Abstract
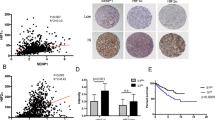
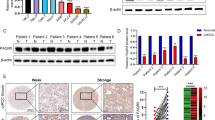
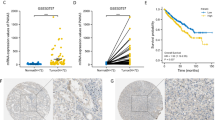
Energy-sensing pathways, normally coordinated by 5′ AMP-activated protein kinase (AMPK), are dysregulated in renal cell carcinoma (RCC). Obesity can accentuate the pre-existing pro-tumorigenic metabolic machinery in RCC cells through its associated obesogenic hormonal milieu, characterized by lower circulating levels of adiponectin. In RCC patients, low adiponectin levels associate clinically with more aggressive disease. We investigated the adiponectin signaling pathway in RCC, focusing on adiponectin receptor 1 (AdipoR1) and associated activation of AMPK. AdipoR1 protein in RCC and normal surrounding renal tissues was determined by Western blot analysis and immunohistochemistry. Anti-tumorigenic effects of adiponectin in RCC cells in vitro were investigated via VEGF and MMP ELISA and invasion assays. Using in vivo models of RCC, the effect of AdipoR1-knockdown (shRNA) on tumor latency, growth and dissemination were determined. AdipoR1 protein was significantly reduced in clear cell RCC specimens. Adiponectin treatment inhibited VEGF, MMP-2 and MMP-9 secretion and activity and invasive and migratory capacities of RCC cells. AMPKα1-knockdown (shRNA) attenuated adiponectin’s effects. In cells stably expressing AdipoR1-specific shRNA, AMPK activation by adiponectin was significantly reduced compared to cells expressing control shRNA. In vivo, AdipoR1 knockdown increased the growth, dissemination and angiogenesis of RCC. These findings suggest that deficiencies in the entire adiponectin hormonal axis (the hormone and its receptor) result in underactivation of AMPK leading to increased angiogenic and invasive capacities of RCC. The established link between obesity and RCC can therefore be further explained by the adiponectin deficiency in obese individuals together with reduced AdipoR1 protein in RCC.







Similar content being viewed by others
References
Chow WH, Gridley G, Fraumeni JF Jr, Jarvholm B (2000) Obesity, hypertension, and the risk of kidney cancer in men. N Engl J Med 343(18):1305–1311
Diez JJ, Iglesias P (2003) The role of the novel adipocyte-derived hormone adiponectin in human disease. Eur J Endocrinol 148(3):293–300
Goldstein BJ, Scalia R (2004) Adiponectin: a novel adipokine linking adipocytes and vascular function. J Clin Endocrinol Metab 89(6):2563–2568
Gavrila A, Chan JL, Yiannakouris N, Kontogianni M, Miller LC, Orlova C, Mantzoros CS (2003) Serum adiponectin levels are inversely associated with overall and central fat distribution but are not directly regulated by acute fasting or leptin administration in humans: cross-sectional and interventional studies. J Clin Endocrinol Metab 88(10):4823–4831
Trujillo ME, Scherer PE (2005) Adiponectin–journey from an adipocyte secretory protein to biomarker of the metabolic syndrome. J Intern Med 257(2):167–175
Yamauchi T, Hara K, Kubota N, Terauchi Y, Tobe K, Froguel P, Nagai R, Kadowaki T (2003) Dual roles of adiponectin/Acrp30 in vivo as an anti-diabetic and anti-atherogenic adipokine. Curr Drug Targets Immune Endocr Metabol Disord 3(4):243–254
