Abstract
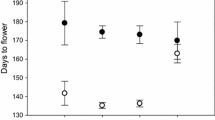
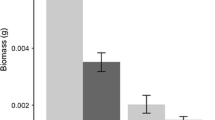
Congeneric species with the same native and non-native ranges, but exhibiting different invasiveness, provide opportunities to assess the relative importance of factors contributing to successful invasions. For example, comparing the competitive ability of such congeners against other species from the native and non-native regions of invasive species can provide insight into the role of evolutionary experience with different competitors. We selected two congeneric Centaurea species with overlapping native and non-native ranges but with strikingly different invasive success, Centaurea solstitialis and C. calcitrapa, and conducted experiments with populations from each region using a suite of different native grass species from each region. When grown with Spanish grass species, competitive responses were 32% stronger for C. calcitrapa and 30% stronger for C. solstitialis, than when in competition with grasses native to California. Centaurea solstitialis from California had 66% higher competitive effects on grasses, all species considered together, than Spanish C. solstitialis; whereas the competitive effects of C. calcitrapa on grasses were not significantly different between C. calcitrapa ranges. Our results are consistent with the prediction that the evolution of increased competitive ability (EICA) might contribute to the devastating success of C. solstitialis in California, and that its absence might contribute to the modest naturalization of the non-invasive C. calcitrapa. Escape from the strong competitive effects and responses of grasses from Spain may also contribute to invasive success in California. Our results suggest that studies of EICA should be based on competitive tests, since biomass alone is not always a good predictor of competitive ability.






Similar content being viewed by others
References
Andonian K, Hierro JL, Khetsuriani L et al (2012) Geographic mosaics of plant–soil microbe interactions in a global plant invasion. J Biogeogr 39:600–608. https://doi.org/10.1111/j.1365-2699.2011.02629.x
Armas C, Ordiales R, Pugnaire FI (2004) Measuring plant interactions: a new comparative index. Ecology 85:2682–2686. https://doi.org/10.1890/03-0650
Aschehoug ET, Metlen KL, Callaway RM, Newcombe G (2012) Fungal endophytes directly increase the competitive effects of an invasive forb. Ecology 93:3–8
Aschehoug ET, Brooker R, Atwater DZ et al (2016) The mechanisms and consequences of interspecific competition among plants. Annu Rev Ecol Evol Syst 47:263–281. https://doi.org/10.1146/annurev-ecolsys-121415-032123
Barker BS, Andonian K, Swope SM et al (2017) Population genomic analyses reveal a history of range expansion and trait evolution across the native and invaded range of yellow starthistle (Centaurea solstitialis). Mol Ecol 26:1131–1147. https://doi.org/10.1111/mec.13998
Bartolomé C (2005) Los tipos de habitat de interés comunitario de España. Ministerio de Medio Ambiente. Dirección General para la Biodiversidad, Madrid
Bartolome JW, Barry WJ, Griggs T, Hopkinson P (2007) Valley grassland. Terr Vegetarian, California, p 432
Blossey B, Nötzold R (1995) Evolution of increased competitive ability in invasive nonindigenous plants: a hypothesis. J Ecol 83:887–889
Bossdorf O, Auge H, Lafuma L et al (2005) Phenotypic and genetic differentiation between native and introduced plant populations. Oecologia 144:1–11. https://doi.org/10.1007/s00442-005-0070-z
Callaway RM, Aschehoug ET (2000) Invasive plants versus their new and old neighbors: a mechanism for exotic invasion. Science 290:521–523. https://doi.org/10.1126/science.290.5491.521
Callaway RM, Kim J, Mahall BE (2006) Defoliation of Centaurea solstitialis stimulates compensatory growth and intensifies negative effects on neighbors. Biol Invasions 8:1389–1397. https://doi.org/10.1007/s10530-006-0003-8
Callaway RM, Waller LP, Diaconu A et al (2011) Escape from competition: neighbors reduce Centaurea stoebe performance at home but not away. Ecology 92:2208–2213
