Abstract
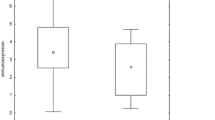
Elevated COX-2 activity is associated with the development of chronic lung diseases leading to bronchial obstruction, including sarcoidosis. The aim of the study was to examine expression pattern of COX-2 messenger RNA (mRNA). Expression was performed by q-PCR method in bronchoalveolar lavage (BAL) cells and peripheral blood (PB) lymphocytes in sarcoidosis patients (n = 61) and control group (n = 30). Analysis of COX-2 mRNA expression level in BAL fluid and PB revealed downregulation in sarcoidosis and control groups. In PB lymphocytes, the statistically significant difference between patients and controls was observed (P = 0.003, Mann–Whitney U test), with higher expression in patients. There were no statistically significant differences between patients without and with parenchymal involvement (stages I vs. II–IV), between patients with acute vs. insidious onset of disease and between patients with abnormal vs. normal spirometry (P > 0.05, Mann–Whitney U test). Results suggest that expression of COX-2 mRNA in patients with pulmonary sarcoidosis is not related to clinical classifications.



Similar content being viewed by others
References
Iannuzzi, M.C., B.A. Rybicki, and A.S. Teirstein. 2007. Sarcoidosis. The New England Journal of Medicine 357: 2153–2165.
Baughman, R.P., A.S. Teirstein, M.A. Judson, M.D. Rossman, H. Yeager Jr., E.A. Bresnitz, L. DePalo, G. Hunninghake, M.C. Iannuzzi, C.J. Johns, et al. 2001. Clinical characteristics of patients in a case control study of sarcoidosis. American Journal of Respiratory and Critical Care Medicine 164: 1885–1889.
Pueringer, R.J., D.A. Schwartz, C.S. Dayton, S.R. Gilbert, and G.W. Hunninghake. 1993. The relationship between alveolar macrophage TNF, IL-1, and PGE2 release, alveolitis, and disease severity in sarcoidosis. Chest 3: 832–838.
Coward, W.R., K. Watts, C.A. Feghali-Bostwick, A. Knox, and L. Pang. 2009. Defective histone acetylation is responsible for the diminished expression of cyclooxygenase 2 in idiopathic pulmonary fibrosis. Molecular and Cellular Biology 15: 4325–39.
Bauman, K.A., S.H. Wettlaufer, K. Okunishi, K.M. Vannella, J.S. Stoolman, S.K. Huang, A.J. Courey, E.S. White, C.M. Hogaboam CM, R.H. Simon, et al. 2010. The antifibrotic effects of plasminogen activation occur via prostaglandin E2 synthesis in humans and mice. Journal of Clinical Investigation 6: 1950–60.
Crofford, L.J. 1997. COX-1 and COX-2 tissue expression: Implications and predictions. Journal of Rheumatology. Supplement 49: 15–9.
Korn, J.H. 1983. Fibroblast prostaglandin E2 synthesis. Persistence of an abnormal phenotype after short-term exposure to mononuclear cell products. Journal of Clinical Investigation 5: 1240–1246.
Goldstein, R.H., and P. Polgar. 1982. The effect and interaction of bradykinin and prostaglandins on protein and collagen production by lung fibroblasts. Journal of Biological Chemistry 15: 8630–8633.
Petkova, D.K., C.A. Clelland, J.E. Ronan, S. Lewis, and A.J. Knox. 2003. Reduced expression of cyclooxygenase (COX) in idiopathic pulmonary fibrosis and sarcoidosis. Histopathology 4: 381–386.
O’Neill, G.P., and A.W. Ford-Hutchinson. 1993. Expression of mRNA for cyclooxygenase-1 and cyclooxygenase-2 in human tissues. FEBS Letters 13: 156–160.
Asano, K., C.M. Lilly, and J.M. Drazen. 1996. Prostaglandin G/H synthase-2 is the constitutive and dominant isoform in cultured human lung epithelial cells. American Journal of Physiology 271: 126–131.
