Abstract
Background
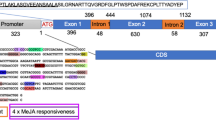
Rhizobial surface polysaccharides are important molecular determinants required for successful symbiosis with legumes. In Sinorhizobium (Ensifer) meliloti Rm41, the rkp-2 region is involved in the biosynthesis of K-antigen polysaccharide (KPS) and lipopolysaccharide (LPS). This region is composed of two genes, lpsL and rkpK, which are respectively responsible for the production of galacturonic and glucuronic acid.
Results
In this work, we show that in S. (Ensifer) fredii HH103 these genes do not form a transcriptional unit and that the transcriptional rate of rkpK is much higher than that of lpsL. Inactivation of each of these genes resulted in alterations in LPS, but did not affect KPS production, which is in agreement with the lack of uronic acids in S. fredii HH103 KPS. Mutation of rkpK also impaired HH103 exopolysaccharide (EPS) production, most probably due to the presence of glucuronic acid in HH103 EPS, as well as increased bacterial autoaggregation and osmosensitivity and decreased biofilm formation on plastic surfaces. Inactivation of rkpK affected negatively symbiosis with cowpea but not with soybean. Mutation of lpsL led to a complete symbiotic impairment with cowpea, whereas soybean plants inoculated with this mutant only formed pseudonodules. In both plants, the lpsL mutant showed defects in root infection.
Conclusion
These results confirm the symbiotic importance of HH103 LPS in symbiosis with legumes.







Similar content being viewed by others
Abbreviations
- ARA:
-
Acetylene reduction assay
- CF:
-
Calcofluor
- CG:
-
Cyclic glucans
- EPS:
-
Exopolysaccharide
- GalA:
-
Galacturonic acid
- GlcA:
-
Glucuronic acid
- KPS:
-
K-antigen polysaccharide
- LPS:
-
Lipopolysaccharide
References
Acosta-Jurado S, Alias-Villegas C, Navarro-Gómez P, Zehner S, Murdoch PD, Rodríguez-Carvajal MA, Soto MJ, Ollero FJ, Ruiz-Sainz JE, Göttfert M, Vinardell JM (2016a) The Sinorhizobium fredii HH103 MucR1 global regulator is connected with the nod regulon and is required for efficient symbiosis with Lotus burttii and Glycine max cv. Williams. Mol Plant-Microbe Interact 29:700–712
Acosta-Jurado S, Navarro-Gómez P, Murdoch P d S, Crespo-Rivas JC, Jie S, Cuesta-Berrio L, Ruiz-Sainz JE, Rodríguez-Carvajal MÁ, Vinardell JM (2016b) Exopolysaccharide production by Sinorhizobium fredii HH103 is repressed by genistein in a NodD1-dependent manner. PLoS One 11:e0160499
Acosta-Jurado S, Rodríguez-Navarro DN, Kawaharada Y, Perea JF, Gil-Serrano A, Jin H, An Q, Rodríguez-Carvajal MA, Andersen SU, Sandal N, Stougaard J, Vinardell JM, Ruiz-Sainz JE (2016c) Sinorhizobium fredii HH103 invades Lotus burttii by crack entry in a Nod factor-and surface polysaccharide-dependent manner. Mol Plant-Microbe Interact 29:925–937
Becker A, Schmidt M, Jäger W, Pühler A (1995) New gentamicin-resistance and lacZ promoter-probe cassettes suitable for insertion mutagenesis and generation of transcripcional fusions. Gene 162:37–39
Beringer JE (1974) R factor transfer in Rhizobium leguminosarum. J Gen Microbiol 84:188–198