Okamoto Y, Kihara S, Ouchi N, Nishida M, Arita Y, Kumada M, Ohashi K, Sakai N, Shimomura I, Kobayashi H, Terasaka N, Inaba T, Funahashi T, Matsuzawa Y (2002) Adiponectin reduces atherosclerosis in apolipoprotein E-deficient mice. Circulation 106(22):2767–2770
Chandran M, Phillips SA, Ciaraldi T, Henry RR (2003) Adiponectin: more than just another fat cell hormone? Diabetes Care 26(8):2442–2450
Miyoshi Y, Funahashi T, Kihara S, Taguchi T, Tamaki Y, Matsuzawa Y, Noguchi S (2003) Association of serum adiponectin levels with breast cancer risk. Clin Cancer Res 9(15):5699–5704
Wei EK, Giovannucci E, Fuchs CS, Willett WC, Mantzoros CS (2005) Low plasma adiponectin levels and risk of colorectal cancer in men: a prospective study. J Natl Cancer Inst 97(22):1688–1694
Dal Maso L, Augustin LS, Karalis A, Talamini R, Franceschi S, Trichopoulos D, Mantzoros CS, La Vecchia C (2004) Circulating adiponectin and endometrial cancer risk. J Clin Endocrinol Metab 89(3):1160–1163
Goktas S, Yilmaz MI, Caglar K, Sonmez A, Kilic S, Bedir S (2005) Prostate cancer and adiponectin. Urology 65(6):1168–1172
Michalakis K, Williams CJ, Mitsiades N, Blakeman J, Balafouta-Tselenis S, Giannopoulos A, Mantzoros CS (2007) Serum adiponectin concentrations and tissue expression of adiponectin receptors are reduced in patients with prostate cancer: a case control study. Cancer Epidemiol Biomarkers Prev 16(2):308–313
Pinthus JH, Kleinmann N, Tisdale B, Chatterjee S, Lu JP, Gillis A, Hamlet T, Singh G, Farrokhyar F, Kapoor A (2008) Lower plasma adiponectin levels are associated with larger tumor size and metastasis in clear-cell carcinoma of the kidney. Eur Urol 54(4):866–873
Horiguchi A, Ito K, Sumitomo M, Kimura F, Asano T, Hayakawa M (2008) Decreased serum adiponectin levels in patients with metastatic renal cell carcinoma. Jpn J Clin Oncol 38(2):106–111
Spyridopoulos TN, Petridou ET, Skalkidou A, Dessypris N, Chrousos GP, Mantzoros CS (2007) Low adiponectin levels are associated with renal cell carcinoma: a case–control study. Int J Cancer 120(7):1573–1578
Rini BI, Small EJ (2005) Biology and clinical development of vascular endothelial growth factor-targeted therapy in renal cell carcinoma. J Clin Oncol 23(5):1028–1043
Brakenhielm E, Veitonmaki N, Cao R, Kihara S, Matsuzawa Y, Zhivotovsky B, Funahashi T, Cao Y (2004) Adiponectin-induced antiangiogenesis and antitumor activity involve caspase-mediated endothelial cell apoptosis. Proc Natl Acad Sci USA 101(8):2476–2481
Chambers AF, Matrisian LM (1997) Changing views of the role of matrix metalloproteinases in metastasis. J Natl Cancer Inst 89(17):1260–1270
Cho NH, Shim HS, Rha SY, Kang SH, Hong SH, Choi YD, Hong SJ, Cho SH (2003) Increased expression of matrix metalloproteinase 9 correlates with poor prognostic variables in renal cell carcinoma. Eur Urol 44(5):560–566
Abdel-Wahed MM, Asaad NY, Aleskandarany M (2004) Expression of matrix metalloproteinase-2 in renal cell carcinoma. J Egypt Natl Cancer Inst 16(3):168–177
Kugler A, Hemmerlein B, Thelen P, Kallerhoff M, Radzun HJ, Ringert RH (1998) Expression of metalloproteinase 2 and 9 and their inhibitors in renal cell carcinoma. J Urol 160(5):1914–1918
