Abstract
Aims
The purpose of this study was to investigate the mechanism of vascular endothelial cell apoptosis induced by acute blood glucose fluctuation.
Methods
Thirty rats were assigned to three groups: normal saline (SAL group), constant high glucose (CHG group) and acute blood glucose fluctuation (AFG) group. Other forty rats were assigned to SAL group, AFG group, LY group (PKCβ inhibitor LY333531 was injected intragastrically to the rats who were under acute blood glucose fluctuation) and SP group (JNK inhibitor SP600125 was injected intraperitoneally to the rats who were under acute blood glucose fluctuation). Oxidative stress and inflammatory cytokines were detected. TUNEL was performed to detect apoptosis. Pro-caspase-3, caspase-3 p17, JNK, PKC-βII and insulin signaling-related protein expression were tested by Western blotting.
Results
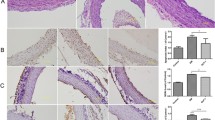
After administration of LY333531, AFG-induced membrane translocation of PKCβII protein was inhibited, but SP600125 failed to affect AFG-induced PKCβII membrane translocation. After administration of LY333531, the AFG-induced increase in JNK activity was significantly compromised. LY333531 inhibited AFG-induced oxidative stress. However, SP600125 only slightly inhibited AFG-induced oxidative stress reaction (P > 0.05). Both LY333531 and SP600125 can reverse AFG-induced endothelial cell apoptosis increase, inflammatory cytokines levels rise and insulin signaling impairment.
Conclusions
It is necessary to actively control blood glucose and avoid significant glucose fluctuation. PKCβII/JNK may serve as a target, and inhibitors of PKCβII/JNK may be used to help prevent cardiovascular diseases in patients with poor glucose control or significant glucose fluctuation.





Similar content being viewed by others
References
Singh S, Usman K, Banerjee M (2016) Pharmacogenetic studies update in type 2 diabetes mellitus. World J Diabetes 7(15):302–315
Jung HS (2015) Clinical implications of glucose variability: chronic complications of diabetes. Endocrinol Metab 30(2):167–174
Torimoto K, Okada Y, Mori H et al (2013) Relationship between fluctuations in glucose levels measured by continuous glucose monitoring and vascular endothelial dysfunction in type 2 diabetes mellitus. Cardiovasc Diabetol 12(1):1
Lorenzi M, Cagliero E (1991) Pathobiology of endothelial and other vascular cells in diabetes mellitus. Call for data. Diabetes 40(40):653–659
Cohen RA (1993) Dysfunction of vascular endothelium in diabetes mellitus. Monogr Am Heart Assoc 87(5):67–76
Risso A, Mercuri F, Quagliaro L et al (2001) Intermittent high glucose enhances apoptosis in human umbilical vein endothelial cells in culture. Am J Physiol Endocrinol Metab 281(5):E924–E930
Liu T, Pei Y, Peng Y et al (2014) Oscillating high glucose enhances oxidative stress and apoptosis in human coronary artery endothelial cells. J Endocrinol Invest 37(7):645–651
Abdelzaher LA, Imaizumi T, Suzuki T et al (2016) Astaxanthin alleviates oxidative stress insults-related derangements in human vascular endothelial cells exposed to glucose fluctuations. Life Sci 150:24–31
Schisano B, Tripathi G, Mcgee K et al (2011) Glucose oscillations, more than constant high glucose, induce p53 activation and a metabolic memory in human endothelial cells. Diabetologia 54(5):1219–1226
Liu T, Gong J, Chen Y et al (2013) Periodic vs constant high glucose in inducing pro-inflammatory cytokine expression in human coronary artery endothelial cells. Inflamm Res 62(7):697–701
Piconi L, Quagliaro L, Assaloni R et al (2006) Constant and intermittent high glucose enhances endothelial cell apoptosis through mitochondrial superoxide overproduction. Diabetes Metab Res Rev 22(3):198–203
Quagliaro L, Piconi L, Assaloni R et al (2003) Intermittent high glucose enhances apoptosis related to oxidative stress in human umbilical vein endothelial cells. Diabetes 52(11):2795–2804
Mita T, Otsuka AK, Uchida T et al (2007) Swings in blood glucose levels accelerate atherogenesis in apolipoprotein E-deficient mice. Biochem Biophys Res Commun 358(358):679–685
