Abstract
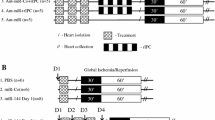
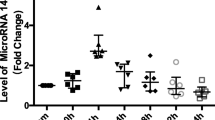
Ischemic postconditioning (PoCo) reduces infarct size following myocardial ischemia/reperfusion. To protect, PoCo must be performed early during reperfusion, and causal cardioprotective signaling must occur then. The role of microRNA (miRNA) in PoCo is unclear. Anesthetized pigs were subjected to 60 min left anterior descending coronary artery (LAD) occlusion and 180 min reperfusion. Immediate full reperfusion (IFR, n = 5) was compared to PoCo (four cycles of 60 s/60 s reperfusion/reocclusion, n = 5). Transmural myocardial biopsies from the LAD territory were sampled at baseline, 60 min ischemia, 10 and 180 min reperfusion. RNA was isolated. The expression of 11 miRNAs, including muscle-specific (miRNA-1, -133a, -206, -208b, -214, and -499), fibrosis- (miRNA-21, -24, and -29b), neovascularization- (miRNA-92a), and inflammation-associated (miRNA-146b) candidates, was quantified using real-time PCR (RT-PCR). mRNA expression at baseline and 180 min reperfusion was quantified and validated (microarray and RT-PCR). PoCo reduced infarct size from 44.9 ± 7.7 to 34.8 ± 5.3 % of the area at risk. The expression of miRNA-1, -24, -29b, -133a, -146b, -208b, and -499 was increased at 10 min reperfusion with PoCo vs. IFR; however, that of miRNA-1, -24, -208b, and -499 was already increased at 60 min ischemia and probably reflects falsely positive results. Five mRNAs were different with PoCo vs. IFR. In silico analysis identified a tentative connection between three miRNAs and five mRNAs with the biological functions “cell death”, “inflammatory response” and/or “glucose metabolism”. If at all, only miRNA-29b, -133a, and -146b fulfill the minimal temporal requirements for a potential causal involvement in cardioprotection by PoCo.


Similar content being viewed by others
References
Arad M, Seidman CE, Seidman JG (2007) AMP-activated protein kinase in the heart: role during health and disease. Circ Res 100:474–488. doi:10.1161/01.RES.0000258446.23525.37
Bartel DP (2004) MicroRNAs: genomics, biogenesis, mechanism, and function. Cell 116:281–297. doi:10.1016/j.bbr.2011.03.031
Benjamini Y, Hochberg Y (1995) Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Statist Soc B 57:289–300. doi:10.1161/hc4001.097183
Bonauer A, Carmona G, Iwasaki M, Mione M, Koyanagi M, Fischer A, Burchfield J, Fox H, Doebele C, Ohtani K, Chavakis E, Potente M, Tjwa M, Urbich C, Zeiher AM, Dimmeler S (2009) MicroRNA-92a controls angiogenesis and functional recovery of ischemic tissues in mice. Science 324:1710–1713. doi:10.1126/science.1174381
Bostjancic E, Zidar N, Stajer D, Glavac D (2009) MicroRNAs miR-1, miR-133a, miR-133b and miR-208 are dysregulated in human myocardial infarction. Cardiology 115:163–169. doi:10.1159/000268088
Chandrasekar B, Freeman GL (1997) Induction of nuclear factor kB and activation protein 1 in postischemic myocardium. FEBS Lett 401:30–34. doi:10.1016/S0014-5793(96)01426-3
