Abstract
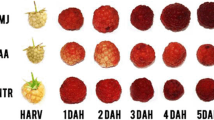
Knowledge of ripeness and regulation of postharvest processes is an important tool to prevent loss of commercial value in both fruit and cut flower markets. The joint analysis of hormones and vitamin E levels can reveal complex interactions between hormones and oxidative stress as key regulators of postharvest processes. Profiling of both groups of metabolic compounds was performed during the ripening of non-climacteric fruits (red raspberry, Rubus idaeus L.) and senescence of ethylene-insensitive flowers (Dutch Iris, Iris x hollandica L.). After an initial extraction of the sample, without further purification steps, the hormonal profile was analyzed by UPLC-MS/MS and vitamin E levels were measured by HPLC. This methodological approach was very fast and had enough sensitivity for the analysis of small samples. Raspberry fruit maturation was characterized by a decline of cytokinin levels [zeatin, zeatin riboside, 2-isopentenyl adenine, and isopentenyl adenosine (Z, ZR, 2-iP, and IPA, respectively)] and gibberellins (GA1 in particular). Exogenous application of ABA prevented δ-tocopherol loss during fruit ripening. Iris floral senescence was also under strict hormonal control, also mediated by cytokinins and gibberellins. Z, ZR, 2-iP, GA9, and GA24 levels decreased in inner tepals, whereas the level of IPA decreased in style-merged-to-stigma tissues, thus suggesting tissue-specific roles for different hormones. α-Tocopherol levels decreased during senescence of inner tepals, hence suggesting enhanced oxidative stress. In conclusion, the rapid and sensitive hormonal and vitamin E profiling presented here can help in understanding the key physiological processes underlying fruit ripening and floral senescence.





Similar content being viewed by others
References
Adams-Phillips L, Barry C, Giovannoni J (2004) Signal transduction systems regulating fruit ripening. Trends Plant Sci 9:331–338
Aharoni A, Keizer LCP, Broeck HCVD et al (2002) Novel insight into vascular, stress, and auxin-dependent and -independent gene expression programs in strawberry, a non-climacteric fruit. Plant Physiol 129:1019–1031
Anttonen MJ, Karjalainen RO (2005) Environmental and genetic variation of phenolic compounds in red raspberry. J Food Compos Anal 18:759–769
Archbold D, Dennis F (1984) Quantification of free ABA and free and conjugated IAA in strawberry achene and receptacle tissue during fruit development. J Am Soc Hortic Sci 109:330–335
Arrom L, Munné-Bosch S (2010) Tocopherol composition in flower organs of Lilium and its variations during natural and artificial senescence. Plant Sci 179:289–295
Beekwilder J, Hall RD, Ric Vos CHD (2005a) Identification and dietary relevance of antioxidants from raspberry. BioFactors 23:197–205
Beekwilder J, Jonker H, Meesters P et al (2005b) Antioxidants in raspberry: on-line analysis links antioxidant activity to a diversity of individual metabolites. J Agric Food Chem 53:3313–3320
Beekwilder J, van der Meer IM, Simic A et al (2008) Metabolism of carotenoids and apocarotenoids during ripening of raspberry fruit. BioFactors 34:57–66
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein dye binding. Anal Biochem 72:248–254
Brigelius-Flohé R (2009) Vitamin E: the shrew waiting to be tamed. Free Rad Biol Med 46:543–554
Burdon JN, Sexton R (1990) The role of ethylene in the shedding of red raspberry fruit. Ann Bot 66:111–120
Celikel FG, van Doorn WG (1995) Solute leakage, lipid peroxidation, and protein degradation during the senescence of Iris tepals. Physiol Plant 94:515–521
Chervin C, El-Kereamy A, Roustan J-P et al (2004) Ethylene seems required for the berry development and ripening in grape, a non-climacteric fruit. Plant Sci 167:1301–1305
De Ancos B, Gonzalez EM, Cano MP (2000) Ellagic acid, vitamin C, and total phenolic contents and radical scavenging capacity affected by freezing and frozen storage in raspberry fruit. J Agric Food Chem 48:4565–4570
