Abstract
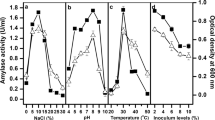
This study investigated the coproduction of alkaline amylase and lipopeptides by Bacillus methylotrophicus DCS1 strain, as well as their biochemical characterisation. The best production of both amylase and biosurfactant was obtained when potato starch (10 g/L) and glutamic acid (5 g/L) were used as carbon and nitrogen sources, respectively. The bacterial strain was incubated for 48 h at 25 °C and 150 rpm. This strain produced a unique amylase as showed by zymography technique. The optima pH and temperature were 60–65 °C and 8.0, respectively. Amylase activity was partially inhibited by EDTA (5 mM). The main hydrolysis products of potato starch were maltose and maltotriose. The alkaline amylase showed excellent stability and compatibility with various solid and liquid detergents. Furthermore, the biosurfactant, produced simultaneously with alkaline amylase, demonstrated high stability at different ranges of salinity, pH, and temperature. Considering its promising properties, B. methylotrophicus DCS1 crude extract containing both biosurfactants and amylase activity may be considered as a potential candidate for future use in detergent processing industries and environmental remediation processes.






Similar content being viewed by others
References
Abouseoud M, Yataghene A, Amrane A, Maachi R (2008) Biosurfactant production by free and alginate entrapped cells of Pseudomonas fluorescens. J Ind Microbiol Biotechnol 35:1303–1308
Arikan B (2008) Highly thermostable, thermophilic, alkaline, SDS and chelator resistant amylase from a thermophilic Bacillus sp. isolate A3-15. Biores Technol 99:3071–3076
Aygan A, Arikan B, Korkmaz H, Dinçer S, Çolak Ö (2008) Highly thermostable and alkaline α-amylase from a halotolerant alkaliphilic Bacillus sp. AB68. Braz J Microbiol 39:547–553
Behel A, Singh MK, Sharma MK, Puri P, Batra N (2006) Characterization of alkaline α-amylase from Bacillus sp. AB 04. Int J Agric Biol 8:80–83
Bhange K, Chaturvedi V, Bhatt R (2016) Simultaneous production of detergent stable keratinolytic protease, amylase and biosurfactant by Bacillus subtilis PF1 using agroindustrial waste. Biotechnol Rep 10:94–104
Bognolo G (1999) Biosurfactants as emulsifying agents for hydrocarbons. Colloids Surf A Physicochem Eng Aspects 152:41–52
Burhan A, Nisa U, Gokhan C, Omer C, Ashabil A, Osman G (2003) Enzymatic properties of a novel thermostable, thermophilic, alkaline and chelator resistant amylase from an alkaliphilic Bacillus sp. isolate ANT-6. Process Biochem 38:1397–1403
Cagri-Mehmetoglu A, Kusakli S, Venter MV (2012) Production of polysaccharide and surfactin by Bacillus subtilis ATCC 6633 using rehydrated whey powder as the fermentation medium. J Dairy Sci 95:3643–3649
Chen YC, Chiang TJ, Liang TW, Wang IL, Wang SL (2012) Reclamation of squid pen by Bacillus licheniformis TKU004 for the production of thermally stable and antimicrobial biosurfactant. Biocatal Agric Biotechnol 1:62–69
Chtioui O, Dimitrov K, Gancel F, Nikov I (2010) Biosurfactants production by immobilized cells of Bacillus subtilis ATCC 21332 and their recovery by pertraction. Process Biochem 45:1795–1799
Colla LM, Rizzardi J, Pinto MH, Reinehr CO, Bertolin TE, Costa JAV (2010) Simultaneous production of lipases and biosurfactants by submerged and solid-state bioprocesses. Biores Technol 101:8308–8314
Dahiya P, Rathi Amity RB (2015) Characterization and application of alkaline α-amylase from Bacillus licheniformis MTCC1483 as a detergent additive. Int Food Res J 22:1293–1297
De Carvalho RV, Côrrea TLR, da Silva JCM, de Oliveira Mansur LRC, Martins MLL (2008) Properties of an amylase from thermophilic Bacillus sp. Braz J Microbiol 39:102–107
De Oliveira DWF, França ÍWL, Félix AKN, Martins JJL, Giro MEA, Melo VMM, Gonçalves LRB (2013) Kinetic study of biosurfactant production by Bacillus subtilis LAMI005 grown in clarified cashew apple juice. Colloids Surf B 101:34–43
De Souza PM, de Oliveira Magalhães P (2010) Application of microbial α-amylase in industry: a review. Braz J Microbiol 41:850–861