Buendía-Clavería AM, Moussaid A, Ollero FJ, Vinardell JM, Torres A, Moreno J, Gil-Serrano AM, Rodríguez-Carvajal MA, Tejero-Mateo P, Peart JL, Brewin NJ, Ruiz-Sainz JE (2003) A purL mutant of Sinorhizobium fredii HH103 is symbiotically defective and altered in its lipopolysaccharide. Microbiology 149:1807–1818
Crespo-Rivas JC, Margaret I, Pérez-Montaño F, López-Baena FJ, Vinardell JM, Ollero FJ, Moreno J, Ruiz-Sainz JE, Buendía-Clavería AM (2007) A pyrF auxothrophic mutant of Sinorhizobium fredii HH103 impaired in its symbiotic interactions with soybean and other legumes. Int Microbiol 10:169–176
De Castro C, Molinaro A, Lanzetta R, Silipo A, Parrilli M (2008) Lipopolysaccharide structures from Agrobacterium and Rhizobium species. Carbohydr Res 343:1924–1933
Downie JA (2010) The roles of extracellular proteins, polysaccharides and signals in the interactions of rhizobia with legume roots. FEMS Microbiol Rev 34:150–170
Finan TM, Hirsch AM, Leigh JA, Johansen E, Kuldau GA, Deegan S, Walker GC, Signer ER (1985) Symbiotic mutants of Rhizobium meliloti that uncouple plant from bacterial differentiation. Cell 40:869–877
Gil-Serrano AM, Rodríguez-Carvajal MA, Tejero Mateo P, Espartero JL, Menéndez M, Corzo J, Ruiz-Sainz JE, Buendía-Clavería AM (1999) Structural determination of a 5-acetamido-3,5,7,9-tetradeoxy-7-(3-hydroxybutyramido)-l-glycero-l-manno-nonulosonic acid-containing homopolysaccharide isolated from Sinorhizobium fredii HH103. Biochem J 342:527–535
Hidalgo A, Margaret I, Crespo-Rivas JC, Parada M, Murdoch PS, López A, Buendía-Clavería AM, Moreno J, Albareda M, Gil-Serrano AM, Rodríguez-Carvajal MA, Palacios JM, Ruiz-Sainz JE, Vinardell JM (2010) The rkpU gene of Sinorhizobium fredii HH103 is required for bacterial K-antigen polysaccharide production and for efficient nodulation with soybean but not with cowpea. Microbiology 156:3398–3411
Janczarek M, Rachwał K, Marzec A, Grządziel J, Palusińska-Szysz M (2015) Signal molecules and cell-surface components involved in early stages of the legume–rhizobium interactions. Appl Soil Ecol 85:94–113
Jiménez-Guerrero I, Pérez-Montaño F, Monreal JA, Preston GM, Fones H, Vioque B, Ollero FJ, Lopez-Baena FJ (2015) The Sinorhizobium (Ensifer) fredii HH103 type 3 secretion system suppresses early defense responses to effectively nodulate soybean. Mol Plant-Microbe Interact 28:790–799
Jones KM, Kobayashi H, Davies BW, Taga ME, Walker GC (2007) How rhizobial symbionts invade plants: the Sinorhizobium-Medicago model. Nature Rev Microbiol 5:619–633
Kawaharada Y, Kelly S, Nielsen MW, Hjuler CT, Gysel K, Muszyński A, Carlson RW, Thygesen MB, Sandal N, Asmussen MH, Vinther M, Andersen SU, Krusell L, Thirup S, Jensen KJ, Ronson CW, Blaise M, Radutoiu S, Stougaard J (2015) Receptor-mediated exopolysaccharide perception controls bacterial infection. Nature 523:308–312
Kawaharada Y, Nielsen MW, Kelly S, James EK, Andersen KR, Rasmussen SR, Füchtbauer W, Madsen LH, Heckmann AB, Radutoiu S, Stougaard J (2017) Differential regulation of the Epr3 receptor coordinates membrane-restricted rhizobial colonization of root nodule primordia. Nat Commun 8:14534
Keating DH, Willits MG, Long SR (2002) A Sinorhizobium meliloti lipopolysaccharide mutant altered in cell surface sulfation. J Bacteriol 184:6681–6689
