Abstract
Background
Prior methods evaluating the changes in cerebral arterial blood volume (∆CaBV) assumed that brain blood transport distal to big cerebral arteries can be approximated with a non-pulsatile flow (CFF) model. In this study, a modified ∆CaBV calculation that accounts for pulsatile blood flow forward (PFF) from large cerebral arteries to resistive arterioles was investigated. The aim was to assess cerebral hemodynamic indices estimated by both CFF and PFF models while changing arterial blood carbon dioxide concentration (EtCO2) in healthy volunteers.
Materials and methods
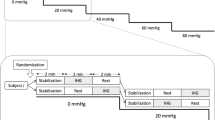
Continuous recordings of non-invasive arterial blood pressure (ABP), transcranial Doppler blood flow velocity (CBFVa), and EtCO2 were performed in 53 young volunteers at baseline and during both hypo- and hypercapnia. The time constant of the cerebral arterial bed (τ) and critical closing pressure (CrCP) were estimated using mathematical transformations of the pulse waveforms of ABP and CBFVa, and with both pulsatile and non-pulsatile models of ∆CaBV estimation. Results are presented as median values ± interquartile range.
Results
Both CrCP and τ gave significantly lower values with the PFF model when compared with the CFF model (p ≪ 0.001 for both). In comparison to normocapnia, both CrCP and τ determined with the PFF model increased during hypocapnia [CrCPPFF (mm Hg): 5.52 ± 8.78 vs. 14.36 ± 14.47, p = 0.00006; τPFF (ms): 47.4 ± 53.9 vs. 72.8 ± 45.7, p = 0.002] and decreased during hypercapnia [CrCPPFF (mm Hg): 5.52 ± 8.78 vs. 2.36 ± 7.05, p = 0.0001; τPFF (ms): 47.4 ± 53.9 vs. 29.0 ± 31.3, p = 0.0003]. When the CFF model was applied, no changes were found for CrCP during hypercapnia or in τ during hypocapnia.
Conclusion
Our results suggest that the pulsatile flow forward model better reflects changes in CrCP and in τ induced by controlled alterations in EtCO2.




Similar content being viewed by others
References
Avezaat CJJ, van Eijndhoven JHM. The role of the pulsatile pressure variations in intracranial pressure monitoring. Neurosurg Rev. 1986;9:113–20.
Alperin N, Sivaramakrishnan A, Lichtor T. Magnetic resonance imaging-based measurements of cerebrospinal fluid and blood flow as indicators of intracranial compliance in patients with Chiari malformation. J Neurosurg. 2005;103:46–52.
Stoquart-Elsankari S, Lehmann P, Villette A, Czosnyka M, Meyer M-E, Deramond H, et al. A phase-contrast MRI study of physiologic cerebral venous flow. J Cereb Blood Flow Metab. 2009;29:1208–15.
Kim DJ, Kasprowicz M, Carrera E, Castellani G, Zweifel C, Lavinio A, et al. The monitoring of relative changes in compartmental compliances of brain. Physiol Meas. 2009;30:647–59.
Carrera E, Kim DJ, Castellani G, Zweifel C, Smielewski P, Pickard JD, et al. Effect of hyper- and hypocapnia on cerebral arterial compliance in normal subjects. J Neuroimaging. 2011;21:121–5.
Nasr N, Czosnyka M, Pavy-Le Traon A, Custaud M-A, Liu X, Varsos GV, et al. Baroreflex and cerebral autoregulation are inversely correlated. Circ J. 2014;78:2460–7.
Czosnyka M, Richards HK, Reinhard M, Steiner L, Budohoski K, Smielewski P, et al. Cerebrovascular time constant: dependence on cerebral perfusion pressure and end-tidal carbon dioxide concentration. Neurol Res. 2012;34:17–24.
Kasprowicz M, Diedler J, Reinhard M, Carrera E, Steiner LA, Smielewski P, et al. Time constant of the cerebral arterial bed in normal subjects. Ultrasound Med Biol. 2012;38:1129–37.
Kasprowicz M, Diedler J, Reinhard M, Carrera E, Smielewski P, Budohoski KP, et al. Time constant of the cerebral arterial bed. Acta Neurochir Suppl. 2012;114:17–21.
Varsos GV, Richards H, Kasprowicz M, Budohoski KP, Brady KM, Reinhard M, et al. Critical closing pressure determined with a model of cerebrovascular impedance. J Cereb Blood Flow Metab. 2013;33:235–43.
Kasprowicz M, Czosnyka M, Soehle M, Smielewski P, Kirkpatrick PJ, Pickard JD, et al. Vasospasm shortens cerebral arterial time constant. Neurocrit Care. 2012;16:213–8.
Carrera E, Kim D-J, Castellani G, Zweifel C, Smielewski P, Pickard JD, et al. Cerebral arterial compliance in patients with internal carotid artery disease. Eur J Neurol. 2011;18:711–8.
Varsos GV, Budohoski KP, Czosnyka M, Kolias AG, Nasr N, Donnelly J, et al. Cerebral vasospasm affects arterial critical closing pressure. J Cereb Blood Flow Metab. 2015;35:285–91.
Varsos GV, Kolias AG, Smielewski P, Brady KM, Varsos VG, Hutchinson PJ, et al. A noninvasive estimation of cerebral perfusion pressure using critical closing pressure. J Neurosurg. 2015;123:638–48.
