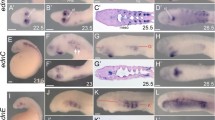
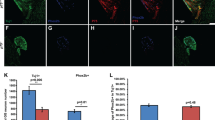
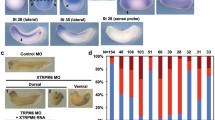
Abstract
P2X receptors are non-selective cation channels operated by extracellular ATP. Currently, little is known concerning the functions of these receptors during development. Previous work from our lab has shown that zebrafish have two paralogs of the mammalian P2X3 receptor subunit. One paralog, p2rx3.1, is expressed in subpopulations of neural and ectodermal cells in the embryonic head. To investigate the role of this subunit in early cranial development, we utilized morpholino oligonucleotides to disrupt its translation. Loss of this subunit resulted in craniofacial defects that included malformation of the pharyngeal skeleton. During formation of these structures, there was a marked increase in cell death within the branchial arches. In addition, the epibranchial (facial, glossopharyngeal, and vagal) cranial sensory ganglia and their circuits were perturbed. These data suggest that p2rx3.1 function in ectodermal cells is involved in purinergic signaling essential for proper craniofacial development and sensory circuit formation in the embryonic and larval zebrafish.







Similar content being viewed by others
References
Burnstock G, Knight GE (2004) Cellular distribution and functions of P2 receptor subtypes in different systems. Int Rev Cytol 240:31–304. doi:10.1016/S0074-7696(04)40002-3
Burnstock G (1996) P2 purinoceptors: historical perspective and classification. Ciba Found Symp 198:1–28 discussion 29-34
Masse K, Bhamra S, Eason R, Dale N, Jones EA (2007) Purine-mediated signalling triggers eye development. Nature 449:1058–1062. doi:10.1038/nature06189
Pearson RA, Dale N, Llaudet E, Mobbs P (2005) ATP released via gap junction hemichannels from the pigment epithelium regulates neural retinal progenitor proliferation. Neuron 46:731–744. doi:10.1016/j.neuron.2005.04.024
Weissman TA, Riquelme PA, Ivic L, Flint AC, Kriegstein AR (2004) Calcium waves propagate through radial glial cells and modulate proliferation in the developing neocortex. Neuron 43:647–661. doi:10.1016/j.neuron.2004.08.015
Burnstock G (2007) Purine and pyrimidine receptors. Cell Mol Life Sci 64:1471–1483. doi:10.1007/s00018-007-6497-0
Burnstock G (2008) Unresolved issues and controversies in purinergic signalling. J Physiol 586:3307–3312. doi:10.1113/jphysiol.2008.155903
Dale N (2008) Dynamic ATP signalling and neural development. J Physiol 586:2429–2436. doi:10.1113/jphysiol.2008.152207
Zimmermann H (2006) Nucleotide signaling in nervous system development. Pflugers Arch 452:573–588. doi:10.1007/s00424-006-0067-4
Egan TM, Samways DS, Li Z (2006) Biophysics of P2X receptors. Pflugers Arch 452:501–512. doi:10.1007/s00424-006-0078-1
North RA (2002) Molecular physiology of P2X receptors. Physiol Rev 82:1013–1067
Egan TM, Khakh BS (2004) Contribution of calcium ions to P2X channel responses. J Neurosci 24:3413–3420. doi:10.1523/JNEUROSCI.5429-03.2004
Hall BK, Miyake T (2000) Craniofacial development of avian and rodent embryos. Methods Mol Biol 135:127–137
Knight RD, Schilling TF (2006) Cranial neural crest and development of the head skeleton. Adv Exp Med Biol 589:120–133. doi:10.1007/978-0-387-46954-6_7
Kuratani S (2005) Craniofacial development and the evolution of the vertebrates: the old problems on a new background. Zoolog Sci 22:1–19. doi:10.2108/zsj.22.1
Schilling TF (1997) Genetic analysis of craniofacial development in the vertebrate embryo. Bioessays 19:459–468. doi:10.1002/bies.950190605
Bronner-Fraser M (1994) Neural crest cell formation and migration in the developing embryo. FASEB J 8:699–706
Holder N, Klein R (1999) Eph receptors and ephrins: effectors of morphogenesis. Development 126:2033–2044
Trainor PA, Krumlauf R (2000) Patterning the cranial neural crest: hindbrain segmentation and Hox gene plasticity. Nat Rev Neurosci 1:116–124. doi:10.1038/35039056
Kucenas S, Li Z, Cox JA, Egan TM, Voigt MM (2003) Molecular characterization of the zebrafish P2X receptor subunit gene family. Neuroscience 121:935–945. doi:10.1016/S0306-4522(03)00566-9