Dehnen-Schmutz K, Holdenrieder O, Jeger MJ, Pautasso M (2010) Structural change in the international horticultural industry: some implications for plant health. Sci Hortic 125:1–15
Evenson RE, Gollin D (2003) Assessing the impact of the Green Revolution, 1960 to 2000. Science 300:758–762
Falk J, Munné-Bosch S (2010) Tocochromanol functions in plants: antioxidation and beyond. J Exp Bot 61:1549–1566
FAO (2011) The state of the world’s land and water resources for food and agriculture. A summary report. Rome, United Nations Food and Agriculture Organization
Gleason M, Lewis D, Nonnecke G, Hatterman-Valenti H (1990) Iowa commercial small fruit spray guide. Iowa State Coop Ext Serv Bull Pm 1375
Hoagland DR, Arnon DI (1950) The water-culture method for growing plants without soil. Calif Agric Exp Sta Circ 347
Hosomi A, Arita M, Sato Y et al (1997) Affinity for α-tocopherol transfer protein as a determinant of the biological activities of vitamin E analogs. FEBS Lett 409:105–108
Jia HF, Chai YM, Li CL et al (2011) Abscisic acid plays an important role in the regulation of strawberry fruit ripening. Plant Physiol 157:188–199
Jiang Y, Joyce DC (2003) ABA effects on ethylene production, PAL activity, anthocyanin and phenolic contents of strawberry fruit. Plant Growth Regul 39:171–174
Kader AA (2005) Increasing food availability by reducing postharvest losses of fresh produce. Acta Hortic 682:2169–2176
Kähkönen MP, Hopia AI, Heinonen M (2001) Berry phenolics and their antioxidant activity. J Agric Food Chem 49:4076–4082
Kalt W, Forney CF, Martin A, Prior RL (1999) Antioxidant capacity, vitamin C, phenolics, and anthocyanins after fresh storage of small fruits. J Agric Food Chem 47:4638–4644
Kamal-Eldin A, Appelqvist LÅ (1996) The chemistry and antioxidant properties of tocopherols and tocotrienols. Lipids 31:671–701
Krüger E, Dietrich H, Schöpplein E et al (2011) Cultivar, storage conditions and ripening effects on physical and chemical qualities of red raspberry fruit. Postharvest Biol Technol 60:31–37
Lawson RH (1996) Economic importance and trends in ornamental horticulture. Acta Hortic 432:226–237
Lichtenthaler HK, Wellburn AR (1983) Determinations of total carotenoids and chlorophylls a and b of leaf extracts in different solvents. Biochem Soc Trans 11:591–592
Macnish AJ, Jiang CZ, Reid MS (2010) Treatment with thidiazuron improves opening and vase life of iris flowers. Postharvest Biol Technol 56:77–84
Maeda H, DellaPenna D (2007) Tocopherol functions in photosynthetic organisms. Curr Opin Plant Biol 10:260–265
Mehrshahi P, Stefano G, Andaloro JM, Brandizzi F, Froehlich JE, DellaPenna D (2013) Transorganellar complementation redefines the biochemical continuity of endoplasmic reticulum and chloroplasts. Proc Natl Acad Sci USA 110:12126–12131
Mezzetti B, Landi L, Pandolfini T, Spena A (2004) The defH9-iaaM auxin-synthesizing gene increases plant fecundity and fruit production in strawberry and raspberry. BMC Biotechnol 4:4
Mullen W, Stewart AJ, Lean MEJ et al (2002) Effect of freezing and storage on the phenolics, ellagitannins, flavonoids, and antioxidant capacity of red raspberries. J Agric Food Chem 50:5197–5201
Müller M, Cela J, Asensi-Fabado MA, Munné-Bosch S (2012) Tocotrienols in plants: occurrence, biosynthesis and function. In: Tan B, Watson RR, Preedy VR (eds) Tocotrienols: vitamin E beyond tocopherols, 2nd edn. CRC Press, Boca Raton, pp 1–16
Neocleous D, Vasilakakis M (2007) Effects of NaCl stress on red raspberry (Rubus idaeus L. “Autumn Bliss”). Sci Hortic 112:282–289
Pak C, van Doorn WG (2005) Delay of Iris flower senescence by protease inhibitors. New Phytol 165:473–480
Perkins-Veazie P (1995) Growth and ripening of strawberry fruit. Hortic Rev 17:267–297
Perkins-Veazie P, Nonnecke G (1992) Physiological changes during ripening of raspberry fruit. HortScience 27:331–333
