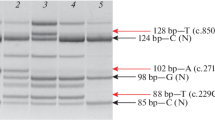
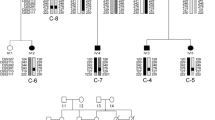
Abstract
Autosomal dominant Emery–Dreifuss muscular dystrophy is caused by mutations in LMNA gene encoding lamins A and C. The disease is characterized by early onset joint contractures during childhood associated with humero-peroneal muscular wasting and weakness, and by the development of a cardiac disease in adulthood. Important intra-familial variability characterized by a wide range of age at onset of myopathic symptoms (AOMS) has been recurrently reported, suggesting the contribution of a modifier gene. Our objective was to identify a modifier locus of AOMS in relation with the LMNA mutation. To map the modifier locus, we genotyped 291 microsatellite markers in 59 individuals of a large French family, where 19 patients carrying the same LMNA mutation, exhibited wide range of AOMS. We performed Bayesian Markov Chain Monte Carlo-based joint segregation and linkage methods implemented in the Loki© software, and detected a strong linkage signal on chromosome 2 between markers D2S143 and D2S2244 (211 cM) with a Bayes factor of 28.7 (empirical p value = 0.0032). The linked region harbours two main candidate genes, DES and MYL1 encoding desmin and light chain of myosin. Importantly, the impact of the genotype on the phenotype for this locus showed an overdominant effect with AOMS 2 years earlier for the homozygotes of the rare allele and 37 years earlier for the heterozygotes than the homozygotes for the common allele. These results provide important highlights for the natural history and for the physiopathology of Emery–Dreifuss muscular dystrophy.




Similar content being viewed by others
References
Allison AC (1956) The sickle-cell and haemoglobin C genes in some African populations. Ann Hum Genet 21:67–89
Alonso S, Lopez S, Izagirre N, de la Rua C (2008) Overdominance in the human genome and olfactory receptor activity. Mol Biol Evol 25:997–1001
Ayroles JF, Hughes KA, Rowe KC, Reedy MM, Rodriguez-Zas SL, Drnevich JM, Caceres CE, Paige KN (2009) A genomewide assessment of inbreeding depression: gene number, function, and mode of action. Conserv Biol 23:920–930
Becane HM, Bonne G, Varnous S, Muchir A, Ortega V, Hammouda EH, Urtizberea JA, Lavergne T, Fardeau M, Eymard B, Weber S, Schwartz K, Duboc D (2000) High incidence of sudden death with conduction system and myocardial disease due to lamins A and C gene mutation. Pacing Clin Electrophysiol 23:1661–1666
Ben Yaou R, Toutain A, Arimura T, Demay L, Massart C, Peccate C, Muchir A, Llense S, Deburgrave N, Leturcq F, Litim KE, Rahmoun-Chiali N, Richard P, Babuty D, Recan-Budiartha D, Bonne G (2007) Multitissular involvement in a family with LMNA and EMD mutations: role of digenic mechanism? Neurology 68:1883–1894
Bione S, Maestrini E, Rivella S, Mancini M, Regis S, Romeo G, Toniolo D (1994) Identification of a novel X-linked gene responsible for Emery–Dreifuss muscular dystrophy. Nat Genet 8:323–327
Bonne G, Di Barletta MR, Varnous S, Becane HM, Hammouda EH, Merlini L, Muntoni F, Greenberg CR, Gary F, Urtizberea JA, Duboc D, Fardeau M, Toniolo D, Schwartz K (1999) Mutations in the gene encoding lamin A/C cause autosomal dominant Emery–Dreifuss muscular dystrophy. Nat Genet 21:285–288
Bonne G, Mercuri E, Muchir A, Urtizberea A, Becane HM, Recan D, Merlini L, Wehnert M, Boor R, Reuner U, Vorgerd M, Wicklein EM, Eymard B, Duboc D, Penisson-Besnier I, Cuisset JM, Ferrer X, Desguerre I, Lacombe D, Bushby K, Pollitt C, Toniolo D, Fardeau M, Schwartz K, Muntoni F (2000) Clinical and molecular genetic spectrum of autosomal dominant Emery–Dreifuss muscular dystrophy due to mutations of the lamin A/C gene. Ann Neurol 48:170–180
