Abstract
Crop phenology changes are important indicators of climate change. Climate change impacts on crop phenology are generally investigated through statistical analysis of the relationship between growth period length and growth period mean temperature. However, growth periods may be either earlier or later in a given year; hence, changes in mean temperature indicate both the effects of climate change and those attributable to seasonal temperature differences. Failure to consider temperature change resulting from seasonal shifts can lead to biased estimation of warming trends and their corresponding impact on phenology. We evaluated this potential bias in rice phenology change in 892 phenology series from China by applying time series regression control for phenological dates. The results indicate that the true magnitudes of climate change for early rice, late rice, and single rice are 0.20–0.56, 0.23–0.86, and 0.28–0.38 K/decade, after correction for the effects of seasonal shifts. The effects of seasonal shifts of growth periods led to underestimates of the magnitude of climate change by 0.16–0.22 and 0.05–0.08 K/decade for early rice and single rice, respectively, and an overestimate of the effect for late rice of 0.02–0.06 K/decade. Correspondingly, the net warming impacts on growth period length after correcting for the effects of seasonal shifts were − 2.7 d/K for early rice, − 4.8 d/K for late rice, and − 3.1 d/K for single rice, which were weaker for early and single rice, but stronger for late rice, relative to previous reports. Changes in growth period length were most closely associated with variation in phenological dates, while their relationship with climate change was less pronounced. Our results indicate that earlier phenological dates and prolonged-duration cultivars have been adopted to offset the impact of climate change, providing further evidence of active adaptation of rice cultivation practice to climate change in China.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Plant phenology and growth stage duration are critical indicators of the responses of vegetation to climate change (Zhang et al. 2004; Estrella et al. 2007; Richardson et al. 2013). Significant changes in vegetation phenology have been identified globally and regionally (Piao et al. 2006; Brown et al. 2012). For crops, changes in phenological dates (PDs) and growth period lengths (GPLs) not only reflect variation in physiological processes (Zhao et al. 2016) but are also potentially related to alteration of cropping patterns (i.e., single cropping to double cropping; Qiu et al. 2017), crop exposure to and risk from seasonal climate extremes (Shimono 2011), potential crop yield (Asseng et al. 2011), and carbon flux (Wu et al. 2012). In addition, due to considerable human intervention, determination of the relative contributions of climate change and human activity (i.e., alterations in cultivars and agro-management) is vitally important for adaptive decision-making (Liu et al. 2018). Hence, alterations in crop phenology are of particular interest in the context of the changing climate.
Rice is currently one of the most important cereal grains globally and serves as a staple food source for more than half of the world’s population (Gross and Zhao 2014). In China, rice accounts for 31.6% of the grain crop area planted, and 36.3% of the total grain harvested (NBSC 2016), and it is vital for feeding the Chinese population. Given the warming trend in mainland China, several studies have explored the potential impact of climate change and cultivar shifts on rice phenology, primarily through statistical analysis of recorded rice phenology data (Tao et al. 2013; Zhang et al. 2013, 2014; Zhao et al. 2016). Published results are highly consistent, confirming significant changes in major rice PDs and GPLs, and the negative impact of temperature on GPLs (higher temperature has shortened GPLs); however, some critical issues remain controversial. First, empirical results indicate that the adoption of short-duration cultivars may be advisable (Zhang et al. 2013); however, adoption of long-duration cultivars has also been suggested (Liu et al. 2012; Tao et al. 2013). Second, while many early studies contended that temperature and cultivar shifts are the two major factors driving the change in rice PDs and GPLs, recent studies (Zhao et al. 2016; Hu et al. 2017) have suggested that (trans)planting dates are most closely associated with PDs and GPLs, while the effect of temperature is less marked.
Recent progress (Zhao et al. 2016; Hu et al. 2017) has highlighted a critical issue; namely that failure to consider PDs in statistical analyses could have led to over- or underestimation of the magnitude of climate change and its impact on phenology. This can be illustrated with a simple schematic chart (Fig. 1). The overall growth period mean temperature change has two components: temperature change induced by climate change, which shifts the entire annual temperature variation upward, and a change induced by seasonal shifts of growth period (Hu et al. 2017). Using the overall growth period average temperature change without considering the effect of seasonal shifts leads to a bias in warming estimates. For example, in case A, as the growth period shifts to earlier in spring, it falls into a lower temperature period. Consequently, the observed overall temperature change would be smaller than the net warming effect, thus underestimating the warming. Conversely, in case B, as the growth period shifts from early autumn to late summer, it falls into a higher temperature period. Consequently, the observed overall temperature change would be larger than the net warming effect, leading to overestimation (Fig. 1). Thus, for different rice cropping systems with varying growth periods, estimates of temperature sensitivity from phenological data are critically dependent on timing within the year.