Xaubet, A., W.J. Fu, M. Li, A. Serrano-Mollar, J. Ancochea, M. Molina-Molina, E. Rodriguez-Becerra, F. Morell, J.M. Rodríguez-Arias, J. Pereda, et al. 2010. A haplotype of cyclooxygenase-2 gene is associated with idiopathic pulmonary fibrosis. Sarcoidosis, Vasculitis, and Diffuse Lung Diseases 27: 121–130.
Bonner, J.C., A.B. Rice, J.L. Ingram, C.R. Moomaw, A. Nyska, A. Bradbury, A.R. Sessoms, P.C. Chulada, D.L. Morgan, D.C. Zeldin, et al. 2002. Susceptibility of cyclooxygenase-2-deficient mice to pulmonary fibrogenesis. American Journal of Pathology 161: 459–470.
Hodges, R.J., R.G. Jenkins, C.P. Wheeler-Jones, D.M. Copeman, S.E. Bottoms, G.J. Bellingan, C.B. Nanthakumar, G.J. Laurent, S.L. Hart, M.L. Foster, et al. 2004. Severity of lung injury in cyclooxygenase-2-deficient mice is dependent on reduced prostaglandin E(2) production. American Journal of Pathology 165: 1663–1676.
Lopez-Campos, J.L., D. Rodriguez-Rodriguez, E. Rodriguez-Becerra, I. Alfageme Michavila, J.F. Guerra, F.J. Hernandez, A. Casanova, J. de Córdoba Gamero Fernández, A. Romero-Ortiz, et al. 2009. Cyclooxygenase-2 polymorphisms confer susceptibility to sarcoidosis but are not related to prognosis. Respiratory Medicine 103: 427–433.
Hill, M.R., A. Papafili, H. Booth, P. Lawson, M. Hubner, H. Beynon, C. Read, G. Lindahl, R.P. Marshall, R.J. McAnulty, et al. 2006. Functional prostaglandin-endoperoxide synthase 2 polymorphism predicts poor outcome in sarcoidosis. American Journal of Respiratory and Critical Care Medicine 15: 915–922.
Ianuzzi, M.C., B.A. Rybicki, and A.S. Teirstein. 2007. Sarcoidosis. NEJM 357: 2153–2165.
American Thoracic Society, European Respiratory Society, and World Association of Sarcoidosis and Other Granulomatous Disorders. 1999. Statement on sarcoidosis. American Journal Respiratory Critical Care Medicine 160: 736–755.
Chciałowski, A., J. Chorostowska-Wynimko, A. Fal, R. Pawłowicz, and J. Domagał-Kulawik. 2011. Recommendation of the Polish Respiratory Society for bronchoalveolar lavage (BAL) sampling processing and analysis methods. Pneumonologia i Alergologia Polska 79: 75–89.
Wilborn, J., L.J. Crofford, M.D. Burdick, S.L. Kunkel, R.M. Strieter, and M. Peters-Golden. 1995. Cultured lung fibroblasts isolated from patients with idiopathic pulmonary fibrosis have a diminished capacity to synthesize prostaglandin E2 and to express cyclooxygenase-2. Journal of Clinical Investigation 95: 1861–1868.
Peebles, R.S.J., K. Hashimoto, J.D. Morrow, R. Dworski, R.D. Collins, Y. Hashimoto, J.W. Christman, K.H. Kang, K. Jarzecka, J. Furlong, et al. 2002. Selective cyclooxygenase-1 and -2 inhibitors each increase allergic inflammation and airway hyperresponsiveness in mice. American Journal of Respiratory and Critical Care Medicine 165: 1154–1160.
Xaubet, A., J. Roca-Ferrer, L. Pujols, J. Ramírez, J. Mullol, A. Marin-Arguedas, A. Torrego, J.M. Gimferrer, and C. Picado. 2004. Cyclooxygenase-2 is up-regulated in lung parenchyma of chronic obstructive pulmonary disease and down-regulated in idiopathic pulmonary fibrosis. Sarcoidosis, Vasculitis, and Diffuse Lung Diseases 21: 35–42.
Lappi-Blanco, E., R. Kaarteenaho-Wiik, P.K. Maasilta, S. Anttila, P. Pääkkö, and H.J. Wolff. 2006. COX-2 is widely expressed in metaplastic epithelium in pulmonary fibrous disorders. American journal of Clinical Pathology 126: 717–24.