Brodsky GL, Muntoni F, Miocic S, Sinagra G, Sewry C, Mestroni L (2000) Lamin A/C gene mutation associated with dilated cardiomyopathy with variable skeletal muscle involvement. Circulation 101:473–476
Carboni N, Mura M, Marrosu G, Cocco E, Ahmad M, Solla E, Mateddu A, Maioli MA, Marini S, Nissardi V, Frau J, Mallarini G, Mercuro G, Marrosu MG (2008) Muscle MRI findings in patients with an apparently exclusive cardiac phenotype due to a novel LMNA gene mutation. Neuromuscul Disord 18:291–298
Celeux GD (1990) The EM and the SEM algorithms for mixtures: statistical and numerical aspects. Cahiers CERO 32:135–151
Cox NJ, Frigge M, Nicolae DL, Concannon P, Hanis CL, Bell GI, Kong A (1999) Loci on chromosomes 2 (NIDDM1) and 15 interact to increase susceptibility to diabetes in Mexican Americans. Nat Genet 21:213–215
Daw EW, Heath SC, Wijsman EM (1999a) Multipoint oligogenic analysis of age-at-onset data with applications to Alzheimer disease pedigrees. Am J Hum Genet 64:839–851
Daw EW, Kumm J, Snow GL, Thompson EA, Wijsman EM (1999b) Monte Carlo Markov chain methods for genome screening. Genet Epidemiol 17(Suppl 1):S133–S138
Daw EW, Liu X, Wu CC (2003a) Age-of-onset of hypertension vs. a single measurement of systolic blood pressure in a combined linkage and segregation analysis. BMC Genet 4(Suppl 1):S80
Daw EW, Wijsman EM, Thompson EA (2003b) A score for Bayesian genome screening. Genet Epidemiol 24:181–190
Daw EW, Chen SN, Czernuszewicz G, Lombardi R, Lu Y, Ma J, Roberts R, Shete S, Marian AJ (2007) Genome-wide mapping of modifier chromosomal loci for human hypertrophic cardiomyopathy. Hum Mol Genet 16:2463–2471
Daw EW, Lu Y, Marian AJ, Shete S (2008) Identifying modifier loci in existing genome scan data. Ann Hum Genet 72:670–675
Emery AE (2000) Emery–Dreifuss muscular dystrophy—a 40 year retrospective. Neuromuscul Disord 10:228–232
Falconer DS, Mackay TFC (1995) (ed) Introduction to quantitative genetics. New York, Longman
Fatkin D, MacRae C, Sasaki T, Wolff MR, Porcu M, Frenneaux M, Atherton J, Vidaillet HJ Jr, Spudich S, De Girolami U, Seidman JG, Seidman C, Muntoni F, Muehle G, Johnson W, McDonough B (1999) Missense mutations in the rod domain of the lamin A/C gene as causes of dilated cardiomyopathy and conduction-system disease. N Engl J Med 341:1715–1724
Gagnon F, Jarvik GP, Badzioch MD, Motulsky AG, Brunzell JD, Wijsman EM (2005) Genome scan for quantitative trait loci influencing HDL levels: evidence for multilocus inheritance in familial combined hyperlipidemia. Hum Genet 117:494–505
Genin E, Feingold J, Clerget-Darpoux F (2008) Identifying modifier genes of monogenic disease: strategies and difficulties. Hum Genet 124:357–368
Gueneau L, Bertrand AT, Jais JP, Salih MA, Stojkovic T, Wehnert M, Hoeltzenbein M, Spuler S, Saitoh S, Verschueren A, Tranchant C, Beuvin M, Lacene E, Romero NB, Heath S, Zelenika D, Voit T, Eymard B, Ben Yaou R, Bonne G (2009) Mutations of the FHL1 gene cause Emery–Dreifuss muscular dystrophy. Am J Hum Genet 85:338–353
Heath SC (1997) Markov chain Monte Carlo segregation and linkage analysis for oligogenic models. Am J Hum Genet 61:748–760
Heath SC, Snow GL, Thompson EA, Tseng C, Wijsman EM (1997) MCMC segregation and linkage analysis. Genet Epidemiol 14:1011–1016