The overall growth period temperature change is the net effect of climate warming and seasonal shift of growth period. Without considering the effects of seasonal shifts, climate warming effects could be over- or underestimated according to the timing of the period in the annual temperature variation and the direction of the shift
Correcting the bias in estimated warming due to seasonal shifts is important for understanding the true impact of climate change on crop phenology, and for measuring the relative importance of climatic and human factors, and development of adaptive strategies. Hu et al. (2017) used a growing degree day–based simulation to distinguish the effects of climate warming, cultivar shifts, and planting date shifts; however, whether their results can be supported by statistical data remains to be determined.
In this study, we revisited rice phenology change in China using phenological records from 157 agricultural meteorological stations, comprising almost all station-observed rice phenology records from China. We performed statistical analyses and paid special attention to the removal of confounding effects induced by seasonal shifts of growth periods. We controlled for phenological dates (PDs) when determining the climate change trend and used regression analysis to identify factors that contribute to changes in growth period length (GPL). Our aims were to (i) reveal the true size of the effect of climate change on rice growth periods, (ii) investigate the true effects of climate change on GPLs, and (iii) distinguish the impacts of changes in PD, climate, and cultivar on GPLs.
2 Materials and methods
2.1 Study area
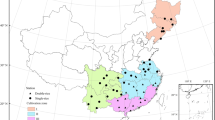
The study area covered the major rice production region in China, including three provinces in northeastern China, and 14 provinces along and south of the Yangtze River basin (Fig. 1). The study area comprised a total area of approximately 3.38 million km2, with about 29.85 million ha rice planted, representing 98% of the total rice area nationwide. There are two typical rice cropping systems in China, single-season cropping rice (hereafter as “single rice”) and double-season cropping rice (hereafter as “double rice”) (Nie and Peng 2017). Single rice is mainly planted in the northeast China plain, plains north of the Yangtze River, and in Sichuan, Yunnan, and Guizhou Provinces up to the upper streams of the Yangtze River. The typical cropping system is a rotation between rice and winter wheat. Double-cropping rice (rotation of early and late rice) is primarily planted in the provinces south of the mid-to-lower Yangtze River.
2.2 Data
Historical observations of rice phenology for the years 1980–2010 were collected from 200 agrometeorological stations operated by the Chinese Meteorological Administration in the study area (http://data.cma.cn/data/cdcindex/cid/4ee1c7fce9cd6a5f.html) (Fig. 2). From this dataset, PDs of sowing, transplanting, heading, and maturity were selected. Data were carefully checked to manage missing values. Phenology series with data for less than 10 years or no observation in 2005–2010 were not considered in our analysis. This resulted in the inclusion of 892 phenology series from 157 stations in the analysis, with 348 series from 88 stations for single rice, 269 series from 69 stations for early rice, and 275 series from 69 stations for late rice.
Historical daily weather data matching the spatial location and time period of the phenological records were required for our analysis. As the spatial locations of national reference meteorological stations collecting daily weather data are mostly different from those of agrometeorological stations, interpolation is generally required to match the two datasets (i.e., Zhang et al. 2013). As interpolation methodology can result in substantial uncertainties, we followed the approach of Zhao et al. (2016), using the China Meteorological Forcing Dataset (http://westdc.westgis.ac.cn/data/7a35329c-c53f-4267-aa07- e0037d913a21) (Chen et al. 2011; He and Yang 2011). The dataset contains three-hourly temperature, precipitation, and solar radiation values, with a spatial resolution of 0.1°. The 3-h data was aggregated to daily mean temperature, and further aggregated into growth period mean temperature (T). The computed growth period means were verified using observations from the national reference stations of the China Meteorological Administration, and good agreement was found.
2.3 Statistical analyses
Trends in rice phenology and climatic factors have frequently been calculated using linear regression with respect to a time index (year t) (Hu et al. 2017). To evaluate changes in rice phenology, trend analysis was applied to each time series of phenological dates (PDs) using linear regression with time index (year t). Changes in growth period lengths (GPLs) were also analyzed using linear regression analysis. In the literature, there are mainly two ways of dividing growth stages. The first type considers the periods of transplanting-to-heading and heading-to-maturity (i.e., Tao et al. 2013; Zhao et al. 2016), and the second type considers the periods of emerging-to-heading and heading-to-maturity (i.e., Zhang et al. 2013; Hu et al. 2017). Taking the advantages of both types, we further divided the vegetation growth stage by transplanting, which is important as the growing environment of rice differ substantially before and after transplanting in China. Therefore, our GPLs included the length of the nursery growth period (NGP) from sowing to transplanting, vegetative growth period (VGP) from transplanting-to-heading, and reproductive growth period (RGP) from heading-to-maturity. These periods were determined from PD records, and linear regression applied to evaluate the changes. Similar methods were also applied to calculate growth period average daily precipitation P and average daily solar radiation S values, for each of the periods: NGP, VGP, and RGP.