Carrera E, Kim D-J, Castellani G, Zweifel C, Czosnyka Z, Kasprowicz M, et al. What shapes pulse amplitude of intracranial pressure? J Neurotrauma. 2010;27:317–24.
Ambarki K, Baledent O, Kongolo G, Bouzerar R, Fall S, Meyer ME. A new lumped-parameter model of cerebrospinal hydrodynamics during the cardiac cycle in healthy volunteers. IEEE Trans Biomed Eng. 2007;54:483–91.
Czosnyka M, Smielewski P, Kirkpatrick P, Piechnik S, Laing R, Pickard JD. Continuous monitoring of cerebrovascular pressure-reactivity in head injury. Acta Neurochir Suppl. 1998;71:74–7.
Czosnyka M, Guazzo E, Whitehouse M, Smielewski P, Czosnyka Z, Kirkpatrick P, et al. Significance of intracranial pressure waveform analysis after head injury. Acta Neurochir. 1996;138:531–42.
Poulin MJ, Robbins PA. Indexes of flow and cross-sectional area of the middle cerebral artery using doppler ultrasound during hypoxia and hypercapnia in humans. Stroke. 1996;27:2244–50.
Valdueza JM, Balzer JO, Villringer A, Vogl TJ, Kutter R, Einhäupl KM. Changes in blood flow velocity and diameter of the middle cerebral artery during hyperventilation: assessment with MR and transcranial Doppler sonography. Am J Neuroradiol. 1997;18:1929–34.
Henriksen JH, Fuglsang S, Bendtsen F, Christensen E, Møller S. Arterial compliance in patients with cirrhosis: stroke volume-pulse pressure ratio as simplified index. Am J Physiol Gastrointest Liver Physiol. 2001;280:G584–94.
Panerai RB, Salinet ASM, Brodie FG, Robinson TG. The influence of calculation method on estimates of cerebral critical closing pressure. Physiol Meas. 2011;32:467–82.
Altman BjM. DG. Statistics notes: calculating correlation coefficients with repeated observations: part 1-correlation within subjects. BMJ. 1995;310:446.
Bland JM, Altman DG. Calculating correlation coefficients with repeated observations: Part 2-correlation between subjects. BMJ. 1995;310:633.
Varsos GV, Kasprowicz M, Smielewski P, Czosnyka M. Model-based indices describing cerebrovascular dynamics. Neurocrit Care. 2014;20:142–57.
Brothers RM, Zhang R. CrossTalk opposing view: the middle cerebral artery diameter does not change during alterations in arterial blood gases and blood pressure. J Physiol. 2016;594:4077–9.
Hoiland RL, Ainslie PN. CrossTalk proposal: The middle cerebral artery diameter does change during alterations in arterial blood gases and blood pressure. J Physiol. 2016;594:4073–5.
Djurberg HG, Seed RF, Price Evans DA, Brohi FA, Pyper DL, Tjan GT, et al. Lack of effect of CO2 on cerebral arterial diameter in man. J Clin Anesth. 1998;10:646–51.
Serrador JM, Picot P, Rutt BK, Shoemaker JK, Bondar RL. MRI measures of middle cerebral artery diameter in conscious humans during simulated orthostasis. Stroke. 2000;31:1672–8.
Schreiber SJ, Gottschalk S, Weih M, Villringer A, Valdueza JM. Assessment of blood flow velocity and diameter of the middle cerebral artery during the acetazolamide provocation test by use of transcranial Doppler sonography and MR imaging. AJNR Am J Neuroradiol. 2000;21:1207–11.
Verbree J, Bronzwaer A-SGT, Ghariq E, Versluis MJ, Daemen MJAP., van Buchem MA, et al. Assessment of middle cerebral artery diameter during hypocapnia and hypercapnia in humans using ultra-high-field MRI. J Appl Physiol. 2014;117:1084–9.
Coverdale NS, Lalande S, Perrotta A, Shoemaker JK. Heterogeneous patterns of vasoreactivity in the middle cerebral and internal carotid arteries. Am J Physiol- Hear Circ Physiol. 2015;308:H1030–8.
Kim S-G, Harel N, Jin T, Kim T, Lee P, Zhao F. Cerebral blood volume MRI with intravascular superparamagnetic iron oxide nanoparticles. NMR Biomed. 2013;26:949–62.
Acknowledgements
We thank Krystian Gruszczyński, Msc. Eng. for assistance with data collection and Tomasz Szczepański, PhD for reviewing medical history and physical examination.
Funding
This research was supported by the National Science Center (Poland) under Grant No. UMO-2013/10/E/ST7/00117.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
ICM + Software is licensed by Cambridge Enterprise, Cambridge, UK, http://www.neurosurg.cam.ac.uk/icmplus/. Prof. Czosnyka has a financial interest in a fraction of the licensing fee for ICM + software. The other authors declare that they have no conflicts of interest.
Informed consent
The protocol complied with the Declaration of Helsinki of the World Medical Association, and all participants gave written informed consent before participating in the study.
Research involving human and animal rights
The study was approved by the bioethical committee of the Wroclaw Medical University (Permission No. KB-170/2014).
Rights and permissions
About this article
Cite this article
Uryga, A., Kasprowicz, M., Calviello, L. et al. Assessment of cerebral hemodynamic parameters using pulsatile versus non-pulsatile cerebral blood outflow models. J Clin Monit Comput 33, 85–94 (2019). https://doi.org/10.1007/s10877-018-0136-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10877-018-0136-1