Colautti RI, MacIsaac HI (2004) A neutral terminology to define “invasive” species. Divers Distrib 10:135–141
Dlugosch KM, Cang FA, Barker BS et al (2015) Evolution of invasiveness through increased resource use in a vacant niche. Nat Plants 1:1–5. https://doi.org/10.1038/nplants.2015.66
Eagle AJ, Eiswerth ME, Johnson WS et al (2007) Costs and losses imposed on California ranchers by yellow starthistle. Rangel Ecol Manag 60:369–377
Eriksen RL, Hierro JL, Eren Ö et al (2014) Dispersal pathways and genetic differentiation among worldwide populations of the invasive weed Centaurea solstitialis L. (Asteraceae). PLoS ONE 9:e114786. https://doi.org/10.1371/journal.pone.0114786
Felker-Quinn E, Schweitzer JA, Bailey JK (2013) Meta-analysis reveals evolution in invasive plant species but little support for Evolution of Increased Competitive Ability (EICA). Ecol Evol 3:739–751. https://doi.org/10.1002/ece3.488
Filipe JC, Montesinos D (2016a) Invasive and non-invasive congeneric Centaurea show contrasting patterns of herbivory by snails. Plant Ecol Evol 149:228–232. https://doi.org/10.5091/plecevo.2016.1231
Filipe JC, Montesinos D (2016b) Inter-regional hybrids of native and non-native Centaurea sulphurea inherit increased competitive ability from the non-natives. Plant Ecol Divers 9:381–386. https://doi.org/10.1080/17550874.2016.1261950
Funk JL, Standish RJ, Stock WD, Valladares F (2015) Plant functional traits of dominant native and invasive species in Mediterranean-climate ecosystems. Ecology 746:23. https://doi.org/10.1890/15-0974.1
García Y, Callaway RM, Diaconu A, Montesinos D (2013) Invasive and non-invasive congeners show similar trait shifts between their same native and non-native ranges. PLoS ONE 8:e82281. https://doi.org/10.1371/journal.pone.0082281
Garcia-Jacas N, Uysal T, Romashchenko K et al (2006) Centaurea revisited: a molecular survey of the Jacea group. Ann Bot 98:741–753
Garren JM, Strauss SY (2009) Population-level compensation by an invasive thistle thwarts biological control from seed predators. Ecol Appl 19:709–721
Graebner RC, Callaway RM, Montesinos D (2012) Invasive species grows faster, competes better, and shows greater evolution toward increased seed size and growth than exotic non-invasive congeners. Plant Ecol 213:545–553. https://doi.org/10.1007/s11258-012-0020-x
He W-M, Feng Y, Ridenour WM et al (2009) Novel weapons and invasion: biogeographic differences in the competitive effects of Centaurea maculosa and its root exudate-catechin. Oecologia 159:803–815. https://doi.org/10.1007/s00442-008-1234-4
Hierro JL, Villarreal D, Eren O et al (2006) Disturbance facilitates invasion: the effects are stronger abroad than at home. Am Nat 168:144–156. https://doi.org/10.1086/505767
Hierro JL, Khetsuriani L, Andonian K et al (2016) The importance of factors controlling species abundance and distribution varies in native and non-native ranges. Ecography 39:1–12. https://doi.org/10.1111/ecog.02633
Hothorn T, Bretz F, Westfall P (2008) Simultaneous inference in general parametric models. Biom J 50:346–363
Joshi J, Vrieling K (2005) The enemy release and EICA hypothesis revisited: incorporating the fundamental difference between specialist and generalist herbivores. Ecol Lett 8:704–714
Laird NNM, Ware JJH (1982) Random-effects models for longitudinal data. Biometrics 38:963–974
Maddox DM, Mayfield A, Poritz NH (1985) Distribution of yellow starthistle (Centaurea solstitialis) and russian knapweed (Centaurea repens). Weed Sci 33:315–327
Maron J, Marler M (2007) Native plant diversity resists invasion at both low and high resource levels. Ecology 88:2651–2661
Montesinos D, Callaway RM (2017) Inter-regional hybrids of native and invasive Centaurea solstitialis display intermediate competitive ability. Ecography 40:801–802. https://doi.org/10.1111/ecog.02653
Montesinos D, Santiago G, Callaway RM (2012) Neo-allopatry and rapid reproductive isolation. Am Nat 180:529–533. https://doi.org/10.1086/667585
Munshaw MG, Lortie CJ (2010) Back to the basics: using density series to test regulation versus limitation for invasive plants. Plant Ecol 211:1–5. https://doi.org/10.1007/s11258-010-9764-3