Applying the same method to detect trends in climate warming will lead to serious bias. As shown in Fig. 1, growth period mean temperature change contains two components, change associated with climate change (decadal signal) and change associated with seasonal shifts of growth period. We need to decompose those two components to reveal the true magnitude of climate change. This could be achieved by introducing a controlling variable indicating the timing of growth period into the linear regression analysis, i.e., the starting dates of the growth period:
where T is the growth period mean temperature, PD− refers to the starting date of its corresponding growth period, t is the time index of year, γ0 is the intercept of the regression, and μ is the random error. For NGP, VGP, and RGP, PD− indicates sowing, transplanting, and heading dates, respectively. The temperature change associated with the seasonal shifts of growth period is represented by γ1, which describes by how many degrees the temperature will change if the starting PD changes by 1 day (K/d), assuming all other conditions remain unchanged. The linear temperature trend associated with year index t is γ2, which represents how many degrees the temperature will change if it is 1 year later (K/a), assuming all other conditions remain unchanged. This is an exact measure of true climate change trend, after correction for seasonal shift effects.
When investigating the relationship between rice phenology and climatic factors, the existing literature generally conducts linear regression analysis of GPL on growth period average temperature T with time index t (i.e., Zhang et al. 2012), or equivalently uses partial correlation analysis between detrended GPL and detrended T (i.e., Tao et al. 2013). Under such analytical framework, a similar issue arises to the biased estimation of warming magnitude as the shift in growth period is not accounted for. In order to address such an issue, we again added the starting phenological dates (PD−) of their corresponding growth period into the regression, as follows:
where GPL refers to the growth period length, including NGP, VGP, and RGP for all three rice cropping systems. The β1 parameters are regression coefficients. T is the growth period mean temperature, PD− denotes the starting phenological dates for the corresponding growth period, and t is the year index. In this way, any seasonal difference in growth period average temperature due to a shift in the growth period can be controlled for, as long as climate change has not changed the seasonal pattern of temperature.
Given the regression models above, a change in a specific GPL is explained by three components: (a) the true climate change impact (β1), which measures GPL’s response to 1 K in temperature change due to climate change, as effects from phenological date shifts and time trend have been controlled for; (b) cultivar shifts impact (β3), the effect of time trend after temperature and phenological dates were controlled for; and (c) seasonal shift effect, which measures GPL’s response to 1 day change (earlier/later) of the starting phenological dates, assuming all other conditions remain unchanged. As starting phenological dates are mostly highly correlated to each other (Zhao et al. 2016), this effect can further be translated to the impact from the earlier/later transplanting or sowing dates, and therefore linked to planting decisions.
The fitting of Eq. (2) for each GPL series was carried out in R 3.3.4. Multi-collinearity diagnostics was also carried out when fitting the model. The variance inflation factor (VIF) was used to test the existence of multi-collinearity. For results with VIF > 10, regression results shall not be considered for later interpretation. Finally, descriptive statistics of all the regression coefficients was derived.
3 Results
3.1 Changes in rice phenology during 1980–2010
The detected changes in rice phenology during 1980–2010 are presented in Figs. 3 and 4.
For early rice, all four phenological dates (PDs) were found to advance to earlier times within the year during the period 1980–2010. The median values of sowing, transplanting, heading, and maturity dates changed by − 0.66, − 2.10, − 1.06, and − 0.54 d/decade, respectively. Advancing trends in sowing, transplanting, heading, and maturity dates were detected at 56%, 72%, 68%, and 57% of the 69 stations, and the advances were significant (p < 0.05) at 26%, 45%, 28%, and 18% of stations, respectively. Conversely, delayed trends in sowing, transplanting, heading, and maturity dates were significant (p < 0.05) in only 19%, 9%, 10%, and 10% of stations, respectively.
For late rice, median values of sowing, transplanting, heading, and maturity dates changed by 2.67, − 0.63, − 0.27, and − 0.77 d/decade, respectively. Trends towards earlier dates in sowing, transplanting, heading, and maturity dates were detected in 25%, 64%, 57%, and 58% of the 69 stations, and these shifts were significant (p < 0.05) in 10%, 23%, 25%, and 32% of the stations, respectively. Delayed trends in sowing, transplanting, heading, and maturity dates were significant (p < 0.05) in 60%, 14%, 17%, and 14% of stations, respectively.
For single rice, trends were detected for earlier sowing and transplanting dates, and delayed heading and maturity dates, respectively. The median value of sowing, transplanting, heading, and maturity dates changed by − 0.44, − 1.24, 0.69, and 1.44 d/decade, respectively. Earlier sowing, transplanting, heading, and maturity dates were detected in approximately 56%, 64%, 43%, and 33% of 88 stations, and these changes were significant (p < 0.05) in 29%, 32%, 12%, and 11% of stations, respectively. Delayed sowing, transplanting, heading, and maturity dates were significant (p < 0.05) in 20%, 12%, 25%, and 29% of stations, respectively.