Kelly SJ, Muszyński A, Kawaharada Y, Hubber AM, Sullivan JT, Sandal N, Carlson RW, Stougaard J, Ronson CW (2013) Conditional requirement for exopolysaccharide in the Mesorhizobium-Lotus symbiosis. Mol Plant-Microbe Interact 26:319–329
Kereszt A, Kiss E, Reuhs BL, Carlson RW, Kondorosi A, Putnoky P (1998) Novel rkp gene clusters of Sinorhizobium meliloti involved in capsular polysaccharide production and invasion of the symbiotic nodule: the rkpK gene codes a UDP-glucose dehydrogenase. J Bacteriol 180:5426–5431
Kiss E, Kereszt A, Barta F, Stephens S, Reuhs BL, Kondorosi A, Putnoky P (2001) The rkp-3 gene region of Sinorhizobium meliloti Rm41 contains strain-specific genes that determine K antigen structure. Mol Plant-Microbe Interact 14:1395–1403
Kondorosi E, Mergaert P, Kereszt A (2013) A paradigm for endosymbiotic life: cell differentiation of Rhizobium bacteria provoked by host plant factors. Annu Rev Microbiol 67:611–628
Leigh A, Signer ER, Walker GC (1985) Exopolysaccharide-deficient mutants of Rhizobium meliloti that form ineffective nodules. Proc Natl Acad Sci U S A 82:6231–6235
López-Baena FJ, Monreal JA, Pérez-Montaño F, Guasch-Vidal B, Bellogín RA, Vinardell JM, Ollero FJ (2009) The absence of Nops secretion in Sinorhizobium fredii HH103 increases GmPR1 expression in Williams soybean. Mol Plant-Microbe Interact 22:1445–1454
López-Baena FJ, Ruiz-Sainz JE, Rodríguez-Carvajal MA, Vinardell JM (2016) Bacterial molecular signals in the Sinorhizobium fredii-soybean symbiosis. Int J Mol Sci 17:E755
Margaret I, Becker A, Blom J, Bonilla I, Goesmann A, Göttfert M, Lloret J, Mittard-Runte V, Rückert C, Ruiz-Sainz JE, Vinardell JM, Weidner S (2011) Symbiotic properties and first analyses of the genomic sequence of the fast growing model strain Sinorhizobium fredii HH103 nodulating soybean. J Biotechnol 155:11–19
Margaret I, Crespo-Rivas JC, Acosta-Jurado S, Buendía-Clavería AM, Cubo MT, Gil-Serrano A, Moreno J, Murdoch PS, Rodríguez-Carvajal MA, Rodríguez-Navarro DN, Ruiz-Sainz JE, Sanjuán J, Soto MJ, Vinardell JM (2012) Sinorhizobium fredii HH103 rkp-3 genes are required for K-antigen polysaccharide biosynthesis, affect lipopolysaccharide structure and are essential for infection of legumes forming determinate nodules. Mol Plant-Microbe Interact 25:825–838
Margaret I, Lucas MM, Acosta-Jurado S, Buendía-Clavería AM, Fedorova E, Hidalgo A, Rodríguez-Carvajal MA, Rodriguez-Navarro DN, Ruiz-Sainz JE, Vinardell JM (2013) The Sinorhizobium fredii HH103 lipopolysaccharide is not only relevant at early soybean nodulation stages but also for symbiosome stability in mature nodules. PLoS One 8:e74717
Margaret-Oliver I, Lei W, Parada M, Rodríguez-Carvajal MA, Crespo-Rivas JC, Hidalgo A, Gil-Serrano A, Moreno J, Rodríguez-Navarro DN, Buendía-Clavería A, Ollero J, Ruiz-Sainz JE, Vinardell JM (2012) Sinorhizobium fredii HH103 does not strictly require KPS and/or EPS to nodulate Glycyrrhiza uralensis, an indeterminate nodule-forming legume. Arch Microbiol 194:87–102
Meyer F, Goesmann A, McHardy AC, Bartels D, Bekel T, Clausen J, Kalinowski J, Linke B, Rupp O, Giegerich R, Pühler A (2003) GenDB--an open source genome annotation system for prokaryote genomes. Nucleic Acids Res 31:2187–2195