Igo RP Jr, Wijsman EM (2008) Empirical significance values for linkage analysis: trait simulation using posterior model distributions from MCMC oligogenic segregation analysis. Genet Epidemiol 32:119–131
Kass RE, Raftery AE (1995) Bayes Factors. J Am Stat Assoc 90:773–795
Liu X, Ramjiganesh T, Chen YH, Chung SW, Hall SR, Schissel SL, Padera RF Jr, Liao R, Ackerman KG, Kajstura J, Leri A, Anversa P, Yet SF, Layne MD, Perrella MA (2009) Disruption of striated preferentially expressed gene locus leads to dilated cardiomyopathy in mice. Circulation 119:261–268
Ma J, Amos CI, Warwick Daw E (2007) Ascertainment correction for Markov chain Monte Carlo segregation and linkage analysis of a quantitative trait. Genet Epidemiol 31:594–604
Mead S, Stumpf MP, Whitfield J, Beck JA, Poulter M, Campbell T, Uphill JB, Goldstein D, Alpers M, Fisher EM, Collinge J (2003) Balancing selection at the prion protein gene consistent with prehistoric kuru like epidemics. Science 300:640–643
Mercuri E, Poppe M, Quinlivan R, Messina S, Kinali M, Demay L, Bourke J, Richard P, Sewry C, Pike M, Bonne G, Muntoni F, Bushby K (2004) Extreme variability of phenotype in patients with an identical missense mutation in the lamin A/C gene: from congenital onset with severe phenotype to milder classic Emery–Dreifuss variant. Arch Neurol 61:690–694
Muchir A, Bonne G, van der Kooi AJ, van Meegen M, Baas F, Bolhuis PA, de Visser M, Schwartz K (2000) Identification of mutations in the gene encoding lamins A/C in autosomal dominant limb girdle muscular dystrophy with atrioventricular conduction disturbances (LGMD1B). Hum Mol Genet 9:1453–1459
Muntoni F, Bonne G, Goldfarb LG, Mercuri E, Piercy RJ, Burke M, Yaou RB, Richard P, Recan D, Shatunov A, Sewry CA, Brown SC (2006) Disease severity in dominant Emery–Dreifuss is increased by mutations in both emerin and desmin proteins. Brain 129:1260–1268
Nadeau JH (2001) Modifier genes in mice and humans. Nat Rev Genet 2:165–174
Nikolova V, Leimena C, McMahon AC, Tan JC, Chandar S, Jogia D, Kesteven SH, Michalicek J, Otway R, Verheyen F, Rainer S, Stewart CL, Martin D, Feneley MP, Fatkin D (2004) Defects in nuclear structure and function promote dilated cardiomyopathy in lamin A/C-deficient mice. J Clin Invest 113:357–369
O’Connell JR, Weeks DE (1998) PedCheck: a program for identification of genotype incompatibilities in linkage analysis. Am J Hum Genet 63:259–266
Rankin J, Auer-Grumbach M, Bagg W, Colclough K, Nguyen TD, Fenton-May J, Hattersley A, Hudson J, Jardine P, Josifova D, Longman C, McWilliam R, Owen K, Walker M, Wehnert M, Ellard S (2008) Extreme phenotypic diversity and nonpenetrance in families with the LMNA gene mutation R644C. Am J Med Genet A 146A:1530–1542
Rosenthal EA, Wijsman EM (2010) Joint linkage and segregation analysis under multiallelic trait inheritance: simplifying interpretations for complex traits. Genet Epidemiol 34:344–353
Seidel U, Bober E, Winter B, Lenz S, Lohse P, Goedde HW, Grzeschik KH, Arnold HH (1988) Alkali myosin light chains in man are encoded by a multigene family that includes the adult skeletal muscle, the embryonic or atrial, and nonsarcomeric isoforms. Gene 66:135–146
Shmulewitz D, Heath SC (2001) Genome scans for Q1 and Q2 on general population replicates using Loki. Genet Epidemiol 21(Suppl 1):S686–S691
Shmulewitz D, Heath SC, Blundell ML, Han Z, Sharma R, Salit J, Auerbach SB, Signorini S, Breslow JL, Stoffel M, Friedman JM (2006) Linkage analysis of quantitative traits for obesity, diabetes, hypertension, and dyslipidemia on the island of Kosrae, Federated States of Micronesia. Proc Natl Acad Sci USA 103:3502–3509