Cumming AP, Hopmans SN, Vukmirovic-Popovic S, Duivenvoorden WC (2011) PSA affects prostate cancer cell invasion in vitro and induces an osteoblastic phenotype in bone in vivo. Prostate Cancer Prostatic Dis 14(4):286–294
Vukmirovic-Popovic S, Escott NG, Duivenvoorden WC (2008) Presence and enzymatic activity of prostate-specific antigen in archival prostate cancer samples. Oncol Rep 20(4):897–903
Duivenvoorden WC, Hirte HW, Singh G (1999) Transforming growth factor beta1 acts as an inducer of matrix metalloproteinase expression and activity in human bone-metastasizing cancer cells. Clin Exp Metastasis 17(1):27–34
Duivenvoorden WC, Hirte HW, Singh G (2001) Quantification of matrix metalloproteinase activity in plasma of patients enrolled in a BAY 12-9566 phase I study. Int J Cancer 91(6):857–862
Lhotak S, Elavathil LJ, Vukmirovic-Popovic S, Duivenvoorden WC, Tozer RG, Singh G (2001) Immunolocalization of matrix metalloproteinases and their inhibitors in clinical specimens of bone metastasis from breast carcinoma. Clin Exp Metastasis 18(6):463–470
Duivenvoorden WC, Beatty LK, Lhotak S, Hill B, Mak I, Paulin G, Gallino D, Austin RC, Pinthus JH (2013) Underexpression of the tumor suppressor LKB1 in clear cell renal cell carcinoma is common and confers growth advantage in vitro and in vivo. Br J Cancer 108(2):327–333
Yamauchi T, Kamon J, Ito Y, Tsuchida A, Yokomizo T, Kita S, Sugiyama T, Miyagishi M, Hara K, Tsunoda M, Murakami K, Ohteki T, Uchida S, Takekawa S, Waki H, Tsuno NH, Shibata Y, Terauchi Y, Froguel P, Tobe K, Koyasu S, Taira K, Kitamura T, Shimizu T, Nagai R, Kadowaki T (2003) Cloning of adiponectin receptors that mediate antidiabetic metabolic effects. Nature 423(6941):762–769
Kadowaki T, Yamauchi T (2005) Adiponectin and adiponectin receptors. Endocr Rev 26(3):439–451
Hardie DG (2008) AMPK: a key regulator of energy balance in the single cell and the whole organism. Int J Obes 32(Suppl 4):S7–S12
Robb VA, Karbowniczek M, Klein-Szanto AJ, Henske EP (2007) Activation of the mTOR signaling pathway in renal clear cell carcinoma. J Urol 177(1):346–352
Jemal A, Siegel R, Xu J, Ward E (2010) Cancer statistics, 2010. CA Cancer J Clin 60(5):277–300
Patard JJ, Rioux-Leclercq N, Fergelot P (2006) Understanding the importance of smart drugs in renal cell carcinoma. Eur Urol 49(4):633–643
Kroog GS, Motzer RJ (2008) Systemic therapy for metastatic renal cell carcinoma. Urol Clin N Am 35(4):687–701
Miller RE, Larkin JM (2009) Combination systemic therapy for advanced renal cell carcinoma. Oncologist 14(12):1218–1224
Bastien L, Culine S, Paule B, Ledbai S, Patard JJ, de la Taille A (2009) Targeted therapies in metastatic renal cancer in. BJU Int 103(10):1334–1342
Chou SH, Tseleni-Balafouta S, Moon HS, Chamberland JP, Liu X, Kavantzas N, Mantzoros CS (2010) Adiponectin receptor expression in human malignant tissues. Horm Cancer 1(3):136–145
Pfeiler G, Hudelist G, Wulfing P, Mattsson B, Konigsberg R, Kubista E, Singer CF (2010) Impact of AdipoR1 expression on breast cancer development. Gynecol Oncol 117(1):134–138
Barresi V, Grosso M, Giuffre G, Tuccari G, Barresi G (2009) The expression of adiponectin receptors Adipo-R1 and Adipo-R2 is associated with an intestinal histotype and longer survival in gastric carcinoma. J Clin Pathol 62(8):705–709