Mitra S, Mukherjee A, Wiley-Kalil A, Das S, Owen H, Reddy PM, Ané JM, James EK, Gyaneshwar P (2016) A rhamnose-deficient lipopolysaccharide mutant of Rhizobium sp. IRBG74 is defective in root colonization and beneficial interactions with its flooding-tolerant hosts Sesbania cannabina and wetland rice. J Exp Bot 67:5869–5884
O’Toole GA, Kolter R (1998) Initiation of biofilm formation in Pseudomonas fluorescens WCS365 proceeds via multiple, convergent signalling pathways: a genetic analysis. Mol Microbiol 28:449–461
Parada M, Vinardell JM, Ollero FJ, Hidalgo A, Gutiérrez R, Buendía-Clavería AM, Lei W, Margaret I, López-Baena FJ, Gil-Serrano AM, Rodríguez-Carvajal MA, Moreno J, Ruiz-Sainz JE (2006) Sinorhizobium fredii HH103 mutants affected in capsular polysaccharide (KPS) are impaired for nodulation with soybean and Cajanus cajan. Mol Plant-Microbe Interact 19:43–52
Pérez-Mendoza D, Rodríguez-Carvajal MÁ, Romero-Jiménez L, Farias GA, Lloret J, Gallegos MT, Sanjuán J (2015) Novel mixed-linkage β-glucan activated by c-di-GMP in Sinorhizobium meliloti. Proc Natl Acad Sci U S A 112:E757–E765
Pérez-Montaño F, Jiménez-Guerrero I, Acosta-Jurado S, Navarro-Gómez P, Ollero FJ, Ruiz-Sainz JE, López-Baena FJ, Vinardell JM (2016) A transcriptomic analysis of the effect of genistein on Sinorhizobium fredii HH103 reveals novel rhizobial genes putatively involved in symbiosis. Sci Rep 6:31592
Putnoky P, Petrovics G, Kereszt A, Grosskopf E, Ha DT, Banfalvi Z, Kondorosi A (1990) Rhizobium meliloti lipopolysaccharide and exopolysaccharide can have the same function in the plant-bacterium interaction. J Bacteriol 172:5450–5458
Reuhs BL, Geller DP, Kim JS, Fox JE, Kolli VSK, Pueppke SG (1998) Sinorhizobium fredii and Sinorhizobium meliloti produce structurally conserved lipopolysaccharides and strain-specific K antigens. Appl Environ Microbiol 64:4930–4938
Rodríguez-Navarro DN, Rodríguez-Carvajal MA, Acosta-Jurado S, Soto MJ, Margaret I, Crespo-Rivas JC, Sanjuan J, Temprano F, Gil-Serrano A, Ruiz-Sainz JE, Vinardell JM (2014) Structure and biological roles of Sinorhizobium fredii HH103 exopolysaccharide. PLoS One 9:e115391
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning. A laboratory manual, 2nd edn. Cold Spring Harbor Laboratory Press, Cold Spring Harbor
Simon R (1984) High frequency mobilization of gram-negative bacterial replicons by the in vivo constructed Tn5-Mob transposon. Mol Gen Genet 196:413–420
Solovyev V, Salamov A (2011) Automatic annotation of microbial genomes and metagenomic sequences. In: Li RW (ed) Metagenomics and its applications in agriculture, biomedicine and environmental studies. Nova Science Publishers, New York, pp 61–78
Sorroche FG, Spesia MB, Zorreguieta A, Giordano W (2012) A positive correlation between bacterial autoaggregation and biofilm formation in native Sinorhizobium meliloti isolates from Argentina. Appl Environ Microbiol 78:4092–4101
Thomas-Oates J, Bereszczak J, Edwards E, Gill A, Noreen S, Zhou JC, Chen MZ, Miao LH, Xie FL, Yang JK, Zhou Q, Yang SS, Li XH, Wang L, Spaink HP, Schlaman HR, Harteveld M, Díaz CL, van Brussel AA, Camacho M, Rodríguez-Navarro DN, Santamaría C, Temprano F, Acebes JM, Bellogín RA, Buendía-Clavería AM, Cubo MT, Espuny MR, Gil AM, Gutiérrez R, Hidalgo A, López-Baena FJ, Madinabeitia N, Medina C, Ollero FJ, Vinardell JM, Ruiz-Sainz JE (2003) A catalogue of molecular, physiological and symbiotic properties of soybean-nodulating rhizobial strains from different soybean cropping areas of China. Syst Appl Microbiol 26:453–465
