Abstract
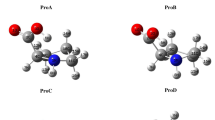
A theoretical study of L-proline-nH2O (n = 1–3) has been performed using the hybrid DFT-B3LYP and MP2 methods together with the 6-311++G(d,p) basis set. The results show that the P2 conformer is energetically favorable when forming a hydrated structure, and the hydration of the carboxyl group leads to the greatest stability. For hydrated complexes, the adiabatic and vertical singlet–triplet excitation energies tend to decrease with the addition of water molecules. The hydration energy indicates that in the hydrated complexes the order of stability is: binding site 2 > binding site 1 > binding site 3, and binding site 12 > binding site 23 > binding site 13. As water molecules are added, the stabilities of these hydrated structures gradually increase. In addition, an infrared frequency analysis indicated that there are some differences in the low-frequency range, which are mainly dominated by the O–H stretching or bending vibrations of different water molecules. All of these results should aid our understanding of molecular behavior and provide reference data for further studies of biological systems.





Similar content being viewed by others
References
Deckert-Gaudig T, Rauls E, Deckert V (2010) J Phys Chem C 114:7412–7420
Soto-Verdugo V, Metiu H, Gwinn E (2010) J Chem Phys 132:195102(1–10)
Parker AW, Lin CY, George MW, Towrie M, Kuimova MK (2010) J Phys Chem B 114:3660–3667
Semencic MC, Heinze K, Forster C, Rapic V (2010) Eur J Inorg Chem 2010:1089–1097
Morera-Boado C, Mora-Diez N, Montero-Cabrera LA, Alonso-Becerra E, González-Jonte RH, de la Vega JMG (2010) J Mol Graph Model 28:604–611
Walch SP (2003) Chem Phys Lett 374:496–500
Evangelista FA, Paul A, Schaefer HF III (2004) J Phys Chem A 108:3565–3571
Magdalena P, Antonio R, Jerzy L (2002) J Phys Chem A 106:11008–11016
Mendham AP, Palmer RA, Potter BS, Dines TJ, Snowden MJ, Withnall R, Chowdhry BZ (2010) J Raman Spectrosc 41:288–302
Fricke H, Schwing K, Gerlach A, Unterberg C, Gerhards M (2010) Phys Chem Chem Phys 12:3511–3521
Clifton E, Jan S, Martin V, Nick CP (2010) J Phys Chem A 114:5919–5927
Rasmussen AM, Lind MC, Kim S, Schaefer HF III (2010) J Chem Theor Comput 6:930–939
Pocker Y (2000) Cell Mol Life Sci 57:1008–1017
Robertson EG, Simons JP (2001) Phys Chem Chem Phys 3:1–18
Chalikian TV, Macgregor RB Jr (2007) Phys Life Rev 4:91–115
Bowers MT, Wyttenbach H (2009) Chem Phys Lett 480:1–16
Pèpe G, Guiliani G, Loustalet S, Halfon P (2002) Eur J Med Chem 37:865–872
Klimovich PV, Mobley DL (2010) J Comput Aided Mol Des 24:307–316
Ashbaugh HS, Kaler EW, Paulaitis ME (1999) J Am Chem Soc 121:9243–9244
Chorny I, Dill KA, Jacobson MP (2005) J Phys Chem B 109:24056–24060
Mobley DL, Barber AE, Fennell CJ, Dill KA (2008) J Phys Chem B 112:2405–2414
Dybal J, Schmidt P, Kriz J, Kurkova D, Rodriguez-Cabello JC, Alonso M (2004) Macromol Symp 205:143–150
Gaigeot MP, Kadri C, Ghomi M (2001) J Mol Struct 565:469–473
Francesco T, Bencini A, Maurizio B, Luca DG, Giuseppe Z, Piercarlo F (2003) J Phys Chem A 107:1188–1196