Desai JD, Banat IM (1997) Microbial production of surfactants and their commercial potential. Microbiol Mol Rev 61:47–64
Deutch CE (2002) Characterization of a salt-tolerant extracellular α-amylase from Bacillus dipsosauri. Lett Appl Microbiol 35:78–84
Fickers P, Leclère V, Guez JS, Béchet M, Coucheney F, Joris B, Jacques P (2008) Temperature dependence of mycosubtilin homologue production in Bacillus subtilis ATCC6633. Res Microbiol 159:449–457
Ghorbel RE, Maktouf S, Massoud EB, Bejar S, Chaabouni SE (2009) New thermostable amylase from Bacillus cohnii US147 with a broad pH applicability. Appl Biochem Biotechnol 157:50–60
Guez JS, Müller CH, Danze PM, Büchs J, Jacques P (2008) Respiration activity monitoring system (RAMOS), an efficient tool to study the influence of the oxygen transfer rate on the synthesis of lipopeptide by Bacillus subtilis ATCC6633. J Biotechnol 134:121–126
Hmidet N, Bayoudh A, Berrin JG, Kanoun S, Juge N, Nasri M (2008) Purification and biochemical characterization of a novel α-amylase from Bacillus licheniformis NH1. Cloning, nucleotide sequence and expression of amyN gene in E. coli. Proc Biochem 43:499–510
Hmidet N, Maalej H, Haddar A, Nasri M (2010) A novel α-amylase from Bacillus mojavensis A21: purification and biochemical characterization. Appl Biochem Biotechnol 162:1018–1030
Hmidet N, Ben Ayed H, Jacques P, Nasri M (2017) Enhancement of Surfactin and Fengycin Production by Bacillus mojavensis A21: application for Diesel Biodegradation. BioMed Res Inter 2017:8
Jemil N, Ben Ayed H, Hmidet N, Nasri M (2016) Characterization and properties of biosurfactants produced by a newly isolated strain Bacillus methylotrophicus DCS1 and their applications in enhancing solubility of hydrocarbon. World J Microbiol Biotechnol 32:175
Jemil N, Manresa A, Rabanal F, Hmidet N, Nasri M (2017a) Structural characterization and identification of cyclic lipopeptides produced by Bacillus methylotrophicus DCS1 strain. J Chromatogr B 1060:374–386
Jemil N, Ben Ayed H, Nasri M, Hmidet N (2017b) Antioxidant and antimicrobial activities of lipopeptides produced by a newly isolated strain Bacillus methylotrophicus DCS1 and their application in biofilm control. BMC Microbiol 17:144
Kavuthodi B, Thomas SK, Sebastian D (2015) Co-production of Pectinase and Biosurfactant by the Newly Isolated Strain Bacillus subtilis BKDS1. Br Microbiol Res J 10:1–12
Kiran KK, Chandra TS (2008) Production of surfactant and detergent-stable, halophilic, and alkalitolerant alpha-amylase by a moderately halophilic Bacillus sp. strain TSCVKK. Appl Microbiol Biotechnol 77:1023–1031
Kuyukina MS, Ivshina IB, Makarov SO, Litvinenko LV, Cunningham CJ, Philp CJ (2005) Effect of biosurfactants on crude oil desorption and mobilization in a soil system. Environ Int 31:155–161
Landy M, Warren GH, RosenmanM SB, Colio LG (1948) Bacillomycin: an antibiotic from Bacillus subtilis active against pathogenic fungi. Exp Biol Med 67:539–541
Luna JM, Rufino RD, Campos-Takakia GM, Sarubbo LA (2012) Properties of the biosurfactant produced by Candida sphaerica cultivated in low-cost substrates. Chem Eng Trans 27:67–72
Machius M, Declerck N, Huber R, Wiegand G (1998) Activation of Bacillus licheniformis alpha-amylase through a disorder → order transition of the substrate-binding site mediated by a calcium–sodium–calcium metal triad. Structure 6:281–292
Makkar RS, Cameotra SS, Banat IM (2011) Advances in utilization of renewable substrates for biosurfactant production. AMB Express 1:5
Marchant R, Banat IM (2012) Microbial biosurfactants: challenges and opportunities for future exploitation. Trends Biotechnol 30:558–565
Miller GL (1959) Use of dinitrosalycilic acid reagent for determination of reducing sugars. Anal Chem 31:426–428
Miller JH (1972) Experiments in moleculer genetics. Cold Spring Harbor Laboratory Press, New York
Mukherjee AK, Borah M, Rai SK (2009) To study the influence of different components of fermentable substrates on induction of extracellular a-amylase synthesis by Bacillus subtilis DM-03 in solid-state fermentation and exploration of feasibility for inclusion of α-amylase in laundry detergent formulations. Biochem Eng J 43:149–156
