Abstract
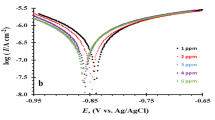
The electrochemical behavior of Zn in 0.5 M NaOH solutions containing various concentrations (0.01–0.1 M) of ClO3 − or ClO4 − anions was studied with potentiodynamic anodic polarization and chronoamperometry techniques. Microstructural and topographical characterization of the pitted surfaces was carried out by ex situ scanning electron microscopy and atomic force microscopy examinations. Addition of either ClO3 − or ClO4 − stimulated general corrosion and ruptured the passive layer (stable pitting), with ClO3 − being more aggressive than ClO4 −. Metastable pitting events appear as current oscillations (spikes) at potentials close to the pitting potential when Cl− ions are produced by cathodic reduction of ClO3 − and ClO4 − before passive layer growth. Current–time measurements are performed at fixed potential after production of Cl− ions and show that the rate of metastable pitting and the intensity of current spikes increase with the potential and the concentration of aggressive anions. Concepts of thin film growth are applied to the passive layer formation in order to explain those results. Metastable events are related to the presence of defects in the passive layer because their frequency and intensity are enhanced in conditions that favor defect formation and roughening in growing films, while stable pitting typically occurs at regions of high metal disorder.








Similar content being viewed by others
References
Pistorius PC, Burstein GT (1994) Corros Sci 36:525
Hashimoto M, Miyajima S, Murata T (1992) Corros Sci 33:885
Gonzalez-Garcia Y, Burstein GT, Gonzalez S, Souto RM (2004) Electrochem Commun 6:637
Trueman AR (2005) Corros Sci 47:2240
Amin MA, Hassan HH, Hazzazi OA, Qhatani MM (2008) J Appl Electrochem 38:1589
Amin MA (2009) Electrochim Acta 54:1857
Gupta RK, Sukiman NL, Cavanaugh MK, Hinton BRW, Hutchinson CR, Birbilis N (2012) Electrochim Acta 66:245
Jiang Z, Norby T, Middleton H (2010) Corros Sci 52:3158
Speckert L, Burstein GT (2011) Corros Sci 53:534
Cavanaugh MK, Birbilis N, Buchheit RG (2012) Electrochim Acta 59:336
Williams DE, Westcott C, Fleischmann M (1985) J Electrochem Soc 132:1796
Williams DE, Stewart J, Balkwill PH (1994) Corros Sci 36:1213
Contreras G, Goidanich S, Maggi S, Piccardi C, Diamanti MV, Pedeferri MP, Lazzari L (2011) Corros Rev 29:241
Souto RM, González-García Y, Battistel D, Daniele S (2012) Chem Eur J 18:230
Abd El Rehim SS, El-Sherbini EEF, Amin MA (2003) J Electroanal Chem 560:175–182
Amin MA (2005) Electrochim Acta 50:1265–1274
Amin MA, Hassan HH, Abd El Rehim SS (2008) Electrochim Acta 53:2600–2609
El-Rehim SSA, Hamed E, Shaltot AM, Amin MA (2012) Z Phys Chem 226:59
Shang X-L, Zhang B, Han E-H, Ke W (2012) Electrochim Acta 65:294
Zheng R, Janssens A, Carmeliet J, Bogaerts W, Hens H (2011) J Build Phys 34:277
Chen Y, Lobo RFM, Santos DMF, Sequeira CAC (2009) Quim Nova 32:387
El Aal EEA, El Wanees SA (2009) Corros Sci 51:1780
Amin MA, Abd El-Rehim SS (2012) Int J Electrochem Sci 7:7600
Macdonald DD (1992) J Electrochem Soc 139:3434
Macdonald DD (1999) Pure Appl Chem 71:951
Haruna T, Macdonald DD (1997) J Electrochem Soc 144:1574
Maurice V, Klein LH, Marcus P (2001) Electrochem Solid-State Lett 4:B1
Kunze J, Maurice V, Klein LH, Strehblow H-H, Marcus P (2001) J Phys Chem B 105:4263
Toney MF, Davenport AJ, Oblonsky LJ, Ryan MP, Vitus CM (1997) Phys Rev Lett 79:4282
Maurice V, Marcus P (2012) Electrochem Acta 84:129
Ohring M (1992) The materials science of thin films. Academic, London
Pimpinelli A, Villain J (1998) Physics of crystal growth. Cambridge University Press, Cambridge
Lide DR (1999) CRC handbook of chemistry and physics, 80th edn. CRC, Boca Raton, p 8
D’Alkaine CV, Boucherit MN (1997) J Electrochem Soc 144:3331
Pagitsas M, Pavlidou M, Sazou D (2008) Electrochim Acta 53:4784
Pagitsas M, Pavlidou M, Papadopoulou S, Sazou D (2007) Chem Phys Lett 434:63
Prinz H, Strehblow H-H (1998) Corros Sci 40:1671
Lee C, Batchelor B, Park SH, Han DS, Abdel-Wahab A, Kramer TA (2011) J Hazard Mater 197:183
Bohni H (1987) Langmiur 3:924
Frankel GS, Newman RG (1992) Critical factors in localized corrosion. The Electrochemical Society, Pennington
Aarao Reis FDA, Stafiej J, Badiali J-P (2006) J Phys Chem B 110:17554
Shibata T (1990) Corros Sci 31:413
Williams DE, Westcott C, Fleischmann M (1985) J Electrochem Soc 132:1804
Metikos-Hukovic M (1992) J Appl Electrochem 22:448
Kolics A, Polkinghorne JC, Wieckowski A (1998) Electrochim Acta 43:3605
Amin MA, Abd El-Rehim SS, El-Sherbini EEF, Mahmoud SR, Abbas MN (2009) Electrochim Acta 54:4288–4296
Galvele JR (1976) J Electrochem Soc 123:464
Budiansky ND, Hudson JL, Scully JR (2004) J Electrochem Soc 151:B233
Chame A, Aarão Reis FDA (2004) Surf Sci 553:145
O’Grady WE, Roeper DF, Natishan PM (2011) J Phys Chem C 115:25298
Conrad BR, Gomar-Nadal E, Cullen WG, Pimpinelli A, Einstein TL, Williams ED (2008) Phys Rev B 77:205328
Sathiyanarayanan R, Hamouda ABH, Pimpinelli A, Einstein TL (2011) Phys Rev B 83:35424
Aarao Reis FDA (2003) Phys Rev B 68:041602
Córdoba-Torres P, Bar-Eli K, Fairén V (2004) J Electroanal Chem 571:189
Evans JW, Thiel PA, Bartelt MC (2006) Surf Sci Rep 61:1
Córdoba-Torres P (2007) Phys Rev B 75:115405
Córdoba-Torres P, Nogueira RP (2008) Electrochim Acta 53:4805
Pistorius PC, Burstein GT (1992) Philos Trans R Soc Lond A 341:531
Burstein GT, Liu C, Souto RM, Vines SP (2005) Corros Eng Sci Technol 39:25
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Amin, M.A., Abd El-Rehim, S.S., Aarão Reis, F.D.A. et al. Metastable and stable pitting events at zinc passive layer in alkaline solutions. Ionics 20, 127–136 (2014). https://doi.org/10.1007/s11581-013-0953-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11581-013-0953-7