Muth NZ, Pigliucci M (2006) Traits of invasives reconsidered: phenotypic comparisons of introduced invasive and introduced noninvasive plant species within two closely related clades. Am J Bot 93:188–196. https://doi.org/10.3732/ajb.93.2.188
Ni G-Y, Schaffner U, Peng S-L, Callaway RM (2010) Acroptilon repens, an Asian invader, has stronger competitive effects on species from America than species from its native range. Biol Invasions 12:3653–3663. https://doi.org/10.1007/s10530-010-9759-y
Oduor AMO, van Kleunen M, Stift M (2017) In the presence of specialist root and shoot herbivory, invasive-range Brassica nigra populations have stronger competitive effects than native-range populations. J Ecol 105:1679–1686. https://doi.org/10.1111/1365-2745.12779
Pinheiro J, Bates D, DebRoy S, Sarkar D, R Core Team (2018) Linear and nonlinear mixed effects models. R package version 3.1-117. https://CRAN.R-project.org/package=nlme. Accessed 05 Aug 2018
Pitcairn MJ, Young JA, Clements CD, Balciunas JOE (2002) Purple starthistle (Centaurea calcitrapa) seed germination. Weed Technol 16:452–456
Qin B, Lau JA, Kopshever J et al (2007) No evidence for root-mediated allelopathy in Centaurea solstitialis, a species in a commonly allelopathic genus. Biol Invasions 9:897–907. https://doi.org/10.1007/s10530-007-9089-x
Qin R-M, Zheng Y-L, Valiente-Banuet A et al (2013) The evolution of increased competitive ability, innate competitive advantages, and novel biochemical weapons act in concert for a tropical invader. New Phytol 197:979–988. https://doi.org/10.1111/nph.12071
R Core Team (2018) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/. Accessed 05 Aug 2018
Ridenour WM, Vivanco JM, Feng Y et al (2008) No evidence for trade-offs: centaurea plants from America are better competitors and defenders. Ecol Monogr 78:369–386
Robbins WW (1940) Alien plants growing without cultivation in California. Agricultural Experiment Station, Berkeley
Rotter MC, Holeski LM (2018) A meta-analysis of the evolution of increased competitive ability hypothesis: genetic-based trait variation and herbivory resistance trade-offs. Biol Invasions 46:1–16. https://doi.org/10.1007/s10530-018-1724-1
Sotes GJ, Cavieres LA, Montesinos D et al (2015) Inter-regional variation on leaf surface defenses in native and non-native Centaurea solstitialis plants. Biochem Syst Ecol 62:208–218. https://doi.org/10.1016/j.bse.2015.09.003
Vilà M, Gómez A, Maron JL (2003) Are alien plants more competitive than their native conspecifics? A test using Hypericum perforatum L. Oecologia 137:211–215. https://doi.org/10.1007/s00442-003-1342-0
Xiao S, Callaway RM, Graebner R et al (2016) Modeling the relative importance of ecological factors in exotic invasion: the origin of competitors matters, but disturbance in the non-native range tips the balance. Ecol Modell 335:39–47. https://doi.org/10.1016/j.ecolmodel.2016.05.005
Zou J, Rogers WE, Siemann E (2008) Increased competitive ability and herbivory tolerance in the invasive plant Sapium sebiferum. Biol Invasions 10:291–302. https://doi.org/10.1007/s10530-007-9130-0
Acknowledgements
We would like to thank Giles Thelen, Andrea Krutulis, David Hooper, and Margarita Zorrilla for their help with greenhouse and laboratory work. DM received funding from the Spanish Micinn (2008-0662), and by the Portuguese Fundação para a Ciência e a Tecnologia—FCT, through national funds and co-funding by the European PT2020, and COMPETE 2020 programs, within the Grants and Projects: IF-00066-2013; PTDC/BIA-PLA/0763/2014; and UID/BIA/04004/2013. RCG was funded by the MILES/HHMI program of The University of Montana. RMC thanks the NSF EPSCoR Track-1 EPS-1101342 (INSTEP 3).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Montesinos, D., Graebner, R.C. & Callaway, R.M. Evidence for evolution of increased competitive ability for invasive Centaurea solstitialis, but not for naturalized C. calcitrapa. Biol Invasions 21, 99–110 (2019). https://doi.org/10.1007/s10530-018-1807-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10530-018-1807-z