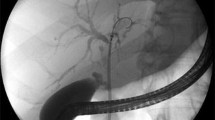
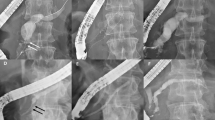
Abstract
Background
Fully-covered, self-expandable metal stents (FCSEMS) have been deployed to treat symptomatic chronic pancreatitis (CP) complicated with main pancreatic duct (MPD) stricture. Although this strategy can be effective, it has the disadvantages of stent migration or stent-induced ductal change. Removal of an FCSEMS can also be challenging in the face of distal migration because of MPD stricture. Dumbbell-type FCSEMS have been developed to prevent stent-induced ductal changes and improve removability when treating benign biliary stricture. This stent might also confer clinical benefits upon patients with MPD stricture.
Aims
The present pilot study aimed to determine the feasibility and safety of deploying dumbbell-type FCSEMS in patients with CP complicated by MPD stricture.
Method
Stents were deployed in 22 patients with MPD stricture caused by CP and complicated by abdominal pain.
Results
Strictures were located at the head (n = 19), body (n = 2), and head and tail (n = 1) of the MPD. Stents were deployed above the papilla in three patients. All stents were deployed for a median duration of 142 (range, 49–190) days and removed. The resolution of MPD strictures was confirmed by pancreatography in 19 (86.3%) patients. Two metal stents that spontaneously tore during removal from two patients had otherwise functioned normally. Only three patients developed recurrent MPD stricture during a median follow-up of 419 (range, 261–484) days..
Conclusions
Deployment of a dumbbell-type FCSEMS seems feasible for MPD stricture, and the rate of adverse events is acceptable.





Similar content being viewed by others
References
Deviere J. Pancreatic stents. Gastrointest Endosc Clin N Am 2011;21:499–510
DiMagno EP, DiMagno MJ. Chronic pancreatitis: Landmark papers, management decisions, and future. Pancreas 2016;45:641–650
Park DH, Kim MH, Moon SH, et al. Feasibility and safety of placement of a newly designed, fully covered self-expandable metal stent for refractory benign pancreatic duct strictures: a pilot study (with video). Gastrointest Endosc 2008;68:1182–1189
Sauer B, Talreja J, Ellen K, et al. Temporary placement of a fully covered self-expandable metal stent in the pancreatic duct for management of symptom refractory chronic pancreatitis: prelim data (with video). Gastrointest Endosc 2008;68:1173–1178
Moon SH, Kim MH, Park DH, et al. Modified fully covered self-expandable metal stents with antimigration features for benign pancreatic-duct strictures in advanced chronic pancreatitis, with a focus on the safety profile and reducing migration. Gastrointest Endosc 2010;72:86–91
Akbar A, Baron TH. Covered self-expanding metal stent use in the pancreatic duct: a case series. Endoscopy 2012;44:869–873
Giacino C, Grandval P, Laugier R. Fully covered self-expandable metal stents for refractory pancreatic duct strictures in chronic pancreatitis. Endoscopy 2012;44:874–877
Ogura T, Onda S, Takagi W, et al. Placement of a 6 mm, fully covered metal stent for main pancreatic head duct stricture due to chronic pancreatitis: a pilot study (with video). Therap Adv Gastroenterol 2016;9:722–728
Matsubara S, Sasahira N, Isayama H, et al. Prospective pilot study of fully covered self-expandable metal stents for refractory benign pancreatic duct strictures: long-term outcomes. Endosc Int Open 2016;4:E1215-E1222
Moon JH, Choi HJ, Koo HC, et al. Feasibility of placing a modified fully covered self-expandable metal stent above the papilla to minimize stent-induced bile duct injury in patients with refractory benign biliary strictures (with videos). Gstrointest Endosc 2012;75:1080–1085
Oh D, Lee JH, Park DH, et al. Long-term outcomes of 6-mm diameter fully covered self-expandable metal stents in benign refractory pancreatic ductal strictures. Dig Endosc 2018.
Cotton PB, Eisen GM, Aabakken L, et al. A lexicon for endoscopic adverse events: report of an ASGE workshop. Gastrointest Endosc 2010;71:746–754
Cahen DL, Gouma DJ, Nio Y, et al. Endoscopic versus surgical drainage of the pancreatic duct in chronic pancreatitis. N Engl J Med 2007;356:676–684
D’Haese JG, Ceyhan GO, Demir IE, et al. Treatment options in painful chronic pancreatitis: a systematic review. HPB(Oxford) 2014;16:512–521
Costamagna G, Bulajic M, Tringali A, et al. Multiple stenting of refractory pancreatic duct strictures in severe chronic pancreatitis: long-term results. Endoscopy 2006;38:254–259
Shen Y, Liu M, Chen M, et al. Covered metal stent or multiple plastic stents for refractory pancreatic ductal strictures in chronic pancreatitis: A systematic review. Pancreatology 2014;14:87–90
Park JS, Lee SS, Song TJ, et al. Long-term outcomes of covered self-expandable metal stents for treating biliary strictures. Endoscopy 2016;48:440–447
Deviere J, Nageshwar Reddy D, Puspok A, et al. Successful management of benign biliary strictures with fully covered self-expandable metal stents. Gastroenterology 2014;147:385–395; quiz e 15
Yan LH, Li L, Chen JS. Chronic pancreatitis. Curr Opin Gastroenterol 2017;33:396–403
Author information
Authors and Affiliations
Contributions
Tadahiro Yamada and Takeshi Ogura wrote a paper. Takeshi Ogura, Rieko Kamiyama, Atsushi Okuda, Nobu Nishioka, Akira Miyano, Miyuki Imanishi, and Kazuhide Higuchi played roles of interpretation of data for the work, revising it critically for important intellectual content, final approval of the version to be published, agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Corresponding author
Ethics declarations
This pilot study proceeded at Osaka Medical College between August 2016 and June 2017 and was approved by the Institutional Review Board at our hospital. All patients provided written, informed consent to participate in the study.
Conflict of Interest
Tadahiro Yamada, Takeshi Ogura, Atsushi Okuda, Miyuki Imanishi, Rieko Kamiyama, Akira Miyano, Nobu Nishioka, and Kazuhide Higuchi declare that there are no conflicts of interest.
Electronic Supplementary Material
Rights and permissions
About this article
Cite this article
Yamada, T., Ogura, T., Okuda, A. et al. Pilot Study of Dumbbell-Type Covered Self-Expandable Metal Stent Deployment for Benign Pancreatic Duct Stricture (with Videos). J Gastrointest Surg 22, 2194–2200 (2018). https://doi.org/10.1007/s11605-018-3901-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11605-018-3901-z