Abstract
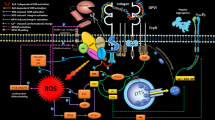
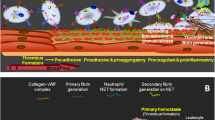
Platelet receptor GPVI plays an important role in platelet firm adhesion to site of vascular injury. Receptor ligation with collagen, in company with other agonist/receptor interactions, augments inside out signaling pathways leading to platelet aggregation and thrombus formation. As GPVI expression is significantly modulated by ectodomain shedding, this study aimed to examine whether GPVI shedding functionally affects collagen-mediated platelet activation during storage. 6 PRP-platelet concentrates were subjected to adhesion analysis on collagen matrix under mild stirring condition as well as collagen-induced aggregation on day 1, 3 and 5 post-storage. Concurrently, platelet supernatants of same samples were fractionated by ultra-centrifugation and obtained micro-particle-free samples were subjected to western blot analysis for the evaluation of GPVI shedding. We showed a direct correlation between collagen-dependent platelet aggregation and adhesion (r = 0.8, p = 0.0001). The increasing levels of GPVI shedding during storage were in reverse correlation with collagen-induced platelet aggregation (r = − 0.82, p = 0.0004) which was significantly reducing during storage. Platelet adhesion to collagen matrix significantly decreased post-storage while it was also reversely correlated with the levels of GPVI shedding during 5 days storage of platelets (r = − 0.69, p = 0.002). Data presented here demonstrated that progressive shedding of surface adhesion receptor GPVI can affect its functional activities in stored platelets. Thereby considering the crucial role of GPVI in platelet adhesion to the site of injury, whether the therapeutic efficacy of banked platelet products could be influenced by storage-dependent shedding of this receptor, remains to be answered in future studies.



Similar content being viewed by others
References
Brass LF, Jiang H, Wu J, Stalker TJ, Zhu L (2006) Contact-dependent signaling events that promote thrombus formation. Blood Cells Mol Dis 36:157–161
Gardiner EE, Arthur JF, Berndt MC, Andrews RK (2005) Role of calmodulin in platelet receptor function. Curr Med Chem 3:283–287
Berger G, Hartwell DW, Wagner DD (1998) P-selectin and platelet clearance. Blood 92:4446–4452
Contin C, Pitard V, Itai T, Nagata S, Moreau JF, Dechanet-Merville J (2003) Membrane-anchored CD40 is processed by the tumor necrosis factor-alpha-converting enzyme. Implications for CD40 signaling. J Biol Chem 278:32801–32809
Bergmeier W, Rabie T, Strehl A, Piffath CL, Prostredna M, Wagner DD, Nieswandt B (2004) GPVI down-regulation in murine platelets through metalloproteinase-dependent shedding. Thromb Haemost 91:951–958
Bergmeier W, Piffath CL, Cheng G, Dole VS, Zhang Y, von Andrian UH, Wagner DD (2004) Tumor necrosis factor-alpha-converting enzyme (ADAM17) mediates GPIbalpha shedding from platelets in vitro and in vivo. Circ Res 95:677–683
Rabie T, Strehl A, Ludwig A, Nieswandt B (2005) Evidence for a role of ADAM17 (TACE) in the regulation of platelet glycoprotein V. J Biol Chem 280:14462–14468
Aktas B, Pozgajova M, Bergmeier W, Sunnarborg S, Offermanns S, Lee D, Wagner DD, Nieswandt B (2005) Aspirin induces platelet receptor shedding via ADAM17 (TACE). J Biol Chem 280:39716–39722
Andrews RK, Karunakaran D, Gardiner EE, Berndt MC (2007) Platelet receptor proteolysis: a mechanism for downregulating platelet reactivity. Arterioscler Thromb Vasc Biol 27:1511–1520
Hosseini E, Ghasemzadeh M, Nassaji F, Jamaat ZP (2016) GPVI modulation during platelet activation and storage: its expression levels and ectodomain shedding compared to markers of platelet storage lesion. Platelets 28:498–508
Ghasemzadeh M, Hosseini E (2017) Platelet granule release is associated with reactive oxygen species generation during platelet storage: a direct link between platelet pro-inflammatory and oxidation states. Thromb Res 156:101–104
Wood B, Padula MP, Marks DC, Johnson L (2016) Refrigerated storage of platelets initiates changes in platelet surface marker expression and localization of intracellular proteins. Transfusion 56:2548–2559
Hosseini E, Ghasemzadeh M (2012) Different stages of platelet adhesion to the site of vascular injury. Iran J Blood Cancer 4:133–142
Ghasemzadeh M (2009) Investigation of signaling cross-talk between platelets and neutrophils. Dissertation, Monash University