Vitorino GP, Barrera GD, Mazzieri MR, Binning RC Jr, Bacelo DE (2006) Chem Phys Lett 432:538–544
Vyas N, Ojha AK (2010) J Mol Struct THEOCHEM 940:95–102
Kim HT (2004) J Mol Struct THEOCHEM 673:121–126
Li Q, Wang N, Yu Z (2007) J Mol Struct THEOCHEM 847:68–74
Mobley DL, Bayly CI, Cooper MD, Dill KA (2009) J Phys Chem B 113:4533–4537
Shi Z, Woody RW, Kallenbach NR (2002) Adv Protein Chem 62:163–240
Carlson KL, Lowe SL, Hoffmann MR, Thomasson KA (2006) J Phys Chem A 110:1925–1933
Che Y, Marshall GR (2006) Biopolymer 81:392–406
Ahmed Z, Myshakina NS, Asher SA (2009) J Phys Chem B 113:11252–11259
Zeng B, Shen T, Wu A, Cai S, Yu X, Xu X, Chen Z (2010) J Phys Chem A 114:5211–5216
Hudaky I, Perczel A (2003) J Mol Struct THEOCHEM 630:135–140
Kang YK (2004) J Mol Struct THEOCHEM 675:37–45
Pfeifer GP, You YH, Besaratinia A (2005) Mutat Res 571:19–31
Cadet J, Sage E, Douki T (2005) Mutat Res 571:3–17
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, Mennucci B, Petersson GA, Nakatsuji H, Caricato M, Li X, Hratchian HP, Izmaylov AF, Bloino J, Zheng G, Sonnenberg JL, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Vreven T, Montgomery JA, Peralta JE Jr, Ogliaro F, Bearpark M, Heyd JJ, Brothers E, Kudin KN, Staroverov VN, Kobayashi R, Normand J, Raghavachari K, Rendell A, Burant JC, Iyengar SS, Tomasi J, Cossi M, Rega N, Millam JM, Klene M, Knox, JE Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Martin RL, Morokuma K, Zakrzewski VG, Voth GA, Salvador P, Dannenberg JJ, Dapprich S, Daniels AD, Farkas O, Foresman JB, Ortiz JV, Cioslowski J, Fox DJ (2009) Gaussian 09, revision A.02. Gaussian Inc., Wallingford
Becke AD (1993) J Chem Phys 98:5648–5652
Lee C, Yang W, Parr RG (1988) Phys Rev B 37:785–789
Møller C, Plesset MS (1934) Phys Rev 46:618–622
Mclean AD, Chandler GS (1980) J Chem Phys 72:5639–5648
Krishnan R, Binkley JS, Seeger R, Pople JA (1980) J Chem Phys 72:650–654
Boys SF, Bernardi F (1970) Mol Phys 19:553–566
Simon S, Duran M, Dannenberg JJ (1996) J Chem Phys 105:11024–11031
Cremer D, Pople JA (1975) J Am Chem Soc 97:1354–1358
Abouaf R, Pommier J, Dunet H (2003) Chem Phys Lett 381:486–494
Abouaf R, Pommier J, Dunet H, Quan P, Nam PC, Nguyen MT (2004) J Chem Phys 121:11668–11674
Nguyen MT, Zhang R, Nam PC, Ceulemans A (2004) J Phys Chem A 108:6554–6561
Daněček P, Kapitán J, Baumruk V, Bednárová L, Kopecký V Jr, Bouĭ P (2007) J Chem Phys 126:224513(1–13)
Swenson CA, Formanek R (1967) J Phys Chem 71:4073–4077
Miller FA (1953) In: Gilman H (ed) Organic chemistry, vol 3. Wiley, New York
Acknowledgments
This work was financially supported from the start-up fund (No.08YKZ010) of the Weinan Teachers University of China.
Author information
Authors and Affiliations
Corresponding author
Additional information
Xiao-Jun Li and Zhi-Jian Zhong contributed equally to this work.
Rights and permissions
About this article
Cite this article
Li, XJ., Zhong, ZJ. & Wu, HZ. DFT and MP2 investigations of L-proline and its hydrated complexes. J Mol Model 17, 2623–2630 (2011). https://doi.org/10.1007/s00894-011-0957-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00894-011-0957-z