Christophi, G.P., T. Caza, C. Curtiss, D. Gumber, P.T. Massa, and S.K. Landas. 2014. Gene expression profiles in granuloma tissue reveal novel diagnostic markers in sarcoidosis. Experimental and Molecular Pathology 96: 393–399.
Hodges, R.J., R.G. Jenkins, C.P. Wheeler-Jones, D.M. Copeman, S.E. Bottoms, G.J. Bellingan, C.B. Nanthakumar, G.J. Laurent, S.L. Hart, and M.L. Foster. 2004. Severity of lung injury in cyclooxygenase-2-deficient mice is dependent on reduced prostaglandin E(2) production. American Journal of Pathology 165: 1663–76.
Brown, J.R., and R.N. DuBois. 2004. Cyclooxygenase as a target in lung cancer. Clinical Cancer Research 15: 4266–4269.
Noor, A., and K.S. Knox. 2007. Immunopathogenesis of sarcoidosis. Clinics in Dermatology 25: 250–258.
Hastürk, S., B. Kemp, S.K. Kalapurakal, J.M. Kurie, W.K. Hong, and J.S. Lee. 2002. Expression of cyclooxygenase-1 and cyclooxygenase-2 in bronchial epithelium and nonsmall cell lung carcinoma. Cancer 15: 1023–1031.
Zhou, T., W. Zhang, N.J. Sweiss, E.S. Chen, D.R. Moller, K.S. Knox, S.F. Ma, M.S. Wade, I. Noth, R.F. Machado, et al. 2012. Peripheral blood gene expression as a novel genomic biomarker in complicated sarcoidosis. PLoS ONE 7(9), e44818.
Davies, P., and D.E. MacIntyre: Prostaglandins and inflammation. Inflammation: Basic Principles and Clinical Correlates. J.I. Gallin, I.M.Goldstein, and R. Snyderman, editors. Raven Press, Ltd., New York. 123–137, 1992
Pablos, J.L., B. Santiago, P.E. Carreira, M. Galindo, and J.J. Gomez-Reino. 1999. Cyclooxygenase-1 and -2 are expressed by human T cells. Clinical and Experimental Immunology 115: 86–90.
Hinz, B., K. Brune, and A. Pahl. 2000. Prostaglandin E(2) upregulates cyclooxygenase-2 expression in lipopolysaccharide-stimulated RAW 264.7 macrophages. Biochemical and Biophysical Research Communications 272: 744–748.
Bonazzi, A., M. Bolla, C. Buccellati, A. Hernandez, S. Zarini, T. Viganò, F. Fumagalli, S. Viappiani, S. Ravasi, P. Zannini, et al. 2000. Effect of endogenous and exogenous prostaglandin E(2) on interleukin-1 beta-induced cyclooxygenase-2 expression in human airway smooth-muscle cells. American Journal of Respiratory and Critical Care Medicine 162: 2272–2277.
Miyara, M., Z. Amoura, C. Parizot, C. Badoual, K. Dorgham, S. Trad, M. Kambouchner, D. Valeyre, C. Chapelon-Abric, P. Debré, et al. 2006. The immune paradox of sarcoidosis and regulatory T cells. Journal of Experimental Medicine 20: 359–370.
Akasaki, Y., G. Liu, N.H. Chung, M. Ehtesham, K.L. Black, and J.S. Yu. 2004. Induction of a CD4+ T regulatory type 1 response by cyclooxygenase-2-overexpressing glioma. Journal of Immunology 173: 4352–4359.
Acknowledgments
This work was financed by the Medical Univeristy of Lodz, number of subsidy: 503/1-151/-4/503-11-002.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The study was approved by the Ethics Committee at the Medical University of Lodz (RNN/141/10/KE). Written informed consent was obtained from each patient.
Conflict of Interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Kiszałkiewicz, J., Piotrowski, W.J., Pastuszak-Lewandoska, D. et al. Altered Cyclooxygenase-2 Expression in Pulmonary Sarcoidosis is not Related to Clinical Classifications. Inflammation 39, 1302–1309 (2016). https://doi.org/10.1007/s10753-016-0362-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10753-016-0362-y