Dong S, Cheng Y, Yang J, Li J, Liu X, Wang X, Wang D, Krall TJ, Delphin ES, Zhang C (2009) MicroRNA expression signature and the role of microRNA-21 in the early phase of acute myocardial infarction. J Biol Chem 284:29514–29525. doi:10.1074/jbc.M109.027896
Fiedler J, Jazbutyte V, Kirchmaier BC, Gupta SK, Lorenzen J, Hartmann D, Galuppo P, Kneitz S, Pena JT, Sohn-Lee C, Loyer X, Soutschek J, Brand T, Tuschl T, Heineke J, Martin U, Schulte-Merker S, Ertl G, Engelhardt S, Bauersachs J, Thum T (2011) MicroRNA-24 regulates vascularity after myocardial infarction. Circulation 124:720–730. doi:10.1161/CIRCULATIONAHA.111.039008
Fleige S, Pfaffl MW (2006) RNA integrity and the effect on the real-time qRT-PCR performance. Mol Aspects Med 27:126–139. doi:10.1016/j.mam.2005.12.003
He B, Xiao J, Ren AJ, Zhang YF, Zhang H, Chen M, Xie B, Gao XG, Wang YW (2011) Role of miR-1 and miR-133a in myocardial ischemic postconditioning. J Biomed Sci 18:22. doi:10.1186/1423-0127-18-22
Heusch G (2004) Postconditioning. Old wine in a new bottle? J Am Coll Cardiol 44:1111–1112. doi:10.1016/j.jacc.2004.06.013
Heusch G (2013) Cardioprotection—chances and challenges of its translation to the clinic. Lancet 381:166–175. doi:10.1016/S0140-6736(12)60916-7
Heusch G, Musiolik J, Gedik N, Skyschally A (2011) Mitochondrial STAT3 activation and cardioprotection by ischemic postconditioning in pigs with regional myocardial ischemia/reperfusion. Circ Res 109:1302–1308. doi:10.1161/CIRCRESAHA.111.255604
Heusch G, Schulz R (2009) Neglect of the coronary circulation: some critical remarks on problems in the translation of cardioprotection. Cardiovasc Res 84:11–14. doi:10.1093/cvr/cvp210
Heusch G, Skyschally A, Schulz R (2011) The in-situ pig heart with regional ischemia/reperfusion—ready for translation. J Mol Cell Cardiol 50:951–963. doi:10.1016/j.yjmcc.2011.02.016
Hinkel R, Penzkofer D, Zuhlke S, Fischer A, Husada W, Xu QF, Baloch E, van Rooij E, Zeiher AM, Kupatt C, Dimmeler S (2013) Inhibition of microRNA-92a protects against ischemia-reperfusion injury in a large animal model. Circulation 128:1066–1075. doi:10.1161/CIRCULATIONAHA.113.00190
Kowallik P, Schulz R, Guth BD, Schade A, Paffhausen W, Gross R, Heusch G (1991) Measurement of regional myocardial blood flow with multiple colored microspheres. Circulation 83:974–982. doi:10.1161/01.CIR.83.3974
Kukreja RC, Yin C, Salloum FN (2011) MicroRNAs: new players in cardiac injury and protection. Mol Pharmacol 80:558–564. doi:10.1124/mol.111.073528
Lassen JF, Botker HE, Terkelsen CJ (2012) Timely and optimal treatment of patients with STEMI. Nat Rev Cardiol 10:41–48. doi:10.1038/nrcardio.2012.156
Li C, Browder W, Kao RL (1999) Early activation of transcription factor NF-kappaB during ischemia in perfused rat heart. Am J Physiol 276:H543–H552
Li G, Labruto F, Sirsjo A, Chen F, Vaage J, Valen G (2004) Myocardial protection by remote preconditioning: the role of nuclear factor kappa-B p105 and inducible nitric oxide synthase. Eur J Cardiothorac Surg 26:968–973. doi:10.1016/j.ejcts.2004.06.015
Li J, Coven DL, Miller EJ, Hu X, Young ME, Carling D, Sinusas AJ, Young LH (2006) Activation of AMPK alpha- and gamma-isoform complexes in the intact ischemic rat heart. Am J Physiol Heart Circ Physiol 291:H1927–H1934. doi:10.1152/ajpheart.00251.2006