Pilati S, Perazzolli M, Malossini A et al (2007) Genome-wide transcriptional analysis of grapevine berry ripening reveals a set of genes similarly modulated during three seasons and the occurrence of an oxidative burst at vèraison. BMC Genomics 8:428
Rao AV, Snyder DM (2010) Raspberries and human health: a review. J Agric Food Chem 58:3871–3883
Remberg SF, Soønsteby A, Aaby K, Heide OM (2010) Influence of postflowering temperature on fruit size and chemical composition of glen ample raspberry (Rubus idaeus L.). J Agric Food Chem 58:9120–9128
Rogers HJ (2012) Is there an important role for reactive oxygen species and redox regulation during floral senescence? Plant, Cell Environ 35:217–233
Rogers HJ (2013) From models to ornamentals: how is flower senescence regulated? Plant Mol Biol 82(6):563–574
Schippers JHM, Jing HC, Hille J, Dijkwel PP (2007) Developmental and hormonal control of leaf senescence. In: Gan S (ed) Senescence processes in plants, 2nd edn. Blackwell, Oxford, pp 145–170
Schuelke M, Elsner A, Finckh B et al (2000) Urinary alpha-tocopherol metabolites in alpha-tocopherol transfer protein-deficient patients. J Lipid Res 41:1543–1551
Siegelman HW, Hendricks SB (1958) Photocontrol of alcohol, aldehyde, and anthocyanin production in apple skin. Plant Physiol 33:409–413
Symons GM, Chua YJ, Ross JJ et al (2012) Hormonal changes during non-climacteric ripening in strawberry. J Exp Bot 63:4741–4750
Talon M, Tadeo FR, Ben-Cheikh W et al (1998) Hormonal regulation of fruit set and abscission in Citrus: classical concepts and new evidence. Acta Hortic 463:209–218
Tan B, Watson RR, Preedy VR (2012) Tocotrienols: vitamin E beyond tocopherols, 2nd edn. CRC Press, Boca Raton
Tilman D, Cassman KG, Matson PA et al (2002) Agricultural sustainability and intensive production practices. Nature 418:671–677
Tripathi SK, Tuteja N (2007) Integrated signaling in flower senescence: an overview. Plant Signal Behav 2:437–445
Van Doorn WG (2001) Categories of petal senescence and abscission: a re-evaluation. Ann Bot 87:447–456
Van Doorn WG, Woltering EJ (2008) Physiology and molecular biology of petal senescence. J Exp Bot 59:453–480
Van Doorn WG, Çelikel FG, Pak C, Harkema H (2012) Delay of Iris flower senescence by cytokinins and jasmonates. Physiol Plant 148:105–120
Wang SY, Lin HS (2000) Antioxidant activity in fruits and leaves of blackberry, raspberry, and strawberry varies with cultivar and developmental stage. J Agric Food Chem 48:140–146
Wang SY, Zheng W (2005) Preharvest application of methyl jasmonate increases fruit quality and antioxidant capacity in raspberries. Int J Food Sci Technol 40:187–195
Wang SY, Chen CT, Wang CY (2009) The influence of light and maturity on fruit quality and flavonoid content of red raspberries. Food Chem 112:676–684
Weberling F (1989) Morphology of flowers and inflorescences. Cambridge University Press, Cambridge
Woltering EJ, van Doorn W (2009) Petal senescence: new concepts for ageing cells. Acta Hortic 847:161–170
Zaharia LI, Walker-Simmon MK, Rodríguez CN, Abrams SR (2005) Chemistry of abscisic acid, abscisic acid catabolites and analogs. J Plant Growth Regul 24:274–284
Acknowledgments
This work was supported by the prize ICREA Academia given to SMB, funded by the Generalitat de Catalunya. JAM holds a FPU fellowship from the Spanish Government. We are very grateful to Serveis de Camps Experimentals and Serveis Científico-tècnics (University of Barcelona) for technical assistance.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Miret, J.A., Cela, J., Bezerra, L.A. et al. Application of a Rapid and Sensitive Method for Hormonal and Vitamin E Profiling Reveals Crucial Regulatory Mechanisms in Flower Senescence and Fruit Ripening. J Plant Growth Regul 33, 34–43 (2014). https://doi.org/10.1007/s00344-013-9375-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00344-013-9375-z