Abstract
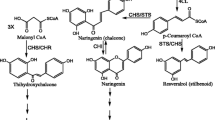
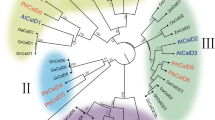
As perennial plants, Populus species are constantly exposed to environmental stresses, such as wounding and pathogen attack, which lead to production of compounds including lignin, flavonoids and phytoalexins. Chalcone synthase (CHS) is a key enzyme in the flavonoid biosynthesis pathway. In this study, a cDNA clone encoding CHS was isolated from Populus trichocarpa by reverse transcription-polymerase chain reaction (RT-PCR). The full-length cDNA, named PtrCHS4, was 1,314 bp with a 1,173 bp open reading frame that corresponded to a deduced protein of 391 amino acid residues. Multiple sequence alignments showed that PtrCHS4 shared high homology with CHS proteins from other plants. Phylogenetic analysis revealed that PtrCHS4 was most closely related to PhCHS from Petunia hybrida and NaCHS from Nicotiana attenuata. Semi-quantitative RT-PCR analysis identified that the PtrCHS4 gene was abundantly expressed in the leaves and stems, while its expression was drastically reduced in the roots. Transcript abundance of PtrCHS4 was stimulated by 2.5-fold within 24 h of wounding treatment. Promoter analysis confirmed that the PtrCHS4 promoter was capable of directing expression of the GUS reporter in both wounded and unwounded leaves of transgenic Chinese white poplar (P. tomentosa Carr.), indicating that the PtrCHS4 promoter is systemically responsive to wounding stimuli. Furthermore, promoter deletion analysis showed that the proximal 1,592 bp from the transcription start site were required for promoter activation by jasmonic acid and the −1,096 to −148 region was proved to be necessary for establishing wound-induced pattern of expression.








Similar content being viewed by others
References
Bevan RD (1984) Trophic effects of peripheral adrenergic nerves on vascular structure. Hypertension 6(6 Pt 2):19–26
Bradforda MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Chem 72:248–254
Constabel CP, Lynn Y, Patton JJ, Christopher ME (2000) Polyphenol oxidase from hybrid poplar. Cloning and expression in response to wounding and herbivory. Plant Physiol 124:285–295
Coyle DR, Nebeker TE, Hart ER, Mattson WJ Jr (2005) Biology and management of insect pests in North American intensively-managed hardwood forest systems. Annu Rev Entomol 50:1–29
Creelman RA, Mullet JE (1997) Biosynthesis and action of jasmonates in plants. Annu Rev Plant Physiol Plant Mol Biol 48:355–381
Davis J, Gordon M, Smit B (1991) Assimilate movement dictates remote sites of wound-induced gene expression in poplar leaves. Proc Natl Acad Sci USA 88:2393–2396
De Bruxelles GL, Roberts MR (2001) Signals regulating multiple responses to wounding and herbivores. Crit Rev Plant Sci 20:487–521
Dixon RA (1986) The phytoalexins response: elicitation, signaling, and control of host gene expression. Biol Rev 61:239–291
Dixon RA, Paiva NL (1995) Stress-induced phenylpropanoid metabolism. Plant Cell 7:1085–1097
Ellard-Ivey M, Douglas CJ (1996) Role of jasmonates in the elicitor- and wound-inducible expression of defense genes in parsley and transgenic tobacco. Plant Physiol 112:183–192
Farmer EE, Johnson RR, Ryan CA (1992) Regulation of expression of proteinase inhibitor genes by methyl jasmonate and jasmonic acid. Plant Physiol 98:995–1002
Feinbaum RL, Ausubel FM, Höfgen R, Willmizer L (1988) Storage of competent cells for Agrobacterium transformation. Nucleic Acids Res 16:9877
Ferrer JL, Jez JM, Bowman ME, Dixon RA, Noel JP (1999) Structure of chalcone synthase and the molecular basis of plant polyketide biosynthesis. Nat Struct Biol 6:775–784
Fukuda-Tanaka S, Hoshino A, Hisatomi Y, Habu Y, Hasebe M, Iida S (1997) Identification of new chalcone synthase genes for flower pigmentation in the Japanese and common morning glories. Plant Cell Physiol 38:754–758
