Abstract
Background and Aims
Glypican-1 (GPC1), a cell-surface heparan sulfate proteoglycan, promotes the pathogenesis of many human cancers. This study focuses on the role of GPC1 in the promotion of cell proliferation and motility in esophageal squamous cell carcinoma (ESCC).
Methods
The expression and distribution of GPC1 were measured in tumor tissues from 248 ESCC patients using immunohistochemical (IHC) assays. Cell counting (kit-8), flow cytometry, Transwell, wound healing, IHC, and Western blotting assays were performed to examine the molecular mechanisms that underlie how GPC1 enhances cell proliferation and motility.
Results
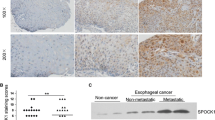
The level of GPC1 was higher in ESCC tumor samples than in para-tumor tissues (IHC score: 5.42 ± 2.15 vs. 0.86 ± 0.96). Ectopic overexpression of GPC1 in EC9706 cells promoted cell growth and the G1/S phase transition; conversely, GPC1 knockdown in Eca109 cells attenuated cell proliferation and induced G2/M phase arrest. In addition, GPC1 upregulation enhanced ESCC cell motility and induced epithelial mesenchymal transition (EMT), as demonstrated by the aberrant expression of EMT markers. Mechanistically, we demonstrated that GPC1 increased levels of p-Akt and β-catenin and reduced PTEN expression in ESCC.
Conclusions
Our study indicated that GPC1 promotes the aggressive proliferation of ESCC cells by regulating the PTEN/Akt/β-catenin pathway. GPC1 may be a promising target for ESCC treatment.






Similar content being viewed by others
References
Rustgi AK, El-Serag HB. Esophageal carcinoma. N Engl J Med. 2014;371:2499–2509.
Murphy G, McCormack V, Abedi-Ardekani B, et al. International cancer seminars: a focus on esophageal squamous cell carcinoma. Ann Oncol. 2017;28:2086–2093.
Song HH, Filmus J. The role of glypicans in mammalian development. Biochim Biophys Acta. 2002;1573:241–246.
Jen YH, Musacchio M, Lander AD. Glypican-1 controls brain size through regulation of fibroblast growth factor signaling in early neurogenesis. Neural Dev. 2009;4:33.
Chamorro-Jorganes A, Araldi E, Rotllan N, Cirera-Salinas D, Suarez Y. Autoregulation of glypican-1 by intronic microRNA-149 fine tunes the angiogenic response to FGF2 in human endothelial cells. J Cell Sci. 2014;127:1169–1178.
Qiao D, Meyer K, Mundhenke C, Drew SA, Friedl A. Heparan sulfate proteoglycans as regulators of fibroblast growth factor-2 signaling in brain endothelial cells. Specific role for glypican-1 in glioma angiogenesis. J Biol Chem. 2003;278:16045–16053.
Su G, Meyer K, Nandini CD, et al. Glypican-1 is frequently overexpressed in human gliomas and enhances FGF-2 signaling in glioma cells. Am J Pathol. 2006;168:2014–2026.
Whipple CA, Lander AD, Korc M. Discovery of a novel molecule that regulates tumor growth and metastasis. Sci World J. 2008;8:1250–1253.
Lu H, Niu F, Liu F, et al. Elevated glypican-1 expression is associated with an unfavorable prognosis in pancreatic ductal adenocarcinoma. Cancer Med. 2017;6:1181–1191.
Hara H, Takahashi T, Serada S, et al. Overexpression of glypican-1 implicates poor prognosis and their chemoresistance in oesophageal squamous cell carcinoma. Br J Cancer. 2016;115:66–75.
Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011;144:646–674.
Luo J, Manning BD, Cantley LC. Targeting the PI3 K-Akt pathway in human cancer: rationale and promise. Cancer Cell. 2003;4:257–262.
Li J, Simpson L, Takahashi M, et al. The PTEN/MMAC1 tumor suppressor induces cell death that is rescued by the AKT/protein kinase B oncogene. Can Res. 1998;58:5667–5672.
