J. I. (2019). Combination antiplatelet and oral anticoagulant therapy in patients with coronary and peripheral artery disease: Focus on the COMPASS trial. Circulation,139(18), 2170–2185.
Swieringa, F., Spronk, H. M., Heemskerk, J. W., & van der Meijden, P. E. (2018). Integrating platelet and coagulation activation in fibrin clot formation. Research and Practice in Thrombosis and Haemostasis,2(3), 450–460.
Akkerman, J. W. N. (2008). From low-density lipoprotein to platelet activation. The International Journal of Biochemistry & Cell Biology,40(11), 2374–2378.
Salomon, G. (2012). Structural identification and cardiovascular activities of oxidized phospholipids. Circulation Research,111, 930–946.
Podrez, E. A., Byzova, T. V., Febbraio, M., Salomon, R. G., Ma, Y., Valiyaveettil, M., et al. (2007). Platelet CD36 links hyperlipidemia, oxidant stress and a prothrombotic phenotype. Nature Medicine,13(9), 1086.
Chatterjee, M., von Ungern-Sternberg, S., Seizer, P., Schlegel, F., Büttcher, M., Sindhu, N., et al. (2015). Platelet-derived CXCL12 regulates monocyte function, survival, differentiation into macrophages and foam cells through differential involvement of CXCR4–CXCR7. Cell Death & Disease,6(11), e1989.
Chatterjee, M., & Gawaz, M. (2013). Platelet-derived CXCL 12 (SDF-1α): Basic mechanisms and clinical implications. Journal of Thrombosis and Haemostasis,11(11), 1954–1967.
Weber, C. (2005). Platelets and chemokines in atherosclerosis: Partners in crime. Circulation Research,96(6), 612–616.
Rath, D., Chatterjee, M., Borst, O., Müller, K., Stellos, K., Mack, A. F., et al. (2013). Expression of stromal cell-derived factor-1 receptors CXCR4 and CXCR7 on circulating platelets of patients with acute coronary syndrome and association with left ventricular functional recovery. European Heart Journal,35(6), 386–394.
Li, X., Zhu, M., Penfold, M. E., Koenen, R. R., Thiemann, A., Heyll, K., et al. (2014). Activation of CXCR7 limits atherosclerosis and improves hyperlipidemia by increasing cholesterol uptake in adipose tissue. Circulation,129(11), 1244–1253.
Stellos, K., Ruf, M., Sopova, K., Kilias, A., Rahmann, A., Stamatelopoulos, K., et al. (2011). Plasma levels of stromal cell-derived factor-1 in patients with coronary artery disease: Effect of clinical presentation and cardiovascular risk factors. Atherosclerosis,219(2), 913–916.
Kile, B. T., Panopoulos, A. D., Stirzaker, R. A., Hacking, D. F., Tahtamouni, L. H., Willson, T. A., et al. (2007). Mutations in the cofilin partner Aip1/Wdr1 cause autoinflammatory disease and macrothrombocytopenia. Blood,110(7), 2371–2380.
Kueh, H. Y., Charras, G. T., Mitchison, T. J., & Brieher, W. M. (2008). Actin disassembly by cofilin, coronin, and Aip1 occurs in bursts and is inhibited by barbed-end cappers. The Journal of Cell Biology,182(2), 341–353.
Rodal, A. A., Tetreault, J. W., Lappalainen, P., Drubin, D. G., & Amberg, D. C. (1999). Aip1p interacts with cofilin to disassemble actin filaments. The Journal of Cell Biology,145(6), 1251–1264.
Chu, S., Becker, R., Berger, P., Bhatt, D., Eikelboom, J., Konkle, B., et al. (2010). Mean platelet volume as a predictor of cardiovascular risk: A systematic review and meta-analysis. Journal of Thrombosis and Haemostasis,8(1), 148–156.
Karpatkin, S. (1969). Heterogeneity of human platelets: II. Functional evidence suggestive of young and old platelets. The Journal of Clinical Investigation,48(6), 1083–1087.
Elbatarny, H. S., & Maurice, D. H. (2005). Leptin-mediated activation of human platelets: Involvement of a leptin receptor and phosphodiesterase 3A-containing cellular signaling complex. American Journal of Physiology-Endocrinology and Metabolism,289(4), E695–E702.
Michelson, A. D., Barnard, M. R., Krueger, L. A., Valeri, C. R., & Furman, M. I. (2001). Circulating monocyte-platelet aggregates are a more sensitive marker of in vivo platelet activation than platelet surface P-selectin: Studies in baboons, human coronary intervention, and human acute myocardial infarction. Circulation,104(13), 1533–1537.