Byeon JS, Jeong JY, Kim MJ, Lee SM, Nam WH, Myung SJ, Kim JG, Yang SK, Kim JH, Suh DJ (2010) Adiponectin and adiponectin receptor in relation to colorectal cancer progression. Int J Cancer 127(12):2758–2767
Kim AY, Lee YS, Kim KH, Lee JH, Lee HK, Jang SH, Kim SE, Lee GY, Lee JW, Jung SA, Chung HY, Jeong S, Kim JB (2010) Adiponectin represses colon cancer cell proliferation via AdipoR1- and -R2-mediated AMPK activation. Mol Endocrinol 24(7):1441–1452
Taliaferro-Smith L, Nagalingam A, Zhong D, Zhou W, Saxena NK, Sharma D (2009) LKB1 is required for adiponectin-mediated modulation of AMPK-S6K axis and inhibition of migration and invasion of breast cancer cells. Oncogene 28(29):2621–2633
Arditi JD, Venihaki M, Karalis KP, Chrousos GP (2007) Antiproliferative effect of adiponectin on MCF7 breast cancer cells: a potential hormonal link between obesity and cancer. Horm Metab Res 39(1):9–13
Dieudonne MN, Bussiere M, Dos Santos E, Leneveu MC, Giudicelli Y, Pecquery R (2006) Adiponectin mediates antiproliferative and apoptotic responses in human MCF7 breast cancer cells. Biochem Biophys Res Commun 345(1):271–279
Dos Santos E, Benaitreau D, Dieudonne MN, Leneveu MC, Serazin V, Giudicelli Y, Pecquery R (2008) Adiponectin mediates an antiproliferative response in human MDA-MB 231 breast cancer cells. Oncol Rep 20(4):971–977
Moon HS, Chamberland JP, Aronis K, Tseleni-Balafouta S, Mantzoros CS (2011) Direct role of adiponectin and adiponectin receptors in endometrial cancer: in vitro and ex vivo studies in humans. Mol Cancer Ther 10(12):2234–2243
Sugiyama M, Takahashi H, Hosono K, Endo H, Kato S, Yoneda K, Nozaki Y, Fujita K, Yoneda M, Wada K, Nakagama H, Nakajima A (2009) Adiponectin inhibits colorectal cancer cell growth through the AMPK/mTOR pathway. Int J Oncol 34(2):339–344
Zakikhani M, Dowling RJ, Sonenberg N, Pollak MN (2008) The effects of adiponectin and metformin on prostate and colon neoplasia involve activation of AMP-activated protein kinase. Cancer Prev Res (Phila PA) 1(5):369–375
Bub JD, Miyazaki T, Iwamoto Y (2006) Adiponectin as a growth inhibitor in prostate cancer cells. Biochem Biophys Res Commun 340(4):1158–1166
Ishikawa M, Kitayama J, Yamauchi T, Kadowaki T, Maki T, Miyato H, Yamashita H, Nagawa H (2007) Adiponectin inhibits the growth and peritoneal metastasis of gastric cancer through its specific membrane receptors AdipoR1 and AdipoR2. Cancer Sci 98(7):1120–1127
Fukata S, Inoue K, Kamada M, Kawada C, Furihata M, Ohtsuki Y, Shuin T (2005) Levels of angiogenesis and expression of angiogenesis-related genes are prognostic for organ-specific metastasis of renal cell carcinoma. Cancer 103(5):931–942
Hawley SA, Boudeau J, Reid JL, Mustard KJ, Udd L, Makela TP, Alessi DR, Hardie DG (2003) Complexes between the LKB1 tumor suppressor, STRAD alpha/beta and MO25 alpha/beta are upstream kinases in the AMP-activated protein kinase cascade. J Biol 2(4):28
Hudes G, Carducci M, Tomczak P, Dutcher J, Figlin R, Kapoor A, Staroslawska E, Sosman J, McDermott D, Bodrogi I, Kovacevic Z, Lesovoy V, Schmidt-Wolf IG, Barbarash O, Gokmen E, O’Toole T, Lustgarten S, Moore L, Motzer RJ (2007) Temsirolimus, interferon alfa, or both for advanced renal-cell carcinoma. N Engl J Med 356(22):2271–2281