Corresponding to the changes in phenological dates, significant changes were also detected in growth period lengths. For early rice, the median lengths of nursery growth period (NGP), vegetative growth period (VGP), and reproductive growth period (RGP) changed by − 1.9, 2.7, and − 0.0 d/decade. Prolonged lengths of NGP, VGP, and RGP were detected in 26%, 73%, and 49% of the 69 stations, and the changes were significant (p < 0.05) in 5%, 40%, and 13% of stations, respectively. NGP, VGP, and RGP lengths were significantly reduced in 36%, 7%, and 6% of stations, respectively.
For late rice, median lengths of NGP, VGP, and RGP changed by − 3.0, 0.9, and − 1.1 d/decade. Prolonged lengths of NGP, VGP, and RGP were detected in 22%, 64%, and 38% of the 69 stations, and the changes were significant (p < 0.05) in 9%, 32%, and 4%, respectively. Significantly reduced NGP, VGP, and RGP lengths were identified at 57%, 9%, and 33% of stations, respectively.
For single rice, the median lengths of NGP, VGP, and RGP changed by − 0.5, 1.5, and 1.2 d/decade. Prolonged lengths of NGP, VGP, and RGP were detected in 45%, 68%, and 68% of 88 stations, and the changes were significant (p < 0.05) in 16%, 32%, and 29%, respectively. Lengths of NGP, VGP, and RGP were significantly reduced at 19%, 6%, and 12% of stations, respectively.
3.2 Changes in growth period climatic factors during 1980–2010
Temperature changes associated with climate change and seasonal shifts of growth period were separated using Eq. (1). For the temperature change associated with seasonal shifts of the growth period, our results (shown in Fig. 5) exactly followed the expected pattern, as described for cases A and B in Fig. 1. For growth periods in the spring (case A; i.e., PD− before day 150 of the year) including all nursery growth periods (NGPs) and vegetative growth periods (VGPs) for early rice and single rice, a delay in the starting date moved the growth period into higher temperatures, and consequently resulted in a positive seasonal shift effect. For the nursery growth period (NGP) of early rice, which is the growth period that occurs earliest in the year, a delay in sowing date of 1 day increased NGP mean temperature by 0.11 K (median value). For growth periods in later summer and fall (case B; i.e., PD− after day 210 of the year), including the vegetative growth period (VGP) of late rice, and reproductive growth periods (RGPs) for late rice and single rice, a delay in the starting date shifted the period into lower temperatures, resulting in a negative seasonal shift. For the RGP of late rice, which is the latest growth period in the year, a delay by 1 day decreased RGP mean temperature by 0.15 K (median). The seasonal effects of growth periods in the summer were balanced, with no obvious sign of influence.
After removing the seasonal effects induced by changing phenological dates (PDs), a net climate change effect was revealed (Fig. 6, left). Climate warming was found in NGP, VGP, and RGP at 81%, 77%, and 79% of stations for early rice, 84%, 79%, and 90% of stations for late rice, and 74%, 84%, and 77% of stations for single rice, respectively. The median trends of climate warming influence on the NGP, VGP, and RGP were 0.56, 0.20, and 0.38 K/decade for early rice, 0.41, 0.23, and 0.86 K/decade for late rice, and 0.38, 0.37, and 0.28 for single rice, respectively.
To quantify the bias caused by lack of control for PDs, we repeated the regression analysis to identify the trend in growth period average temperature, but removed PD− from Eq. (1). This collapsed the model to a univariate linear regression with respect to year t (which is a regression used in earlier studies, e.g., Hu et al. 2017). The results are presented in Fig. 6 (right).
Controlling for PDs made a critical difference to the distributions of warming trends. The difference in median values provided evidence of both under- and overestimation. In addition, the interquartile ranges were narrower and the trends were generally more positive in the left panel than those in the right. The median trends of growth period mean temperature in NGP, VGP, and RGP were 0.41, − 0.03, and 0.40 K/decade for early rice, 0.43, 0.29, and 0.91 K/decade for late rice, and 0.30, 0.32, and 0.20 K/decade for single rice, respectively. Therefore, for early rice, warming effects could have been underestimated for NGP and VGP if the negative seasonal effects due to earlier sowing and transplanting dates (Fig. 3) were not removed. The median sizes of underestimates were 0.15 and 0.20 K/decade for NGP and VGP, respectively. For late rice, warming effects may have been overestimated for VGP and RGP if the positive seasonal effects due to earlier transplanting and heading dates (Fig. 3) were not removed. The median sizes of the overestimation were 0.02, 0.05, and 0.08 K/decade for NGP, VGP, and RGP, respectively. For single rice, warming effects were underestimated for all growth periods, due to earlier sowing and transplanting dates, and delayed heading dates (Fig. 3). The median sizes of underestimates were 0.07, 0.09, and 0.07 K/decade for NGP, VGP, and RGP, respectively. The biases were substantial, ranging from 5 to 100% of the magnitude of climate warming during corresponding growth periods.