Martinez-Gonzalez J, Rius J, Castello A, Cases-Langhoff C, Badimon L (2003) Neuron-derived orphan receptor-1 (NOR-1) modulates vascular smooth muscle cell proliferation. Circ Res 92:96–103. doi:10.1161/01.RES.0000050921.53008.47
Matthews JM, Lester K, Joseph S, Curtis DJ (2013) LIM-domain-only proteins in cancer. Nat Rev Cancer 13:111–122. doi:10.1038/nrc3418
Morgan EN, Boyle EM Jr, Yun W, Griscavage-Ennis JM, Farr AL, Canty TG Jr, Pohlman TH, Verrier ED (1999) An essential role of NF-kappaB in the cardioadaptive response to ischemia. Ann Thorac Surg 68:377–382. doi:10.1016/S0003-4975(99)00646-3
Musi N, Hirshman MF, Arad M, Xing Y, Fujii N, Pomerleau J, Ahmad F, Berul CI, Seidman JG, Tian R, Goodyear LJ (2005) Functional role of AMP-activated protein kinase in the heart during exercise. FEBS Lett 579:2045–2050. doi:10.1016/j.febslet.2005.02.052
Ohtani K, Dimmeler S (2011) Control of cardiovascular differentiation by microRNAs. Basic Res Cardiol 106:5–11. doi:10.1007/s00395-010-0139-7
Ovize M, Baxter GF, Di Lisa F, Ferdinandy P, Garcia-Dorado D, Hausenloy DJ, Heusch G, Vinten-Johansen J, Yellon DM, Schulz R (2010) Postconditioning and protection from reperfusion injury: where do we stand? Cardiovasc Res 87:406–423. doi:10.1093/cvr/cvq129
Park SY, Lee JH, Ha M, Nam JW, Kim VN (2009) miR-29 miRNAs activate p53 by targeting p85 alpha and CDC42. Nat Struct Mol Biol 16:23–29. doi:10.1038/nsmb.1533
Roy S, Khanna S, Hussain SR, Biswas S, Azad A, Rink C, Gnyawali S, Shilo S, Nuovo GJ, Sen CK (2009) MicroRNA expression in response to murine myocardial infarction: miR-21 regulates fibroblast metalloprotease-2 via phosphatase and tensin homologue. Cardiovasc Res 82:21–29. doi:10.1093/cvr/cvp015
Rozen S, Skaletsky H (2000) Primer3 on the WWW for general users and for biologist programmers. Methods Mol Biol 132:365–386
Russell RR III, Li J, Coven DL, Pypaert M, Zechner C, Palmeri M, Giordano FJ, Mu J, Birnbaum MJ, Young LH (2004) AMP-activated protein kinase mediates ischemic glucose uptake and prevents postischemic cardiac dysfunction, apoptosis, and injury. J Clin Invest 114:495–503. doi:10.1172/JCI21233
Shan ZX, Lin QX, Fu YH, Deng CY, Zhou ZL, Zhu JN, Liu XY, Zhang YY, Li Y, Lin SG, Yu XY (2009) Upregulated expression of miR-1/miR-206 in a rat model of myocardial infarction. Biochem Biophys Res Commun 381:597–601. doi:10.1016/j.bbrc.2009.02.097
Siegrist F, Ebeling M, Certa U (2011) The small interferon-induced transmembrane genes and proteins. J Interferon Cytokine Res 31:183–197. doi:10.1089/jir.2010.0112
Skyschally A, van Caster P, Boengler K, Gres P, Musiolik J, Schilawa D, Schulz R, Heusch G (2009) Ischemic postconditioning in pigs: no causal role for RISK activation. Circ Res 104:15–18. doi:10.1161/CIRCRESAHA.108.186429
Skyschally A, van Caster P, Iliodromitis EK, Schulz R, Kremastinos DT, Heusch G (2009) Ischemic postconditioning—experimental models and protocol algorithms. Basic Res Cardiol 104:469–483. doi:10.1007/s00395-009-0040-4
