Abstract
Objectives
To develop a new tract imaging technique for visualising small fibre tracts of the brainstem and for detecting the abnormalities in multiple system atrophy of the cerebellar type (MSA-C) using a phase difference enhanced (PADRE) imaging technique, in which the phase difference between the target and surrounding tissue is selectively enhanced.
Methods
Two neuroradiologists compared the high-spatial-resolution PADRE imaging, which was acquired from six healthy volunteers, three patients with MSA-C, and 7 patients with other types of neurodegenerative diseases involving the brainstem or cerebellum.
Results
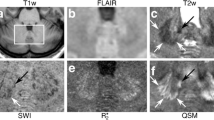
Various fine fibre tracts in the brainstem, the superior and inferior cerebellar peduncles, medial lemniscus, spinothalamic tract, medial longitudinal fasciculus, central tegmental tract, corticospinal tract and transverse pontine fibres, were identified on PADRE imaging. PADRE imaging from MSA-C demonstrated the disappearance of transverse pontine fibres and significant atrophy of the inferior cerebellar peduncles, while the superior cerebellar peduncles were intact. PADRE imaging also demonstrated that the transverse pontine fibres and inferior cerebellar peduncle were not involved in the other neurodegenerative diseases.
Conclusion
PADRE imaging can offer a new form of tract imaging of the brainstem and may have the potential to reinforce the clinical utility of MRI in differentiating MSA from other conditions.



Similar content being viewed by others
References
Wakana S, Jiang H, Nagae-Poetscher LM, van Zijl PC, Mori S (2004) Fiber tract-based atlas of human white matter anatomy. Radiology 230:77–87
Nagae-Poetscher LM, Jiang H, Wakana S, Golay X, van Zijl PC, Mori S (2004) High-resolution diffusion tensor imaging of the brain stem at 3T. AJNR Am J Neuroradiol 25:1325–1330
Haacke EM, Xu Y, Cheng YC, Reichenbach JR (2004) Susceptibility weighted imaging (SWI). Magn Reson Med 52:612–618
Haacke EM, Cheng NY, House MJ, Liu Q, Neelavalli J, Ogg RJ, Khan A, Ayaz M, Kirsch W, Obenaus A (2005) Imaging iron stores in the brain using magnetic resonance imaging. Magn Reson Imaging 23:1–25
Haacke EM, Ayaz M, Khan A, Manova ES, Krishnamurthy B, Gollapalli L, Ciulla C, Kim I, Petersen F, Kirsch W (2007) Establishing a baseline phase behavior in magnetic resonance imaging to determine normal vs. abnormal iron content in the brain. J Magn Reson Imaging 26:256–264
Kakeda S, Korogi Y, Kamada K, Ohnari N, Moriya J, Sato T, Kitajima M, Hasnine H, Hirata N (2008) Signal intensity of the motor cortex on phase-weighted imaging at 3T. AJNR Am J Neuroradiol 29:1171–1175
Yoneda T (2009) Triple-layer appearance of human cerebral cortices on phase-difference enhanced imaging using 3D principle of echo shifting with a train of observations (PRESTO) sequence. Proc Int Soc Magn Reson Med 17:27
Wenning GK, Ben Shlomo Y, Magalhaes M, Daniel SE, Quinn NP (1994) Clinical features and natural history of multiple system atrophy. An analysis of 100 cases. Brain 117(Pt 4):835–845
Gilman S, Low PA, Quinn N, Albanese A, Ben-Shlomo Y, Fowler CJ, Kaufmann H, Klockgether T, Lang AE, Lantos PL, Litvan I, Mathias CJ, Oliver E, Robertson D, Schatz I, Wenning GK (1999) Consensus statement on the diagnosis of multiple system atrophy. J Neurol Sci 163:94–98
Harding AE, Deufel T (eds) (1993) Inherited ataxias. In: Advances in neurology, vol 61. New York: Raven
Nolte J (2003) The human brain, 5th edn. Mosby Year Book, St Louis
Matsusue E, Fujii S, Kanasaki Y, Kaminou T, Ohama E, Ogawa T (2009) Cerebellar lesions in multiple system atrophy: postmortem MR imaging-pathologic correlations. AJNR Am J Neuroradiol 30:1725–1730
Wenning GK, Tison F, Elliott L, Quinn NP, Daniel SE (1996) Olivopontocerebellar pathology in multiple system atrophy. Mov Disord 11:157–162