Trovati, M., & Anfossi, G. (2002). Influence of insulin and of insulin resistance on platelet and vascular smooth muscle cell function. Journal of Diabetes and Its Complications,16(1), 35–40.
Shoji, T., Koyama, H., Fukumoto, S., Maeno, T., Yokoyama, H., Shinohara, K., et al. (2006). Platelet activation is associated with hypoadiponectinemia and carotid atherosclerosis. Atherosclerosis,188(1), 190–195.
Koyama, H., Maeno, T., Fukumoto, S., Shoji, T., Yamane, T., Yokoyama, H., et al. (2003). Platelet P-selectin expression is associated with atherosclerotic wall thickness in carotid artery in humans. Circulation,108(5), 524–529.
Pereira, J., Soto, M., Palomo, I., Ocqueteau, M., Coetzee, L.-M., Astudillo, S., et al. (2002). Platelet aging in vivo is associated with activation of apoptotic pathways: Studies in a model of suppressed thrombopoiesis in dogs. Thrombosis and Haemostasis,87(05), 905–909.
Pastori, D., Pignatelli, P., Carnevale, R., & Violi, F. (2015). Nox-2 up-regulation and platelet activation: Novel insights. Prostaglandins & Other Lipid Mediators,120, 50–55.
Dayal, S., Wilson, K. M., Motto, D. G., Miller, F. J., Jr., Chauhan, A. K., & Lentz, S. R. (2013). Hydrogen peroxide promotes aging-related platelet hyperactivation and thrombosis. Circulation,127, 1308–1316.
Fuentes, E., & Palomo, I. (2016). Role of oxidative stress on platelet hyperreactivity during aging. Life Sciences,148, 17–23.
Carnevale, R., Loffredo, L., Pignatelli, P., Nocella, C., Bartimoccia, S., Di Santo, S., et al. (2012). Dark chocolate inhibits platelet isoprostanes via NOX2 down-regulation in smokers. Journal of Thrombosis and Haemostasis,10(1), 125–132.
Mangiacapra, F., De Bruyne, B., Muller, O., Trana, C., Ntalianis, A., Bartunek, J., et al. (2010). High residual platelet reactivity after clopidogrel: Extent of coronary atherosclerosis and periprocedural myocardial infarction in patients with stable angina undergoing percutaneous coronary intervention. Cardiovascular Interventions.,3(1), 35–40.
Parodi, G., Marcucci, R., Valenti, R., Gori, A. M., Migliorini, A., Giusti, B., et al. (2011). High residual platelet reactivity after clopidogrel loading and long-term cardiovascular events among patients with acute coronary syndromes undergoing PCI. JAMA,306(11), 1215–1223.
Flierl, U., Bauersachs, J., & Schäfer, A. (2015). Modulation of platelet and monocyte function by the chemokine fractalkine (CX 3 CL 1) in cardiovascular disease. European Journal of Clinical Investigation,45(6), 624–633.
Semple, J. W., Italiano, J. E., & Freedman, J. (2011). Platelets and the immune continuum. Nature Reviews Immunology,11(4), 264.
Lievens, D., & von Hundelshausen, P. (2011). Platelets in atherosclerosis. Thrombosis and Haemostasis,105(05), 827–838.
Langer, H. F., Bigalke, B., Seizer, P., Stellos, K., Fateh-Moghadam, S., Gawaz, M. (2010). Interaction of platelets and inflammatory endothelium in the development and progression of coronary artery disease. In Seminars in thrombosis and hemostasis. Thieme Medical Publishers.
Schäfer, A., Schulz, C., Fraccarollo, D., et al. (2007). The CX3C chemokine fractalkine induces vascular dysfunction by generation of superoxide anions. Arteriosclerosis, Thrombosis, and Vascular Biology,27, 55–62.
Schulz, C., Schäfer, A., Stolla, M., et al. (2007). Chemokine fractalkine mediates leukocyte recruitment to inflammatory endothelial cells in flowing whole blood, a critical role for P-selectin expressed on activated platelets. Circulation,116, 764–773.
Schäfer, A., Schulz, C., Eigenthaler, M., Fraccarollo, D., Kobsar, A., Gawaz, M., et al. (2004). Novel role of the membrane-bound chemokine fractalkine in platelet activation and adhesion. Blood,103(2), 407–412.