Gläßgen WE, Rose A, Madlung J, Koch W, Gleitz J, Seitz HU (1998) Regulation of enzymes involved in anthocyanin biosynthesis in carrot cell cultures in response to treatment with ultraviolet light and fungal elicitors. Planta 204:490–498
Guerineau F, Benjdia M, Zhou DX (2003) A jasmonate-responsive element within the A. thaliana vsp1 promoter. J Exp Bot 54:1153–1162
Gundlach H, Müller MJ, Kutchan TM, Zenk MH (1992) Jasmonic acid is a signal transducer in elicitor-induced plant cell cultures. Proc Natl Acad Sci USA 89:2389–2393
Hahn K, Strittmatter G (1994) Pathogen defence gene prp1–1from potato encodes an auxin-responsive glutathione Stransferease. Eur J Biochem 226:619–626
Heller W, Hahlbrock K (1980) Highly purified “flavanone synthase” from parsley catalyzes the formation of naringenin chalcone. Arch Biochem Biophys 200:617–619
Higo K, Ugawa Y, Iwamoto M, Korenaga T (1999) Plant cis-acting regulatory DNA elements (PLACE) database. Nucleic Acids Res 27:297–300
Höfgen R, Willmitzer L (1988) Storage of competent cell for Agrobacterium transformation. Nucleic Acids Res 16:9877
Hollick JB, Gordon MP (1995) Transgenic analysis of a hybrid poplar wound-inducible promoter reveals developmental patterns of expression similar to that of storage protein genes. Plant Physiol 109:73–85
Ito M, Ichinose Y, Kato H, Shiraishi T, Yamada T (1997) Molecular evolution and functional relevance of the chalcone synthase genes of pea. Mol Gen Genet 255:28–37
Jefferson RA (1987) Assaying chimeric genes in plants: the gus gene fusion system. Plant Mol Biol Rep 5:387–405
Jefferson RA, Kavanagh TA, Bevan MW (1987) GUS fusions: β-glucuronidase as a sensitive and versatile gene fusion marker in higher plants. EMBO J 6:3901–3907
Jez JM, Noel JP (2000) A kaleidoscope of carotenoids. Nat Biotech 18:825–826
Jia ZC, Gou JQ, Sun YM, Yuan L, Tang Q, Yang XY, Pei Y, Luo KM (2010) Enhanced resistance to fungal pathogens in transgenic Populus tomentosa Carr. by overexpression of an nsLTP-like antimicrobial protein gene from motherwort (Leonurus japonicus). Tree Physiol 30:1599–1605
Kim SR, Choi JL, Costa MA, An G (1992) Identification of G-box sequence as an essential element for methyl jasmonate response of potato proteinase inhibitor II promoter. Plant Physiol 99:627–631
Kreuzaler F, Ragg H, Fautz E, Kuhn DN, Hahlbrock K (1983) UV-induction of chalcone synthase mRNA in cell suspension cultures of Petroselinum hortense. Proc Natl Acad Sci USA 80:2591–2593
Lawton MA, Lamb CJ (1987) Transcriptional activation of plant defense genes by fungal elicitor, wounding, and infection. Mol Cell Biol 7:335–341
Lindroth RL, Hwang SY (1996) Diversity, redundancy, and multiplicity in chemical defense systems of aspen. In: Romeo JT, Saunders JA, Barbosa P (eds) Phytochemical diversity and redundancy in ecological interactions. Plenum Press, New York, pp 25–56
Lloyd G, McCown B (1980) Commercially feasible micropropagation of mountain laurel, Kalmia latifolia, by use of shoot-tip culture. Int Plant Prop Soc Proc 30:421–427
Luo KM, Deng W, Xiao YH, Zheng XZ, Li Y, Pei Y (2006) Leaf senescence is delayed in tobacco plants expressing the maize knotted1gene under the control of a wound-inducible promoter. Plant Cell Rep 25:1246–1254
Maleck K, Levine A, Eulgem T, Morgen A, Schmid J, Lawton K, Dangl JL, Dietrich RA (2000) The transcriptome of Arabidopsis thaliana during systemic acquired resistance. Nat Genet 26:403–410
Martin CR (1993) Structure, function, and regulation of the chalcone synthase. Int Rev Cytol 147:233–284
Mason HS, DeWald DB, Mullet JE (1993) Identification of a methyl jasmonate-responsive domain in the soybean vspB promoter. Plant Cell 5:241–251
Matton DP, Prescott G, Bertrand C, Camirand A, Brisson N (1993) Identification of cis-acting elements involved in the regulation of the 17 kDa pathogenesis-related gene STH-2 in potato. Plant Mol Biol 22:279–291
Mellway RD, Tran LT, Prouse MB, Campbell MM, Constabel CP (2009) The wound-, pathogen-, and ultraviolet B-responsive MYB134 gene encodes R2R3 MYB transcription factor that regulates proanthocyanidin synthesis in poplar. Plant Physiol 150:924–941