Papp MI, Kahn JE, Lantos PL (1989) Glial cytoplasmic inclusions in the CNS of patients with multiple system atrophy (striatonigral degeneration, olivopontocerebellar atrophy and Shy-Drager syndrome). J Neurol Sci 94:79–100
Dickson DW, Lin W, Liu WK, Yen SH (1999) Multiple system atrophy: a sporadic synucleinopathy. Brain Pathol 9:721–732
Naka H, Ohshita T, Murata Y, Imon Y, Mimori Y, Nakamura S (2002) Characteristic MRI findings in multiple system atrophy: comparison of the three subtypes. Neuroradiology 44:204–209
Schott JM, Simon JE, Fox NC, King AP, Khan MN, Cipolotti L, Paviour DC, Stevens JM, Rossor MN (2003) Delineating the sites and progression of in vivo atrophy in multiple system atrophy using fluid-registered MRI. Mov Disord 18:955–958
Lee EA, Cho HI, Kim SS, Lee WY (2004) Comparison of magnetic resonance imaging in subtypes of multiple system atrophy. Parkinsonism Relat Disord 10:363–368
Papp MI, Lantos PL (1994) The distribution of oligodendroglial inclusions in multiple system atrophy and its relevance to clinical symptomatology. Brain 117(Pt 2):235–243
Steele JC, Richardson JC, Olszewski J (1964) Progressive supranuclear palsy. A heterogeneous degeneration involving the brain stem, basal ganglia and cerebellum with vertical gaze and pseudobulbar palsy, nuchal dystonia and dementia. Arch Neurol 10:333–359
Tsuboi Y, Slowinski J, Josephs KA, Honer WG, Wszolek ZK, Dickson DW (2003) Atrophy of superior cerebellar peduncle in progressive supranuclear palsy. Neurology 60:1766–1769
Giussani C, Poliakov A, Ferri RT, Plawner LL, Browd SR, Shaw DW, Filardi TZ, Hoeppner C, Geyer JR, Olson JM, Douglas JG, Villavicencio EH, Ellenbogen RG, Ojemann JG (2010) DTI fiber tracking to differentiate demyelinating diseases from diffuse brain stem glioma. Neuroimage 52:217–223
Griswold MA, Jakob PM, Nittka M, Goldfarb JW, Haase A (2000) Partially parallel imaging with localized sensitivities (PILS). Magn Reson Med 44:602–609
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Supplementary Figures (1–26) shows the sequential PADRE imaging.
The numbers in the PADRE images represent the major anatomical structures analysed in this study. These are 1, inferior cerebellar peduncle; 2, transverse pontine fibre; 3, superior cerebellar peduncle; 4, central tegmental tract; 5, medial longitudinal fasciculus; 6, medial lemniscus; 7, spinothalamic tract; 8, facial nerve; 9, trigeminal nerve; 10, corticospinal tract; 11, inferior olivary nucleus; 12, red nucleus; 13, substantia nigra; and 14, cerebral aqueduct of Sylvius.
Supplementary Fig. 1
(JPEG 32 kb)
Supplementary Fig. 2
(JPEG 34 kb)
Supplementary Fig. 3
(JPEG 34 kb)
Supplementary Fig. 4
(JPEG 30 kb)
Supplementary Fig. 5
(JPEG 30 kb)
Supplementary Fig. 6
(JPEG 33 kb)
Supplementary Fig. 7
(JPEG 28 kb)
Supplementary Fig. 8
(JPEG 35 kb)
Supplementary Fig. 9
(JPEG 33 kb)
Supplementary Fig. 10
(JPEG 38 kb)
Supplementary Fig. 11
(JPEG 32 kb)
Supplementary Fig. 12
(JPEG 35 kb)
Supplementary Fig. 13
(JPEG 33 kb)
Supplementary Fig. 14
(JPEG 35 kb)
Supplementary Fig. 15
(JPEG 36 kb)
Supplementary Fig. 16
(JPEG 32 kb)
Supplementary Fig. 17
(JPEG 34 kb)
Supplementary Fig. 18
(JPEG 35 kb)
Supplementary Fig. 19
(JPEG 33 kb)
Supplementary Fig. 20
(JPEG 28 kb)
Supplementary Fig. 21
(JPEG 29 kb)
Supplementary Fig. 22
(JPEG 27 kb)
Supplementary Fig. 23
(JPEG 30 kb)
Supplementary Fig. 24
(JPEG 29 kb)
Supplementary Fig. 25
(JPEG 27 kb)
Supplementary Fig. 26
(JPEG 24 kb)
Rights and permissions
About this article
Cite this article
Kakeda, S., Korogi, Y., Yoneda, T. et al. A novel tract imaging technique of the brainstem using phase difference enhanced imaging: normal anatomy and initial experience in multiple system atrophy. Eur Radiol 21, 2202–2210 (2011). https://doi.org/10.1007/s00330-011-2158-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-011-2158-7