Postea, O., Vasina, E. M., Cauwenberghs, S., et al. (2012). Contribution of platelet CX(3)CR1 to platelet-monocyte complex formation and vascular recruitment during hyperlipidemia. Arteriosclerosis, Thrombosis, and Vascular Biology,32, 1186–1193.
Flierl, U., Fraccarollo, D., Lausenmeyer, E., Rosenstock, T., Schulz, C., Massberg, S., et al. (2012). Fractalkine activates a signal transduction pathway similar to P2Y12 and is associated with impaired clopidogrel responsiveness. Arteriosclerosis, Thrombosis, and Vascular Biology,32(8), 1832–1840.
García, Á., Senis, Y. A., Antrobus, R., Hughes, C. E., Dwek, R. A., Watson, S. P., et al. (2006). A global proteomics approach identifies novel phosphorylated signaling proteins in GPVI-activated platelets: Involvement of G6f, a novel platelet Grb2-binding membrane adapter. Proteomics,6(19), 5332–5343.
Senis, Y. A., Antrobus, R., Severin, S., Parguina, A. F., Rosa, I., Zitzmann, N., et al. (2009). Proteomic analysis of integrin αIIbβ3 outside-in signaling reveals Src-kinase-independent phosphorylation of Dok-1 and Dok-3 leading to SHIP-1 interactions. Journal of Thrombosis and Haemostasis,7(10), 1718–1726.
Tadokoro, S., Shattil, S. J., Eto, K., Tai, V., Liddington, R. C., de Pereda, J. M., et al. (2003). Talin binding to integrin ß tails: A final common step in integrin activation. Science,302(5642), 103–106.
Honda, S., Shirotani-Ikejima, H., Tadokoro, S., Maeda, Y., Kinoshita, T., Tomiyama, Y., et al. (2009). Integrin-linked kinase associated with integrin activation. Blood,113(21), 5304–5313.
Legate, K. R., Montañez, E., Kudlacek, O., & Füssler, R. (2006). ILK, PINCH and parvin: The tIPP of integrin signalling. Nature Reviews Molecular Cell Biology,7(1), 20.
Tucker, K. L., Sage, T., Stevens, J. M., Jordan, P. A., Jones, S., Barrett, N. E., et al. (2008). A dual role for integrin-linked kinase in platelets: Regulating integrin function and α-granule secretion. Blood,112(12), 4523–4531.
Mallat, Z., Benamer, H., Hugel, B., Benessiano, J., Steg, P. G., Freyssinet, J. M., et al. (2000). Elevated levels of shed membrane microparticles with procoagulant potential in the peripheral circulating blood of patients with acute coronary syndromes. Circulation,101(8), 841–843.
Brill, A., Dashevsky, O., Rivo, J., Gozal, Y., & Varon, D. (2005). Platelet-derived microparticles induce angiogenesis and stimulate post-ischemic revascularization. Cardiovascular Research,67(1), 30–38.
Coppinger, J. A., Cagney, G., Toomey, S., Kislinger, T., Belton, O., McRedmond, J. P., et al. (2004). Characterization of the proteins released from activated platelets leads to localization of novel platelet proteins in human atherosclerotic lesions. Blood,103(6), 2096–2104.
Dobaczewski, M., Gonzalez-Quesada, C., & Frangogiannis, N. G. (2010). The extracellular matrix as a modulator of the inflammatory and reparative response following myocardial infarction. Journal of Molecular and Cellular Cardiology,48(3), 504–511.
Schellings, M. W., Vanhoutte, D., Swinnen, M., Cleutjens, J. P., Debets, J., van Leeuwen, R. E., et al. (2009). Absence of SPARC results in increased cardiac rupture and dysfunction after acute myocardial infarction. Journal of Experimental Medicine,206(1), 113–123.
Shahjahani, M., Khodadi, E., Seghatoleslami, M., Asl, J. M., Golchin, N., Zaieri, Z. D., et al. (2015). Rare cytogenetic abnormalities and alteration of microRNAs in acute myeloid leukemia and response to therapy. Oncology Reviews,9(1), 261.
Khodadi, E., Asnafi, A. A., Mohammadi-Asl, J., Hosseini, S. A., Malehi, A. S., & Saki, N. (2017). Evaluation of miR-21 and miR-150 expression in immune thrombocytopenic purpura pathogenesis: A case-control study. Frontiers in Biology,12(5), 361–369.
Yao, R., Ma, Y., Du, Y., Liao, M., Li, H., Liang, W., et al. (2011). The altered expression of inflammation-related microRNAs with microRNA-155 expression correlates with Th17 differentiation in patients with acute coronary syndrome. Cellular & Molecular Immunology,8(6), 486.