3.3 Impacts of phenological dates and temperature on growth period length
The results of the regression of the growth period lengths (GPLs) to temperature while controlling for time trend and corresponding starting phenological dates (PD−) are reported in Table 1. The multi-collinearity diagnostics helped excluding results with VIF values > 10, and coefficients of all remaining GPL series were included in the summary statistics. A summary table containing goodness-of-fit and multi-collinearity statistics is provided in the supplementary material (Table S2). The percentage of stations with positive coefficients (%+), positive coefficients that are statistically significant at the 5% level (%+sig.05), negative coefficients that are statistically significant at the 5% level (%−sig.05), and the median coefficients (median) are also presented (Table 1).
Among the factors considered, GPLs were negatively influenced by PD− for more than 90% of stations for late rice and single rice, and for approximately 80% of stations for early rice. Significantly, negative influences were detected for 30–46% of stations for early rice, 46%–62% stations for late rice, and 44%–56% of stations for single rice. Median coefficients indicated that an earlier sowing date by 1 day could prolong NGP by 0.27–0.49 d, and an earlier transplanting by 1 day could prolong VGP by 0.36–0.49 d, while an earlier heading by 1 day could prolong RGP by 0.16–0.42 d.
GPL was mainly negatively influenced by temperature (higher temperature led to shorter GPL), after controlling for PD−. The percentage of positive coefficients was generally below 39%. Regarding the percentage of statistically significant coefficients, substantial higher partition of negative effects than that of positive effects were detected for the NGP of early rice and late rice, VGPs, and RGPs of all rice cropping systems. For the NGP of single rice, there were more balanced percentages of significantly positive and negative effects; however, their values were both 8%. Median coefficients indicated that an increase by 1 K in growth period mean temperature could shorten NGP by approximately 0.42–0.91 d, VGP by 0.82–2.18 d, and RGP by 0.95–1.90 d, depending on cropping types.
Failing to consider shifts in the PDs in the statistical analysis would be predicted to lead to biased estimates of the impacts of climate change on GPLs. To reveal the size of this bias, we performed the regression in Eq. (2) without controlling for PD−. The results are presented in Table S1. We found that for NGP, VGP, and RGP of early rice, and NGP and VGP of single rice, whose PD− were mostly before day 180 of the year, and whose climate trends in temperature have been underestimated, the shortening effects of climate change on GPL were likely to have been overestimated if PD− was not controlled for. The shortening effects were weaker, and the percentages of (significantly) negative coefficients were all smaller. In contrast, for VGP and RGP of late rice, and RGP of single rice, the shortening effects of climate change on GPL may have been underestimated. The impact of climate change was − 2.7 d/K for early rice, − 4.8 d/K for late rice, and − 3.1 d/K for single rice (calculated from Table 1), but − 4.0 d/K for early rice, − 3.0 d/K for late rice, and − 4.2 d/K for single rice without controlling for PD− (calculated from Table S1). Therefore, the overall effect of not controlling for PD is an overestimation of the impact of climate change on GPLs for early rice and single rice, but an underestimation for late rice.
The effects of cultivar shifts can be revealed by observing the time trend in GPLs after removing the influence of PDs and climatic factors, or equivalently, the coefficients of the time index (t) in Eq. (2). Our results provide strong evidence for the use of shortened nursery growth period (NGP), 0.5–1.8 d/decade, but prolonged vegetative growth period (VGP) cultivars for all rice cropping systems, and prolonged reproductive growth period (RGP) cultivars for single rice. Our results also weakly supported that cultivar change had prolonged RGPs of early and late rice in terms of the median values.
4 Discussion
4.1 Changes in rice phenology
Our analyses of data from 892 rice phenology series from 157 agrometeorological stations showed that there were changes to earlier dates within the year for almost all phenological dates (PDs), except for the sowing dates of late rice, and heading/maturity dates of single rice, in terms of median values. The direction of changes in transplanting, heading, and maturity dates detected in our study was consistent with published findings (Tao et al. 2013; Zhao et al. 2016; Hu et al. 2017), although our study differs in the number of stations and the years considered. Moreover, the pattern of change indicated by our results agrees with the findings of Zhao et al. (2016). For both early and late rice, the transplanting date exhibited the largest magnitude of change to earlier dates, while the heading date had the smallest. This differs from the findings of Tao et al. (2013), in which heading dates showed the largest magnitude of change to earlier dates for both early and late rice. The difference in the magnitude of change in PDs led to completely different results regarding changes in GPL (see below). Earlier studies have rarely analyzed changes in sowing date in their analyses, primarily because of the strong human influence on sowing decisions (Estrella et al. 2007).