Najafi MF, Deobagkar D, Deobagkar D (2005) Purification and characterization of an extracellular α-amylase from Bacillus subtilis AX20. Prot Exp Purif 41:349–354
Neves LCMD, Miyamura TTMO, Kobayashi MJ, Penna TCV, Converti A (2007) Production of biosurfactant by a genetically-modified strain of Bacillus subtilis expressing green fluorescent protein. Ann Microbiol 57:377–381
Nitschke M, Costa SGAO (2007) Biosurfactants in food industry. Trends Food Sci Technol 18:252–259
Nitschke M, Pastore GM (2004) Biosurfactant production by Bacillus subtilis using cassava processing effluent. Appl Biochem Biotechnol 112:163–172
Niyonzima FN, More SS (2014) Detergent-Compatible Bacterial Amylases. Appl Biochem Biotechnol 174:1215–1232
Nusrat A, Rahman SR (2007) Comparative studies on the production of extracellular amylase by three mesophilic Bacillus isolates. Bangladesh J Microbiol 24:129–132
Ohno A, Ano T, Shoda M (1995) Production of a lipopeptide antibiotic, surfactin, by recombinant Bacillus subtilis in solid state fermentation. Biotechnol Bioeng 47:209–214
Piedrahίta-Aguirre CA, Bastos RG, Carvalho AL, Monte Alegre R (2014) The influence of process parameters in production of lipopeptide iturin A using aerated packed bed bioreactors in solid-state fermentation. Bioprocess Biosyst Eng 37:1569–1576
Ramnani P, Kumar SS, Gupta R (2005) Concomitant production and downstream processing of alkaline protease and biosurfactant from Bacillus licheniformis RG1: bioformulation as detergent additive. Process Biochem 40:3352–3359
Rodrigues LR, Banat IM, Teixeira JA, Oliveira R (2006) Biosurfactants: potential applications in medicine. J Antimicrob Chemother 57:609–618
Roohi R, Kuddus M, Saima S (2013) Cold-active detergent-stable extracellular α-amylase from Bacillus cereus GA6: biochemical characteristics and its perspectives in laundry detergent formulation. J Biochemical Technol 4:636–644
Roy JK, Rai SK, Mukherjee AK (2012) Characterization and application of a detergent-stable alkaline alpha-amylase from Bacillus subtilis strain AS-S01a. Int J Biol Macromol 50:219–229
Saxena KR, Dutt K, Agarwal L, Nayyar P (2007) A highly thermostable and alkaline amylase from a Bacillus sp. PN5. Biores Technol 98:260–265
Schallmey M, Singh A, Ward OP (2004) Developments in the use of Bacillus species for industrial production. Can J Microbiol 50:1–17
Sobrinho HB, Luna JM, Rufino RD, Porto ALF, Sarubbo LA (2013) Biosurfactants: classification, properties and environmental applications. In: Govil JN (ed) Recent developments in biotechnology. Studium Press LLC, Houston, pp 1–29
Wang Q, Chen S, Zhang J, Sun M, Liu Z, Ziniu Y (2008) Co-producing lipopeptides and poly-γ-glutamic acid by solid-state fermentation of Bacillus subtilis using soybean and sweet potato residues and its biocontrol and fertilizer synergistic effects. Bioresour Technol 99:3318–3323
Xie F, Quan S, Liu D, Ma H, Li F, Zhou F, Chen G (2014) Purification and characterization of a novel α-amylase from a newly isolated Bacillus methylotrophicus strain P11-2. Process Biochem 49:47–53
Yao D, Ji Z, Wang C, Qi G, Zhang L, Ma X, Chen S (2012) Co-producing iturin A and poly-γ-glutamic acid from rapeseed meal under solid state fermentation by the newly isolated Bacillus subtilis strain 3–10. World J Microbiol Biotechnol 28:985–991
Zhang J, Xue Q, Gao H, Lai H, Wang P (2016) Production of lipopeptide biosurfactants by Bacillus atrophaeus 5-2a and their potential use in microbial enhanced oil recovery. Microb Cell Fact 15:168
Acknowledgements
This work was funded by «Ministry of Higher Education and Scientific Research-Tunisia».
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hmidet, N., Jemil, N. & Nasri, M. Simultaneous production of alkaline amylase and biosurfactant by Bacillus methylotrophicus DCS1: application as detergent additive. Biodegradation 30, 247–258 (2019). https://doi.org/10.1007/s10532-018-9847-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10532-018-9847-8