Al-Saleem T, Wessner LL, Scheithauer BW, Patterson K, Roach ES, Dreyer SJ, Fujikawa K, Bjornsson J, Bernstein J, Henske EP (1998) Malignant tumors of the kidney, brain, and soft tissues in children and young adults with the tuberous sclerosis complex. Cancer 83(10):2208–2216
Brugarolas JB, Vazquez F, Reddy A, Sellers WR, Kaelin WG Jr (2003) TSC2 regulates VEGF through mTOR-dependent and -independent pathways. Cancer Cell 4(2):147–158
Romanelli A, Dreisbach VC, Blenis J (2002) Characterization of phosphatidylinositol 3-kinase-dependent phosphorylation of the hydrophobic motif site Thr(389) in p70 S6 kinase 1. J Biol Chem 277(43):40281–40289
Chiang GG, Abraham RT (2005) Phosphorylation of mammalian target of rapamycin (mTOR) at Ser-2448 is mediated by p70S6 kinase. J Biol Chem 280(27):25485–25490
Linehan WM, Srinivasan R, Schmidt LS (2010) The genetic basis of kidney cancer: a metabolic disease. Nat Rev Urol 7(5):277–285
Tong WH, Sourbier C, Kovtunovych G, Jeong SY, Vira M, Ghosh M, Romero VV, Sougrat R, Vaulont S, Viollet B, Kim YS, Lee S, Trepel J, Srinivasan R, Bratslavsky G, Yang Y, Linehan WM, Rouault TA (2011) The glycolytic shift in fumarate-hydratase-deficient kidney cancer lowers AMPK levels, increases anabolic propensities and lowers cellular iron levels. Cancer Cell 20(3):315–327
Baba M, Hong SB, Sharma N, Warren MB, Nickerson ML, Iwamatsu A, Esposito D, Gillette WK, Hopkins RF 3rd, Hartley JL, Furihata M, Oishi S, Zhen W, Burke TR Jr, Linehan WM, Schmidt LS, Zbar B (2006) Folliculin encoded by the BHD gene interacts with a binding protein, FNIP1, and AMPK, and is involved in AMPK and mTOR signaling. Proc Natl Acad Sci USA 103(42):15552–15557
Vazquez-Martin A, Oliveras-Ferraros C, del Barco S, Martin-Castillo B, Menendez JA (2009) The antidiabetic drug metformin: a pharmaceutical AMPK activator to overcome breast cancer resistance to HER2 inhibitors while decreasing risk of cardiomyopathy. Ann Oncol 20(3):592–595
Konturek PC, Burnat G, Rau T, Hahn EG, Konturek S (2008) Effect of adiponectin and ghrelin on apoptosis of Barrett adenocarcinoma cell line. Dig Dis Sci 53(3):597–605
Miyazaki T, Bub JD, Uzuki M, Iwamoto Y (2005) Adiponectin activates c-Jun NH2-terminal kinase and inhibits signal transducer and activator of transcription 3. Biochem Biophys Res Commun 333(1):79–87
Acknowledgments
We are indebted to Drs. S. Popovic and J.C. Cutz, for sharing their expertise in pathology, to Dr. J.P. Lu for excellent technical assistance, and Dr. A. Kapoor for providing clinical RCC specimens. This work was supported by the Canadian Institutes of Health Research (Grant 93465), a young investigators research grant by the Northeastern section of the American Urological Association, McMaster Surgical Associates, and an early career award by Hamilton Health Sciences (to JHP).
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kleinmann, N., Duivenvoorden, W.C.M., Hopmans, S.N. et al. Underactivation of the adiponectin–adiponectin receptor 1 axis in clear cell renal cell carcinoma: implications for progression. Clin Exp Metastasis 31, 169–183 (2014). https://doi.org/10.1007/s10585-013-9618-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10585-013-9618-1