Vinardell JM, López-Baena FJ, Hidalgo A, Ollero FJ, Bellogín R, Espuny MR, Temprano F, Romero F, Krishnan HB, Pueppke SG, Ruiz-Sainz JE (2004a) The effect of FITA mutations on the symbiotic properties of Sinorhizobium fredii varies in a chromosomal-background-dependent manner. Arch Microbiol 181:144–154
Vinardell JM, Ollero FJ, Hidalgo A, López-Baena FJ, Medina C, Ivanov-Vangelov K, Parada M, Madinabeitia N, Espuny MR, Bellogín RA, Camacho M, Rodríguez-Navarro DN, Soria-Díaz ME, Gil-Serrano AM, Ruiz-Sainz JE (2004b) NolR regulates diverse symbiotic signals of Sinorhizobium fredii HH103. Mol Plant-Microbe Interact 17:676–685
Vinardell JM, Acosta-Jurado S, Göttfert M, Zehner S, Becker A, Baena-Ropero I, Blom J, Crespo-Rivas JC, Goesmann A, Jaenicke S, Krol E, McIntosh M, Margaret I, Pérez-Montaño F, Schneiker-Bekel S, Serrania J, Szczepanowski R, Buendia-Claveria AM, Lloret J, Bonilla I, Pühler A, Ruiz-Sainz JE, Weidner S (2015) The Sinorhizobium fredii HH103 genome: a comparative analysis with S. fredii strains differing in their symbiotic behaviour with soybean. Mol. Plant-Microbe Interact 28:811–824
Vincent JM (1970) Appendix III. The modified Fåhraeus slide technique. In: A manual for the practical study of root nodule bacteria. Blackwell Scientific Publications, Oxford, pp 144–145
Weidner S, Becker A, Bonilla I, Jaenicke S, Lloret J, Margaret I, Pühler A, Ruiz-Sainz JE, Schneiker-Bekel S, Szczepanowski R, Vinardell JM, Zehner S, Göttfert M (2012) Genome sequence of the soybean symbiont Sinorhizobium fredii HH103. J Bacteriol 194:1617–1618
Westphal O, Jann K (1965) Bacterial lipopolysaccharides, extraction with phenol-water and further application of the procedure. Meth Carbohydr Chem 5:83–91
Zehner S, Schober G, Wenzel M, Lang K, Göttfert M (2008) Expression of the Bradyrhizobium japonicum type III secretion system in legume nodules and analysis of the associated tts box promoter. Mol Plant-Microbe Interact 21:1087–1093
Acknowledgments
This work was supported by grants from the Spanish Ministry of Science and Innovation (BIO2011-30229 and BIO2016-78409-R) and the Andalusia Government (P07-CVI-07500). SAJ and PNG are recipients of PhD grants from the VPPI of the University of Seville. We thank the Biology and NMR Facilities of the Centro de Investigación, Tecnología e Innovación (CITIUS), of the University of Seville. We also thank Isamar Moyano for technical assistance. Finally, we are grateful to the three anonymous reviewers who have evaluated this work.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Euan James.
Rights and permissions
About this article
Cite this article
Acosta-Jurado, S., Navarro-Gómez, P., Crespo-Rivas, JC. et al. The Sinorhizobium (Ensifer) fredii HH103 rkp-2 region is involved in the biosynthesis of lipopolysaccharide and exopolysaccharide but not in K-antigen polysaccharide production. Plant Soil 417, 415–431 (2017). https://doi.org/10.1007/s11104-017-3268-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-017-3268-z