Nursery growth period (NGP) was reduced and vegetative growth periods (VGP) prolonged for all rice cropping systems in this study, while reproductive growth period (RGP) was almost unchanged for early rice, shortened for late rice, and prolonged for single rice. The majority of our findings are in agreement with the existing literature. For example, a shortened seedbed duration (equivalent to our NGP) was reported by Zhang et al. (2013), and the prolonged VGPs for early rice and late rice are consistent with the findings of Zhao et al. (2016). However, our results disagree with those of Tao et al. (2013), because of the difference in the magnitudes of shifts in transplanting and heading dates, as mentioned above. Our results are consistent with the findings of Hu et al. (2017), in that the growth periods from emerging to heading (comparable to our results for NGP and VGP), and RGPs were prolonged for early rice and single rice, but shortened for late rice.
4.2 Impact of climate change, cultivar shifts, and phenological date shifts on GPL
Our results regarding the shortening effect of temperature rise on growth period length (GPL) support those of previous reports (Sadras and Monzon 2006; Liu et al. 2012; Tao et al. 2012; Zhang et al. 2013); however, our data indicate less overall impact on GPL than previous statistical analyses have suggested. After controlling for phenological dates (PDs) in the regression to remove the effects of seasonal shift, only 80% of investigated GPL series exhibited negative relationships between GPL and temperature, of which 28% were statistically significant. Correspondingly, in Zhang et al. (2013), negative and significantly negative effects of temperature were reported in over 95% and 61.9% of the data series, respectively, and negative effects were found in over 80% of the data series in Tao et al. (2013). Regarding the magnitude of impacts, the estimated temperature effects were − 3.6 d/K for early rice, − 3.8 d/K for late rice, and − 5.5 d/K for single rice (Zhang et al. 2013), consistent with our results without controlling for PD−s.
Regarding the effects of cultivar shifts, our results are consistent with those of previous studies. For instance, statistical analyses (Tao et al. 2013; Zhang et al. 2014) indicated that cultivars of longer growth duration VGP were adopted for single rice, and cultivars with longer growth durations of RGP were adopted for single and early rice. Analysis based on rice growth models also indicated that longer VGP and RGP cultivars were used for early and single rice, but not for late rice (Hu et al. 2017). Our result for single rice was also supported by the crop simulation analysis (Liu et al. 2012; Liu et al. 2013) that cultivar change led to a significant increase in actual thermal time requirement to complete the whole rice growing period for single rice. Regarding the shortened RGP of late rice, our model tended to attribute the change to heading date variation and climate change, but not to cultivar shifts. Tao et al. (2013) discussed that the avoidance of cold damage in the fall may explain the popularity of short-duration RGP cultivars.
In our results, GPL changes exhibited the strongest association with PDs, consistent with the findings of Zhao et al. (2016). Overall, 91% of investigated GPL series (for all GPLs and rice cropping systems) exhibited negative associations between GPLs and their corresponding starting PDs, and 48% were significantly negatively associated. Therefore, assuming all other conditions are the same, later starting PDs led to shorter GPLs. As PDs are highly mutually correlated (Zhao et al. 2016), the effect of PDs on GPLs can further be interpreted as the effect of planting dates (sowing dates)Footnote 1 or transplanting dates, and lead to similar conclusions as those based on a simulation of growing degree days (Hu et al. 2017; Liu et al. 2018).
In summary, our results indicate that climate warming has shortened rice GPLs, which may have harmed yield production (Yao et al. 2007; Zhang et al. 2013). Nevertheless, the warming climate enables growth earlier in the spring and later in the fall (Jeong et al. 2011; Cong et al. 2013; Ai et al. 2014). As rice sowing dates are restricted by minimum temperature requirements,Footnote 2 earlier spring and delayed fall can expand the time window suitable for rice planting within a specific year, providing farmers with more flexibility for their planting decisions, since the temperature constraint is relaxed. A prolonged time window suitable for rice cultivation further allows the use of longer vegetative growth period cultivars, which could make the full period suitable for rice cultivation and may alleviate the potential damage attributable to a shortened growth period resulting from higher temperatures. This provides further evidence of active adaptation in rice production practices in response to climate change in China (Liu et al. 2012; Hu et al. 2017).
4.3 Limitations
The study has several limitations. First, the model used is still based on statistical analysis, which has hampered us for more insightful discussion. For instance, the impact of cultivar change cannot be modeled explicitly with the available data due to the lack of yearly cultivar change data, particularly at the farm level. Because of this, our results on late rice disagree with earlier reports. To further investigate the contributions of the climatic and technological factors, agronomic analysis, and/or process-based crop, simulation models may provide more insights.