Tam JL, Triantaphyllopoulos K, Todd H, Raguz S, de Wit T, Morgan JE, Partridge TA, Makrinou E, Grosveld F, Antoniou M (2006) The human desmin locus: gene organization and LCR-mediated transcriptional control. Genomics 87:733–746
Verrelli BC, McDonald JH, Argyropoulos G, Destro-Bisol G, Froment A, Drousiotou A, Lefranc G, Helal AN, Loiselet J, Tishkoff SA (2002) Evidence for balancing selection from nucleotide sequence analyses of human G6PD. Am J Hum Genet 71:1112–1128
Wijsman EM, Yu D (2004) Joint oligogenic segregation and linkage analysis using bayesian Markov chain Monte Carlo methods. Mol Biotechnol 28:205–226
Wijsman EM, Rothstein JH, Thompson EA (2006) Multipoint linkage analysis with many multiallelic or dense diallelic markers: Markov chain-Monte Carlo provides practical approaches for genome scans on general pedigrees. Am J Hum Genet 79:846–858
Wijsman EM, Rothstein JH, Igo RP Jr, Brunzell JD, Motulsky AG, Jarvik GP (2010) Linkage and association analyses identify a candidate region for apoB level on chromosome 4q32.3 in FCHL families. Hum Genet 127:705–719
Worman HJ, Bonne G (2007) “Laminopathies”: a wide spectrum of human diseases. Exp Cell Res 313:2121–2133
Zhang Q, Bethmann C, Worth NF, Davies JD, Wasner C, Feuer A, Ragnauth CD, Yi Q, Mellad JA, Warren DT, Wheeler MA, Ellis JA, Skepper JN, Vorgerd M, Schlotter-Weigel B, Weissberg PL, Roberts RG, Wehnert M, Shanahan CM (2007) Nesprin-1 and -2 are involved in the pathogenesis of Emery Dreifuss muscular dystrophy and are critical for nuclear envelope integrity. Hum Mol Genet 16:2816–2833
Acknowledgments
We thank Michel Krawczyk from the Computer Center and Research Network of Jussieu (Paris, France) and Jean-Louis Gaunet from Pitié-Salpétrière Hospital (Paris, France) for their valuable methodological assistance; Véronique Ortega, Andoni Urtizberea, Denis Duboc, El-Hadi Hammouda and Henri-Marc Bécane for their precious help in the clinical evaluation of the EDM1 family. This work was supported by the Institut National de la Santé et de la Recherche Médicale (INSERM); the Université Paris 06; the Centre National de la Recherche Scientifique (CNRS); the Association Française contre les Myopathies [#11057, #11034 and financial support for L.G.]; the European Union Sixth Framework Programme [Euro-laminopathies #018690] and F. G. holds a Canada Research Chair.
Conflict of interest
There is no direct or indirect conflict of interest for any of the authors for the data presented in the manuscript in whole or in part.
Author information
Authors and Affiliations
Corresponding author
Additional information
The authors wish it to be known that, in their opinion, the B. Granger, L. Gueneau authors should be regarded as joint first authors and the S. Tezenas du Montcel, G. Bonne authors should be regarded as joint last authors.
Electronic supplementary material
Below is the link to the electronic supplementary material.
439_2010_909_MOESM1_ESM.jpeg
Schematic presentation of candidate gene regions that were sequenced. Arrows indicated positions of the primers used for sequencing Supplementary material 2 (DOCX 29 kb)
Rights and permissions
About this article
Cite this article
Granger, B., Gueneau, L., Drouin-Garraud, V. et al. Modifier locus of the skeletal muscle involvement in Emery–Dreifuss muscular dystrophy. Hum Genet 129, 149–159 (2011). https://doi.org/10.1007/s00439-010-0909-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00439-010-0909-1