Gatsiou, A., Boeckel, J. N., Randriamboavonjy, V., & Stellos, K. (2012). MicroRNAs in platelet biogenesis and function: Implications in vascular homeostasis and inflammation. Current Vascular Pharmacology,10(5), 524–531.
Widera, C., Gupta, S. K., Lorenzen, J. M., Bang, C., Bauersachs, J., Bethmann, K., et al. (2011). Diagnostic and prognostic impact of six circulating microRNAs in acute coronary syndrome. Journal of Molecular and Cellular Cardiology,51(5), 872–875.
Siasos, G., Kollia, C., Tsigkou, V., Basdra, E. K., Lymperi, M., Oikonomou, E., et al. (2013). MicroRNAs: Novel diagnostic and prognostic biomarkers in atherosclerosis. Current Topics in Medicinal Chemistry,13(13), 1503–1517.
Bartel, D. P. (2009). MicroRNAs: Target recognition and regulatory functions. Cell,136(2), 215–233.
Onselaer, M. B., Oury, C., Hunter, R., Eeckhoudt, S., Barile, N., Lecut, C., et al. (2014). The Ca2 +/calmodulin-dependent kinase kinase β-AMP-activated protein kinase-α1 pathway regulates phosphorylation of cytoskeletal targets in thrombin-stimulated human platelets. Journal of Thrombosis and Haemostasis,12(6), 973–986.
Pula, G., Schuh, K., Nakayama, K., Nakayama, K. I., Walter, U., & Poole, A. W. (2006). PKCδ regulates collagen-induced platelet aggregation through inhibition of VASP-mediated filopodia formation. Blood,108(13), 4035–4044.
Pitsilos, S., Hunt, J., Mohler, E. R., Prabhakar, A. M., Poncz, M., Dawicki, J., et al. (2003). Platelet factor 4 localization in carotid atherosclerotic plaques: Correlation with clinical parameters. Thrombosis and Haemostasis,89(06), 1112–1120.
Von Hundelshausen, P., Koenen, R. R., Sack, M., Mause, S. F., Adriaens, W., Proudfoot, A. E., et al. (2005). Heterophilic interactions of platelet factor 4 and RANTES promote monocyte arrest on endothelium. Blood,105(3), 924–930.
Scheuerer, B., Ernst, M., Dürrbaum-Landmann, I., Fleischer, J., Grage-Griebenow, E., Brandt, E., et al. (2000). The CXC-chemokine platelet factor 4 promotes monocyte survival and induces monocyte differentiation into macrophages. Blood,95(4), 1158–1166.
Von Hundelshausen, P., Weber, K. S., Huo, Y., Proudfoot, A. E., Nelson, P. J., Ley, K., et al. (2001). RANTES deposition by platelets triggers monocyte arrest on inflamed and atherosclerotic endothelium. Circulation,103(13), 1772–1777.
Smith, D. F., Galkina, E., Ley, K., & Huo, Y. (2005). GRO family chemokines are specialized for monocyte arrest from flow. American Journal of Physiology-Heart and Circulatory Physiology,289(5), H1976–H1984.
Abi-Younes, S., Sauty, A., Mach, F., Sukhova, G., Libby, P., & Luster, A. (2000). The stromal cell–derived factor-1 chemokine is a potent platelet agonist highly expressed in atherosclerotic plaques. Circulation Research,86(2), 131–138.
Abi-Younes, S., Si-Tahar, M., & Luster, A. D. (2001). The CC chemokines MDC and TARC induce platelet activation via CCR4. Thrombosis Research,101(4), 279–289.
Gear, A. R., & Camerini, D. (2003). Platelet chemokines and chemokine receptors: Linking hemostasis, inflammation, and host defense. Microcirculation,10(3–4), 335–350.
Lippi, G., Franchini, M., & Targher, G. (2011). Arterial thrombus formation in cardiovascular disease. Nature Reviews Cardiology,8(9), 502.
Strehl, A., Munnix, I. C., Kuijpers, M. J., van der Meijden, P. E., Cosemans, J. M., Feijge, M. A., et al. (2007). Dual role of platelet protein kinase C in thrombus formation stimulation OF pro-aggregatory and suppression of procoagulant activity in plateletS. Journal of Biological Chemistry,282(10), 7046–7055.