Second, we conclude that sowing and transplanting dates are critically influential on growth period lengths, with even stronger effects than climate change; however, it does not lead to a complete attribution of phenology changes to human and climate factors. Sowing and transplanting dates are subject to both climate constraints and human planting decisions; the degree of influence of these factors on sowing and transplanting dates remains controversial in the literature. For example, an association between temperature before planting and planting date was reported by Tao et al. (2006), while others have suggested that the relationship is weak (Zhao et al. 2016). Zhang et al. (2013) suggested that the association is stronger if the sowing dates are earlier in the year; therefore, further analyses will be required to distinguish the relative contribution of human decisions and climatic factors in planting dates, to finally reveal a full picture of climate, cultivar shifts, and planting decision impacts on growth period lengths.
5 Conclusion
In conclusion, changes in rice phenology include not only changes in the length of growth periods, but also the phenological dates and corresponding seasonal shifts of growth periods in a year. Consequently, changes in climatic factors, particularly temperature, comprise both climate change effects and seasonal effects due to growth period shifts. We distinguished the effects of seasonal shifts by controlling for the start dates of each growth period in the regression model. We found that the true magnitude of climate warming for early rice and single rice has been underestimated, and that for late rice it has been overestimated in previous studies. Correspondingly, the net climate change impact on growth period length after removing seasonal shift effects was weaker for early rice and single rice, but stronger for late rice. Phenological date changes were found to be most closely associated with alterations in growth period length changes, while the linkage to climate change was weaker. As phenological date changes are subject to strong human influence, in terms of planting decisions, together with inferred changes in cultivars, our results suggest active adaptation to climate change in rice cultivation practices in China. Although climate change results in more heat stress on rice growth, earlier spring and delayed autumn growth of rice allow farmers to have greater flexibility and to determine the appropriate timing for rice cultivation.
Notes
Our observation implies that GPLs are strongly linked to transplanting and sowing dates. This is supported by our data generated by replacing all PD−s in Eq. (2) with sowing or transplanting dates. After such replacement, the overall percentage with negative relationships with GPL was 71.6%, and the overall percentage with significantly negative effects was 23.3%, respectively; stronger than the effects of climate warming.
A 5-day moving average temperature, with a threshold of 10 °C, is believed to be the critical temperature for thermophiles to start growth, and is therefore used as an indicator for paddy rice research and field management (Jiang et al. 2011)
References
Ai Z, Guo X, Liu W et al (2014) Analysis on possible influences of agricultural climate resources change on double-season rice production. J Nat Resour 29:2089–2102. https://doi.org/10.11849/zrzyxb.2014.12.010
Asseng S, Foster I, Turner NC (2011) The impact of temperature variability on wheat yields. Glob Chang Biol 17:997–1012. https://doi.org/10.1111/j.1365-2486.2010.02262.x
Brown ME, de Beurs KM, Marshall M (2012) Global phenological response to climate change in crop areas using satellite remote sensing of vegetation, humidity and temperature over 26 years. Remote Sens Environ 126:174–183. https://doi.org/10.1016/j.rse.2012.08.009
Chen Y, Yang K, He J et al (2011) Improving land surface temperature modeling for dry land of China. J Geophys Res Atmos 116. https://doi.org/10.1029/2011JD015921
Cong N, Wang T, Nan H et al (2013) Changes in satellite-derived spring vegetation green-up date and its linkage to climate in China from 1982 to 2010: a multimethod analysis. Glob Chang Biol 19:881–891. https://doi.org/10.1111/gcb.12077
Estrella N, Sparks TH, Menzel A (2007) Trends and temperature response in the phenology of crops in Germany. Glob Chang Biol 13:1737–1747. https://doi.org/10.1111/j.1365-2486.2007.01374.x
Gross BL, Zhao Z (2014) Archaeological and genetic insights into the origins of domesticated rice. Proc Natl Acad Sci 111:6190–6197. https://doi.org/10.1073/pnas.1308942110
He J, Yang K (2011) China meteorological forcing dataset. In: Cold and arid regions science data center at Lanzhou. https://doi.org/10.3972/westdc.002.2014.db
Hu X, Huang Y, Sun W, Yu L (2017) Shifts in cultivar and planting date have regulated rice growth duration under climate warming in China since the early 1980s. Agric For Meteorol 247:34–41. https://doi.org/10.1016/j.agrformet.2017.07.014