Tamura M, Gu J, Takino T, Yamada KM. Tumor suppressor PTEN inhibition of cell invasion, migration, and growth: differential involvement of focal adhesion kinase and p130Cas. Cancer Res. 1999;59:442–449.
Harada E, Serada S, Fujimoto M, et al. Glypican-1 targeted antibody-based therapy induces preclinical antitumor activity against esophageal squamous cell carcinoma. Oncotarget. 2017;8:24741–24752.
Shiau CE, Hu N, Bronner-Fraser M. Altering Glypican-1 levels modulates canonical Wnt signaling during trigeminal placode development. Dev Biol. 2010;348:107–118.
Ma J, Guo X, Zhang J, et al. PTEN gene induces cell invasion and migration via regulating AKT/GSK-3beta/beta-catenin signaling pathway in human gastric cancer. Dig Dis Sci. 2017;62:3415–3425. https://doi.org/10.1007/s10620-017-4764-y
Matsuda K, Maruyama H, Guo F, et al. Glypican-1 is overexpressed in human breast cancer and modulates the mitogenic effects of multiple heparin-binding growth factors in breast cancer cells. Can Res. 2001;61:5562–5569.
Aikawa T, Whipple CA, Lopez ME, et al. Glypican-1 modulates the angiogenic and metastatic potential of human and mouse cancer cells. J Clin Invest. 2008;118:89–99.
Qiao D, Yang H, Meyer K, Friedl A. Glypican-1 regulates anaphase promoting complex-cyclosome substrates and cell cycle progression in endothelial cells. Mol Biol Cell. 2008;19:2789–2801.
Bianco C, Strizzi L, Rehman A, et al. A Nodal-and ALK4-independent signaling pathway activated by Cripto-1 through Glypican-1 and c-Src. Can Res. 2003;63:1192–1197.
Kalluri R, Weinberg RA. The basics of epithelial–mesenchymal transition. J Clin Invest. 2009;119:1420–1428.
Lin DC, Hao JJ, Nagata Y, et al. Genomic and molecular characterization of esophageal squamous cell carcinoma. Nat Genet. 2014;46:467–473.
Haddadi N, Lin Y, Travis G, et al. PTEN/PTENP1: ‘regulating the regulator of RTK-dependent PI3 K/Akt signalling’, new targets for cancer therapy. Mol Cancer. 2018;17:37.
Trotman LC, Pandolfi PP. PTEN and p53: who will get the upper hand? Cancer Cell. 2003;3:97–99.
Mayo LD, Donner DB. A phosphatidylinositol 3-kinase/Akt pathway promotes translocation of Mdm2 from the cytoplasm to the nucleus. Proc Natl Acad Sci USA. 2001;98:11598–11603.
Qi J, Yu Y, Akilli Ozturk O, et al. New Wnt/beta-catenin target genes promote experimental metastasis and migration of colorectal cancer cells through different signals. Gut. 2016;65:1690–1701.
White BD, Chien AJ, Dawson DW. Dysregulation of Wnt/beta-catenin signaling in gastrointestinal cancers. Gastroenterology. 2012;142:219–232.
Mulholland DJ, Dedhar S, Wu H, Nelson CC. PTEN and GSK3beta: key regulators of progression to androgen-independent prostate cancer. Oncogene. 2006;25:329–337.
Grossmann AH, Yoo JH, Clancy J, et al. The small GTPase ARF6 stimulates beta-catenin transcriptional activity during WNT5A-mediated melanoma invasion and metastasis. Sci Signal. 2013;6:14.
Acknowledgments
This work was sponsored by Shanghai Sailing Program (No. 16YF1401500), National Nature Science Foundation of China (Nos. 81301820, 81472673, 81672720, 81672334), the Fund of Shanghai Science and Technology Commission(16ZR1406100), and the National Clinical Key Special Subject of China.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Li, J., Chen, Y., Zhan, C. et al. Glypican-1 Promotes Tumorigenesis by Regulating the PTEN/Akt/β-Catenin Signaling Pathway in Esophageal Squamous Cell Carcinoma. Dig Dis Sci 64, 1493–1502 (2019). https://doi.org/10.1007/s10620-019-5461-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-019-5461-9