Masselli, E., Carubbi, C., Gobbi, G., Mirandola, P., Galli, D., Martini, S., et al. (2015). Protein kinase Cɛ inhibition restores megakaryocytic differentiation of hematopoietic progenitors from primary myelofibrosis patients. Leukemia,29(11), 2192.
Dann, R., Hadi, T., Montenont, E., Boytard, L., Alebrahim, D., Feinstein, J., et al. (2018). Platelet-derived MRP-14 induces monocyte activation in patients with symptomatic peripheral artery disease. Journal of the American College of Cardiology,71(1), 53–65.
Huo, Y., Schober, A., Forlow, S. B., Smith, D. F., Hyman, M. C., Jung, S., et al. (2003). Circulating activated platelets exacerbate atherosclerosis in mice deficient in apolipoprotein E. Nature Medicine,9(1), 61.
Vorchheimer, D. A., Becker, R., eds. (2006). Platelets in atherothrombosis. Mayo Clinic Proceedings; Elsevier.
Wang, Y., Fang, C., Gao, H., Bilodeau, M. L., Zhang, Z., Croce, K., et al. (2014). Platelet-derived S100 family member myeloid-related protein-14 regulates thrombosis. The Journal of Clinical Investigation,124(5), 2160–2171.
Liu, G., Liang, B., Song, X., Bai, R., Qin, W., Sun, X., et al. (2016). P-selectin increases angiotensin II-induced cardiac inflammation and fibrosis via platelet activation. Molecular Medicine Reports,13(6), 5021–5028.
Edelstein, L. C., Simon, L. M., Montoya, R. T., Holinstat, M., Chen, E. S., Bergeron, A., et al. (2013). Racial differences in human platelet PAR4 reactivity reflect expression of PCTP and miR-376c. Nature Medicine,19(12), 1609.
Rao, A. K. (2017). Transcription factor RUNX1 regulates platelet PCTP (phosphatidylcholine transfer protein): Implications for cardiovascular events. Circulation,136(10), 927–939.
Lonial, S., Waller, E. K., Richardson, P. G., Jagannath, S., Orlowski, R. Z., Giver, C. R., et al. (2005). Risk factors and kinetics of thrombocytopenia associated with bortezomib for relapsed, refractory multiple myeloma. Blood,106, 3777–3784.
Nayak, M. K., Kulkarni, P. P., & Dash, D. (2013). Regulatory role of proteasome in determination of platelet life span. Journal of Biological Chemistry,288(10), 6826–6834.
Shen, Y., Zhou, X., Wang, Z., Yang, G., Jiang, Y., Sun, C., et al. (2011). Coagulation profiles and thromboembolic events of bortezomib plus thalidomide and dexamethasone therapy in newly diagnosed multiple myeloma. Leukemia Research,35, 147–151.
Gupta, N., Li, W., Willard, B., Silverstein, R. L., & McIntyre, T. M. (2014). Proteasome proteolysis supports stimulated platelet function and thrombosis. Arteriosclerosis, Thrombosis, and Vascular Biology,34(1), 160–168.
Gurbel, P. A., & Tantry, U. S. (2010). Combination antithrombotic therapies. Circulation,121, 569–583.
Gibson, C. M., Chakrabarti, A. K., Mega, J., Bode, C., Bassand, J.-P., Verheugt, F. W., et al. (2013). Reduction of stent thrombosis in patients with acute coronary syndromes treated with rivaroxaban in ATLAS-ACS 2 TIMI 51. Journal of the American College of Cardiology,62(4), 286–290.
Bhatt, D. L., Fox, K. A., Hacke, W., Berger, P. B., Black, H. R., Boden, W. E., et al. (2006). Clopidogrel and aspirin versus aspirin alone for the prevention of atherothrombotic events. New England Journal of Medicine,354(16), 1706–1717.
Bonaca, M. P., Bhatt, D. L., Cohen, M., Steg, P. G., Storey, R. F., Jensen, E. C., et al. (2015). Long-term use of ticagrelor in patients with prior myocardial infarction. New England Journal of Medicine,372(19), 1791–1800.
Acknowledgements
We wish to thank all our colleagues in Research Center of Thalassemia & Hemoglobinopathy, Health research institute, Ahvaz Jundishapur University of Medical Sciences, Ahvaz, Iran.
Funding
Funding resources were not applicable to this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Handling Editor: Y. James Kang.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Khodadi, E. Platelet Function in Cardiovascular Disease: Activation of Molecules and Activation by Molecules. Cardiovasc Toxicol 20, 1–10 (2020). https://doi.org/10.1007/s12012-019-09555-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12012-019-09555-4