Azuma K, Kawamori R, Toyofuku Y et al (2006) Repetitive fluctuations in blood glucose enhance monocyte adhesion to the endothelium of rat thoracic aorta. Arterioscler Thromb Vasc Biol 26(10):2275–2280
Zhang W, Zhao S, Li Y et al (2013) Acute blood glucose fluctuation induces myocardial apoptosis through oxidative stress and nuclear factor-κB activation. Cardiology 124(1):11–17
Na W, Shen H, Liu H et al (2016) Acute blood glucose fluctuation enhances rat aorta endothelial cell apoptosis, oxidative stress and pro-inflammatory cytokine expression in vivo. Cardiovas Diabetol 15(1):109
Na W, Yan L, Bing H et al (2010) Taurine prevents free fatty acid-induced hepatic insulin resistance in association with inhibiting JNK1 activation and improving insulin signaling in vivo. Diabetes Res Clin Pract 90(3):288–296
Li H, Li H, Bao Y et al (2011) Free fatty acids induce endothelial dysfunction and activate protein kinase C and nuclear factor-κB pathway in rat aorta. Int J Cardiol 152(2):218–224
Kavanagh E, Rodhe J, Burguillos MA et al (2013) Regulation of caspase-3 processing by cIAP2 controls the switch between pro-inflammatory activation and cell death in microglia. Cell Death Dis 5(12):e1565
Huang J, Zhang X, Li J et al (2014) Impact of glucose fluctuation on acute cerebral infarction in type 2 diabetes. Can J Neurol Sci 41(4):486
Tang C, Han P, Oprescu AI et al (2007) Evidence for a role of superoxide generation in glucose-induced beta-cell dysfunction in vivo. Diabetes 56:2722–2731
Qin W, Bei R, Wang S et al (2016) Apigenin and naringenin ameliorate PKCβII-associated endothelial dysfunction via regulating ROS/caspase-3 and NO pathway in endothelial cells exposed to high glucose. Vascul Pharmacol 85:39–49
Qin W, Xi J, He B et al (2016) Ameliorative effects of hispidulin on high glucose-mediated endothelial dysfunction via inhibition of PKCβII-associated NLRP3 inflammasome activation and NF-κB signaling in endothelial cells. J Funct Foods 27:392–405
Feng MH, Liu SH, Liau CS et al (2000) High glucose-induced apoptosis in human endothelial cells is mediated by sequential activations of c-Jun NH(2)-terminal kinase and caspase-3. Circulation 101(22):2618–2624
Liu W, Schoenkerman A, Lowe WL Jr (2000) Activation of members of the mitogen-activated protein kinase family by glucose in endothelial cells. Am J Physiol Endocrinol Metab 279(4):E782
Chen YR, Meyer CF, Tan TH (1996) Persistent activation of c-Jun N-terminal kinase 1 (JNK1) in gamma radiation-induced apoptosis. J Biol Chem 271(2):631–634
Maeda M, Hayashi T, Mizuno N et al (2015) Intermittent high glucose implements stress-induced senescence in human vascular endothelial cells: role of superoxide production by NADPH oxidase. PLoS ONE 10(4):e0123169
Tang Z, Lei M (2010) Involvement of JNK signal transduction pathway in endothelial cell apoptosis induced by intermittent high glucose. J Cent South Univ (Med Sci) 35(6):616–621
Yamawaki H, Saito K, Okada M et al (2008) Methylglyoxal mediates vascular inflammation via JNK and p38 in human endothelial cells. AJP Cell Physiol 295(6):C1510–C1517
Acknowledgements
The research was supported by Grants from Project of Liaoning Provincial Educational Bureau (LK201603, LK201633, L2015568).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical standard
This article does not contain any study with human participants. All procedures performed in this study involving animals were in accordance with the ethical standards of “Ethics Committee Acts” of Shengjing Hospital of China Medical University.
Human and animal rights
This article does not contain any studies with human performed by the any of the authors. The experimental procedures for all animals were in accordance with the Ethics Committee Acts of Shengjing Hospital of China Medical University.
Informed consent
For this type of study formal consent is not required.
Additional information
Managed by Massimo Porta.
Rights and permissions
About this article
Cite this article
Wu, N., Shen, H., Wang, Y. et al. Role of the PKCβII/JNK signaling pathway in acute glucose fluctuation-induced apoptosis of rat vascular endothelial cells. Acta Diabetol 54, 727–736 (2017). https://doi.org/10.1007/s00592-017-0999-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00592-017-0999-5