Kucenas S, Soto F, Cox JA, Voigt MM (2006) Selective labeling of central and peripheral sensory neurons in the developing zebrafish using P2X(3) receptor subunit transgenes. Neuroscience 138:641–652. doi:10.1016/j.neuroscience.2005.11.058
Lawson ND, Weinstein BM (2002) In vivo imaging of embryonic vascular development using transgenic zebrafish. Dev Biol 248:307–318. doi:10.1006/dbio.2002.0711
Westerfield M (2000) The zebrafish book. University of Oregon, Eugene
Kimmel CB, Ballard WW, Kimmel SR, Ullmann B, Schilling TF (1995) Stages of embryonic development of the zebrafish. Dev Dyn 203:253–310
Lewis C, Neidhart S, Holy C, North RA, Buell G, Surprenant A (1995) Coexpression of P2X2 and P2X3 receptors subunits can account for ATP-gated currents in sensory neurons. Nature 377:432–435. doi:10.1038/377432a0
Chomczynski P, Sacchi N (1987) Single-step method of RNA isolation by acid guanidinium thiocyanate-phenol-chloroform extraction. Anal Biochem 162:156–159. doi:10.1016/0003-2697(87)90021-2
Dutton KA et al (2001) Zebrafish colourless encodes sox10 and specifies non-ectomesenchymal neural crest fates. Development 128:4113–4125
Pohl BS, Knochel W (2001) Overexpression of the transcriptional repressor FoxD3 prevents neural crest formation in Xenopus embryos. Mech Dev 103:93–106. doi:10.1016/S0925-4773(01)00334-3
Knight RD et al (2003) lockjaw encodes a zebrafish tfap2a required for early neural crest development. Development 130:5755–5768. doi:10.1242/dev.00575
Baker CV, Bronner-Fraser M (2000) Establishing neuronal identity in vertebrate neurogenic placodes. Development 127:3045–3056
Knight RD, Javidan Y, Nelson S, Zhang T, Schilling T (2004) Skeletal and pigment cell defects in the lockjaw mutant reveal multiple roles for zebrafish tfap2a in neural crest development. Dev Dyn 229:87–98. doi:10.1002/dvdy.10494
Knight RD, Javidan Y, Zhang T, Nelson S, Schilling TF (2005) AP2-dependent signals from the ectoderm regulate craniofacial development in the zebrafish embryo. Development 132:3127–3138. doi:10.1242/dev.01879
Li W, Cornell RA (2007) Redundant activities of Tfap2a and Tfap2c are required for neural crest induction and development of other non-neural ectoderm derivatives in zebrafish embryos. Dev Biol 304:338–354. doi:10.1016/j.ydbio.2006.12.042
Ekker SC (2000) Morphants: a new systematic vertebrate functional genomics approach. Yeast 17:302–306. doi:10.1002/1097-0061(200012)17:4<302::AID-YEA53>3.0.CO;2-#
Egan TM, Haines WR, Voigt MM (1998) A domain contributing to the ion channel of ATP-gated P2X2 receptors identified by the substituted cysteine accessibility method. J Neurosci 18:2350–2359
Torres GE, Egan TM, Voigt MM (1999) Identification of a domain involved in ATP-gated ionotropic receptor subunit assembly. J Biol Chem 274:22359–22365. doi:10.1074/jbc.274.32.22359
Boue-Grabot E, Akimenko MA, Seguela P (2000) Unique functional properties of a sensory neuronal P2X ATP-gated channel from zebrafish. J Neurochem 75:1600–1607. doi:10.1046/j.1471-4159.2000.0751600.x
Egan TM, Cox JA, Voigt MM (2000) Molecular cloning and functional characterization of the zebrafish ATP-gated ionotropic receptor P2X(3) subunit. FEBS Lett 475:287–290. doi:10.1016/S0014-5793(00)01685-9
Kimmel CB, Miller CT, Kruze G, Ullmann B, BreMiller RA, Larison KD, Snyder HC (1998) The shaping of pharyngeal cartilages during early development of the zebrafish. Dev Biol 203:245–263. doi:10.1006/dbio.1998.9016
Miller CT, Schilling TF, Lee K, Parker J, Kimmel CB (2000) sucker encodes a zebrafish endothelin-1 required for ventral pharyngeal arch development. Development 127:3815–3828
Richman JM, Lee SH (2003) About face: signals and genes controlling jaw patterning and identity in vertebrates. Bioessays 25:554–568. doi:10.1002/bies.10288
Hall BK, Miyake T (2000) All for one and one for all: condensations and the initiation of skeletal development. Bioessays 22:138–147. doi:10.1002/(SICI)1521-1878(200002)22:2<138::AID-BIES5>3.0.CO;2-4
Furutani-Seiki M et al (1996) Neural degeneration mutants in the zebrafish, Danio rerio. Development 123:229–239
Barrallo-Gimeno A, Holzschuh J, Driever W, Knapik EW (2004) Neural crest survival and differentiation in zebrafish depends on mont blanc/tfap2a gene function. Development 131:1463–1477. doi:10.1242/dev.01033