Kulkarni S, Woollard KJ, Thomas S, Oxley D, Jackson SP (2007) Conversion of platelets from a proaggregatory to a proinflammatory adhesive phenotype: role of PAF in spatially regulating neutrophil adhesion and spreading. Blood 110:1879–1886
Fu Q, Garnham CP, Elliott ST, Bovenkamp DE, Van Eyk JE (2005) A robust, streamlined, and reproducible method for proteomic analysis of serum by delipidation, albumin and IgG depletion, and two-dimensional gel electrophoresis. Proteomics 5:2656–2664
Ghasemzadeh M, Kaplan ZS, Alwis I, Schoenwaelder SM, Ashworth KJ, Westein E, Hosseini E, Salem HH, Slattery R, McColl SR, Hickey MJ, Ruggeri ZM, Yuan Y, Jackson SP (2013) The CXCR1/2 ligand NAP-2 promotes directed intravascular leukocyte migration through platelet thrombi. Blood 121:4555–4566
Apelseth TO, Hervig T (2007) In vitro evaluation of platelet concentrates during storage: platelet counts and markers of platelet destruction. Transfus Apheresis Sci 37:261–268
Cardigan R, Turner C, Harrison P (2005) Current methods of assessing platelet function: relevance to transfusion medicine. Vox Sang 88:153–163
Gardiner EE, Andrews RK (2014) Platelet receptor expression and shedding: glycoprotein Ib-IX-V and glycoprotein VI. Transfus Med Rev 28:56–60
Gardiner EE, Arthur JF, Kahn ML, Berndt MC, Andrews RK (2004) Regulation of platelet membrane levels of glycoprotein VI by a platelet-derived metalloproteinase. Blood 104:3611–3617
Schoenwaelder SM, Jarman KE, Gardiner EE, Hua M, Qiao J, White MJ, Josefsson EC, Alwis I, Ono A, Willcox A, Andrews RK, Mason KD, Salem HH, Huang DC, Kile BT (2011) Roberts AW and Jackson SP: Bcl-xL-inhibitory BH3 mimetics can induce a transient thrombocytopathy that undermines the hemostatic function of platelets. Blood 118:1663–1674
Siljander PR, Munnix IC, Smethurst PA, Deckmyn H, Lindhout T, Ouwehand WH, Farndale RW, Heemskerk JW (2004) Platelet receptor interplay regulates collagen-induced thrombus formation in flowing human blood. Blood 103:1333–1341
Baker J, Griggs RK, Falati S, Poole AW (2004) GPIb potentiates GPVI-induced responses in human platelets. Platelets 15:207–214
Arthur JF, Gardiner EE, Matzaris M, Taylor SG, Wijeyewickrema L, Ozaki Y, Kahn ML, Andrews RK, Berndt MC (2005) Glycoprotein VI is associated with GPIb-IX-V on the membrane of resting and activated platelets. Thromb Haemost 93:716–723
Nieswandt B, Bergmeier W, Eckly A, Schulte V, Ohlmann P, Cazenave J-P, Zirngibl H, Offermanns S, Gachet C (2001) Evidence for cross-talk between glycoprotein VI and Gi-coupled receptors during collagen-induced platelet aggregation. Blood 97:3829–3835
Nieswandt B, Schulte V, Zywietz A, Gratacap M-P, Offermanns S (2002) Costimulation of Gi-and G12/G13-mediated signaling pathways induces integrin αIIbβ3 activation in platelets. J Biol Chem 277:39493–39498
Boomgaard MN, Gouwerok CW, Homburg CH, de Groot G, MJ, IJ, de Korte D (1994) The platelet adhesion capacity to subendothelial matrix and collagen in a flow model during storage of platelet concentrates for 7 days. Thromb Haemost 72:611–616
Tynngard N, Wallstedt M, Sodergren AL, Faxalv L, Ramstrom S (2015) Platelet adhesion changes during storage studied with a novel method using flow cytometry and protein-coated beads. Platelets 26:177–185
Author information
Authors and Affiliations
Contributions
MG supervised and designed the study, did the experiments, analyzed the data and wrote the paper. EH did some experiments, helped with analyzing data and writing the paper. PB did some experiments.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest. This work was part of Dr. Ghasemzadeh’s approved project (No. 1394-01-33-1861) which is also considered as research material for a PhD student thesis supported by Iranian blood transfusion organization and High Institute for Research and Education in Transfusion Medicine in Iran.
Ethical approval
Ethical approval for this research was provided by ethic committee in High Institute for Research and Education in Transfusion Medicine in accordance with national legislation and local rules.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Hosseini, E., Beshkar, P. & Ghasemzadeh, M. Reverse correlations of collagen-dependent platelet aggregation and adhesion with GPVI shedding during storage. J Thromb Thrombolysis 46, 534–540 (2018). https://doi.org/10.1007/s11239-018-1739-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11239-018-1739-6