Jeong SJ, Ho CH, Gim HJ, Brown ME (2011) Phenology shifts at start vs. end of growing season in temperate vegetation over the Northern Hemisphere for the period 1982-2008. Glob Chang Biol 17:2385–2399. https://doi.org/10.1111/j.1365-2486.2011.02397.x
Jiang X, Tang L, Liu X et al (2011) Spatial and temporal characteristics of rice production climatic resources in main growing regions of China. Trans Chinese Soceity Agric Eng 27:238–245
Liu L, Wang E, Zhu Y, Tang L (2012) Contrasting effects of warming and autonomous breeding on single-rice productivity in China. Agric Ecosyst Environ 149:20–29. https://doi.org/10.1016/j.agee.2011.12.008
Liu L, Wang E, Zhu Y, Tang L (2013) Quantifying three-decade changes of single rice cultivars in China using crop modeling. Field Crop Res 149:84–94. https://doi.org/10.1016/j.agee.2011.12.008
Liu Y, Chen Q, Ge Q et al (2018) Modelling the impacts of climate change and crop management on phenological trends of spring and winter wheat in China. Agric For Meteorol 248:518–526. https://doi.org/10.1016/j.agrformet.2017.09.008
National Bureau of Statistics of China (NBSC) (2016) China statistical yearbook 2016. http://www.stats.gov.cn/tjsj/ndsj/2016/indexeh.htm.. Accessed 09 Nov 2017
Nie L, Peng S (2017) Rice production in China. In: Chauhan B, Jabran K, Mahajan G (eds) Rice production worldwide. Springer, Cham
Piao S, Fang J, Zhou L et al (2006) Variations in satellite-derived phenology in China’s temperate vegetation. Glob Chang Biol 12:672–685. https://doi.org/10.1111/j.1365-2486.2006.01123.x
Qiu B, Lu D, Tang Z et al (2017) Mapping cropping intensity trends in China during 1982–2013. Appl Geogr 79:212–222. https://doi.org/10.1016/j.apgeog.2017.01.001
Richardson AD, Keenan TF, Migliavacca M et al (2013) Climate change, phenology, and phenological control of vegetation feedbacks to the climate system. Agric For Meteorol 169:156–173. https://doi.org/10.1016/j.agrformet.2012.09.012
Sadras VO, Monzon JP (2006) Modelled wheat phenology captures rising temperature trends: shortened time to flowering and maturity in Australia and Argentina. F Crop Res 99:136–146. https://doi.org/10.1016/j.fcr.2006.04.003
Shimono H (2011) Earlier rice phenology as a result of climate change can increase the risk of cold damage during reproductive growth in northern Japan. Agric Ecosyst Environ 144:201–207. https://doi.org/10.1016/j.agee.2011.08.006
Tao F, Yokozawa M, Xu Y et al (2006) Climate changes and trends in phenology and yields of field crops in China, 1981–2000. Agric For Meteorol 138:82–92. https://doi.org/10.1016/j.agrformet.2006.03.014
Tao F, Zhang S, Zhang Z (2012) Spatiotemporal changes of wheat phenology in China under the effects of temperature, day length and cultivar thermal characteristics. Eur J Agron 43:201–212. https://doi.org/10.1016/j.eja.2012.07.005
Tao F, Zhang Z, Shi W et al (2013) Single rice growth period was prolonged by cultivars shifts, but yield was damaged by climate change during 1981-2009 in China, and late rice was just opposite. Glob Chang Biol 19:3200–3209. https://doi.org/10.1111/gcb.12250
Wu C, Gonsamo A, Chen JM et al (2012) Interannual and spatial impacts of phenological transitions, growing season length, and spring and autumn temperatures on carbon sequestration: a North America flux data synthesis. Glob Planet Change 92–93:179–190. https://doi.org/10.1016/j.gloplacha.2012.05.021
Yao F, Xu Y, Lin E et al (2007) Assessing the impacts of climate change on rice yields in the main rice areas of China. Clim Chang 80:395–409. https://doi.org/10.1007/s10584-006-9122-6
Zhang X, Friedl MA, Schaaf CB, Strahler AH (2004) Climate controls on vegetation phenological patterns in northern mid- and high latitudes inferred from MODIS data. Glob Chang Biol 10:1133–1145. https://doi.org/10.1111/j.1529-8817.2003.00784.x
Zhang T, Huang Y, Yang X (2013) Climate warming over the past three decades has shortened rice growth duration in China and cultivar shifts have further accelerated the process for late rice. Glob Chang Biol 19:563–570. https://doi.org/10.1111/gcb.12057
Zhang S, Tao F, Zhang Z (2014) Rice reproductive growth duration increased despite of negative impacts of climate warming across China during 1981-2009. Eur J Agron 54:70–83. https://doi.org/10.1016/j.eja.2013.12.001
Zhao H, Fu YH, Wang X et al (2016) Timing of rice maturity in China is affected more by transplanting date than by climate change. Agric For Meteorol 216:215–220. https://doi.org/10.1016/j.agrformet.2015.11.001
Funding
This study was supported by the National Key R&D Program of China (No. 2016YFA0602404), the project of State Key Laboratory of Earth Surface Processes and Resource Ecology, and the Fund for Creative Research Groups of National Natural Science Foundation of China (No. 41621061).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 31 kb)
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Ye, T., Zong, S., Kleidon, A. et al. Impacts of climate warming, cultivar shifts, and phenological dates on rice growth period length in China after correction for seasonal shift effects. Climatic Change 155, 127–143 (2019). https://doi.org/10.1007/s10584-019-02450-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10584-019-02450-5