Robu ME, Larson JD, Nasevicius A, Beiraghi S, Brenner C, Farber SA, Ekker SC (2007) p53 activation by knockdown technologies. PLoS Genet 3:e78. doi:10.1371/journal.pgen.0030078
Holzschuh J et al (2005) Requirements for endoderm and BMP signaling in sensory neurogenesis in zebrafish. Development 132:3731–3742. doi:10.1242/dev.01936
Nechiporuk A, Linbo T, Raible DW (2005) Endoderm-derived Fgf3 is necessary and sufficient for inducing neurogenesis in the epibranchial placodes in zebrafish. Development 132:3717–3730. doi:10.1242/dev.01876
Baker CV, Bronner-Fraser M (2001) Vertebrate cranial placodes I. Embryonic induction. Dev Biol 232:1–61. doi:10.1006/dbio.2001.0156
Kim CH, Ueshima E, Muraoka O, Tanaka H, Yeo SY, Huh TL, Miki N (1996) Zebrafish elav/HuC homologue as a very early neuronal marker. Neurosci Lett 216:109–112. doi:10.1016/0304-3940(96)13021-4
Lee SA, Shen EL, Fiser A, Sali A, Guo S (2003) The zebrafish forkhead transcription factor Foxi1 specifies epibranchial placode-derived sensory neurons. Development 130:2669–2679. doi:10.1242/dev.00502
Kimmel CB, Miller CT, Moens CB (2001) Specification and morphogenesis of the zebrafish larval head skeleton. Dev Biol 233:239–257. doi:10.1006/dbio.2001.0201
Yelick PC, Schilling TF (2002) Molecular dissection of craniofacial development using zebrafish. Crit Rev Oral Biol Med 13:308–322. doi:10.1177/154411130201300402
Schlosser G (2006) Induction and specification of cranial placodes. Dev Biol 294:303–351. doi:10.1016/j.ydbio.2006.03.009
Moreau M, Leclerc C (2004) The choice between epidermal and neural fate: a matter of calcium. Int J Dev Biol 48:75–84. doi:10.1387/ijdb.15272372
Webb SE, Moreau M, Leclerc C, Miller AL (2005) Calcium transients and neural induction in vertebrates. Cell Calcium 37:375–385. doi:10.1016/j.ceca.2005.01.005
D'Amico-Martel A (1982) Temporal patterns of neurogenesis in avian cranial sensory and autonomic ganglia. Am J Anat 163:351–372. doi:10.1002/aja.1001630407
Northcutt RG, Brandle K (1995) Development of branchiomeric and lateral line nerves in the axolotl. J Comp Neurol 355:427–454. doi:10.1002/cne.903550309
Zou D, Silvius D, Fritzsch B, Xu PX (2004) Eya1 and Six1 are essential for early steps of sensory neurogenesis in mammalian cranial placodes. Development 131:5561–5572. doi:10.1242/dev.01437
Cockayne DA et al (2005) P2X2 knockout mice and P2X2/P2X3 double knockout mice reveal a role for the P2X2 receptor subunit in mediating multiple sensory effects of ATP. J Physiol 567:621–369
Boldogkoi Z, Schutz B, Sallach J, Zimmer A (2002) P2X(3) receptor expression at early stage of mouse embryogenesis. Mech Dev 118:255–260. doi:10.1016/S0925-4773(02)00280-0
Cheung KK, Burnstock G (2002) Localization of P2X(3) receptors and coexpression with P2X(2) receptors during rat embryonic neurogenesis. J Comp Neurol 443:368–382. doi:10.1002/cne.10123
Khakh BS et al (2001) International union of pharmacology. XXIV. Current status of the nomenclature and properties of P2X receptors and their subunits. Pharmacol Rev 53:107–118
Sun SK, Dee CT, Tripathi VB, Rengifo A, Hirst CS, Scotting PJ (2007) Epibranchial and otic placodes are induced by a common Fgf signal, but their subsequent development is independent. Dev Biol 303:675–686. doi:10.1016/j.ydbio.2006.12.008
Acknowledgments
We would like to thank Jasmina Mandzukic for help with fish husbandry, and Dr. Steve Johnson (Washington Univ. St. Louis) for his great insight and help on all things zebrafish. The zn8 antibody developed by Dr. W. Trevarrow was obtained from the Developmental Studies Hybridoma Bank developed under the auspices of the NICHD and maintained by The University of Iowa, Department of Biological Sciences, Iowa City, IA 52242. This work was supported by the NIH grants NS50261 (MMV), NS051140 (SK), and GM008306 (SK and AL).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kucenas, S., Cox, J.A., Soto, F. et al. Ectodermal P2X receptor function plays a pivotal role in craniofacial development of the zebrafish. Purinergic Signalling 5, 395–407 (2009). https://doi.org/10.1007/s11302-009-9165-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11302-009-9165-z