Staat P, Rioufol G, Piot C, Cottin Y, Cung TT, L’Huillier I, Aupetit J-F, Bonnefoy E, Finet G, Andre-Fouet X, Ovize M (2005) Postconditioning the human heart. Circulation 112:2143–2148. doi:10.1161/CIRCULATIONAHA.105.558122
Tian R, Musi N, D’Agostino J, Hirshman MF, Goodyear LJ (2001) Increased adenosine monophosphate-activated protein kinase activity in rat hearts with pressure-overload hypertrophy. Circulation 104:1664–1669. doi:10.1161/hc4001.097183
Valen G (2009) Extracardiac approaches to protecting the heart. Eur J Cardiothorac Surg 35:651–657. doi:10.1016/j.ejcts.2008.12.023
van Rooij E, Sutherland LB, Thatcher JE, DiMaio JM, Naseem RH, Marshall WS, Hill JA, Olson EN (2008) Dysregulation of microRNAs after myocardial infarction reveals a role of miR-29 in cardiac fibrosis. Proc Natl Acad Sci U S A 105:13027–13032. doi:10.1073/pnas.0805038105
Wang JX, Jiao JQ, Li Q, Long B, Wang K, Liu JP, Li YR, Li PF (2011) miR-499 regulates mitochondrial dynamics by targeting calcineurin and dynamin-related protein-1. Nat Med 17:71–78. doi:10.1038/nm.2282
Weiss JB, Eisenhardt SU, Stark GB, Bode C, Moser M, Grundmann S (2012) MicroRNAs in ischemia-reperfusion injury. Am J Cardiovasc Dis 2:237–247
Welman E, Colbeck JF, Selwyn AP, Fox KM, Orr I (1980) Plasma lysosomal enzyme activity in acute myocardial infarction and the effects of drugs. Adv Myocardiol 2:359–369
Welman E, Selwyn AP, Peters TJ, Colbeck JF, Fox KM (1978) Plasma lysosomal enzyme activity in acute myocardial infarction. Cardiovasc Res 12:99–105
Xu C, Lu Y, Pan Z, Chu W, Luo X, Lin H, Xiao J, Shan H, Wang Z, Yang B (2007) The muscle-specific microRNAs miR-1 and miR-133 produce opposing effects on apoptosis by targeting HSP60, HSP70 and caspase-9 in cardiomyocytes. J Cell Sci 120:3045–3052. doi:10.1242/jcs.098830
Ye Y, Hu Z, Lin Y, Zhang C, Perez-Polo JR (2010) Downregulation of microRNA-29 by antisense inhibitors and a PPAR-g agonist protects against myocardial ischaemia-reperfusion injury. Cardiovasc Res 87:535–544. doi:10.1093/cvr/cvq053
Yellon DM, Hausenloy DJ (2007) Myocardial reperfusion injury. N Engl J Med 357:1121–1135. doi:10.1093/cvr/cvq053
Zhao Z-Q, Corvera JS, Halkos ME, Kerendi F, Wang N-P, Guyton RA, Vinten-Johansen J (2003) Inhibition of myocardial injury by ischemic postconditioning during reperfusion: comparison with ischemic preconditioning. Am J Physiol Heart Circ Physiol 285:H579–H588. doi:10.1152/ajpheart.01064.2002
Acknowledgments
The authors’ studies were supported by the Dr. Heinz-Horst-Deichmann Foundation. The technical assistances of Sandra Krüger and Jelena Löblein are gratefully acknowledged.
Ethical standards
The experimental protocols were approved by the Bioethical Committee of the district of Düsseldorf (G1240/11).
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Baars, T., Skyschally, A., Klein-Hitpass, L. et al. microRNA expression and its potential role in cardioprotection by ischemic postconditioning in pigs. Pflugers Arch - Eur J Physiol 466, 1953–1961 (2014). https://doi.org/10.1007/s00424-013-1429-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-013-1429-3