Miranda M, Ralph SG, Mellway R, White R, Heath MC, Bohlmann J, Constabel CP (2007) The transcriptional response of hybrid poplar (Populus trichocarpa × P. deltoides) to infection by Melampsora medusae leaf rust involves induction of flavonoid pathway genes leading to the accumulation of proanthocyanidins. Mol Plant Microbe Interact 20:816–831
Mueller MJ, Brodschelm W, Spannagl E, Zenk MH (1993) Signaling in the elicitation process is mediated through the octadecanoid pathway leading to jasmonic acid. Proc Natl Acad Sci USA 90:7490–7494
Nishiuchi T, Kodama H, Yanagisawa S, Iba K (1999) Wound-induced expression of the FAD7 gene is mediated by different regulatory domains of its promoter in leaves/stems and roots. Plant Physiol 121:1239–1246
Nishiuchi T, Shinshi H, Suzuki K (2004) Rapid and transient activation of transcription of the ERF3 gene by wounding in tobacco leaves: possible involvement of NtWRKYs and autorepression. J Biol Chem 279:55355–55361
Pang YZ, Shen GA, Liu CH, Liu XJ, Tan F, Sun XF, Tang KX (2004) Molecular cloning and sequence analysis of a novel chalcone synthase cDNA from Ginkgo biloba. DNA Seq 15:283–290
Peters DJ, Constabel CP (2002) Molecular analysis of herbivore-induced condensed tannin synthesis: cloning and expression of dihydroflavonol reductase from trembling aspen (Populus tremuloides). Plant J 32:701–712
Richard S, Lapointe G, Rutledge RG, Séguin A (2000) Induction of chalcone synthase expression in white spruce by wounding and jasmonate. Plant Cell Physiol 41:982–987
Ronald EK, Cornelis ES, Jos NMM, Anton GMG (1987) The chalcone synthase multigene family of Petunia hybrida (V30): sequence homology, chromosomal localization, and evolutionary aspects. Plant Mol Biol 10:159–169
Rouster J, Leah R, Mundy J, Cameron-Mills V (1997) Identification of a methyl jasmonate-responsive region in the promoter of a lipoxygenase 1 gene expressed in barley grain. Plant J 11:513–523
Ruíz-Rivero OJ, Prat S (1998) A −308 deletion of the tomato LAP promoters is able to direct flower-specific and MeJA-induced expression in transgenic plants. Plant Mol Biol 36:639–648
Rushton PJ, Reinstadler A, Lipka V, Lippok B, Somssich IE (2002) Synthetic plant promoters containing defined regulatory elements provide novel insights into pathogen- and wound-induced signaling. Plant Cell 14:749–762
Rushton PJ, Torres JT, Parniske M, Wernert P, Hahlbrock K, Somssich IE (1996) Interaction of elicitor-induced DNA binding proteins with elicitor response elements in the promoters of parsley PR1 genes. EMBO J 15:5690–5700
Sallaud C, El-Turk J, Breda C, Buffard D, De Kozak I, Esnault R, Kondorosi A (1995) Differential expression of cDNA coding for chalcone reductase, a key enzyme of the 5-deoxyflavonoid pathway, under various stress conditions in Medicago sativa. Plant Sci 109:179–190
Sambrook J, Russell DW (2001) Molecular cloning. a laboratory manual, 3rd edn. Cold Spring Harbor Laboratory Press, Cold Spring Harbor
Scheffler BE, Reddy A, Hoffmann I, Wienand U (1995) Chalcone synthase cDNA from Rice (Oryza sativa). Plant Physiol 109:722
Schmelzer E, Jahnen W, Hahlbrock K (1988) In situ localization of light-induced chalcone synthase mRNA, chalcone synthase, and flavonoid end products in epidermal cells of parsley leaves. Proc Natl Acad Sci USA 85:2989–2993
Schubert R, Fischer R, Hain R, Schreier PH, Bahnweg G, Ernst D, Sandermann HJ (1997) An ozone-responsive region of the grapevine resveratrol synthase promoter differs from the basal pathogen-responsive sequence. Plant Mol Biol 34:417–426
Schultz JC, Baldwin IT (1982) Oak leaf quality declines in response to defoliation by gypsy moth larvae. Science 217:149–151
Sembdner G, Parthier B (1993) The biochemistry and the physiological and molecular actions of jasmonates. Annu Rev Plant Physiol Plant Mol Biol 44:569–589
Stevens MT, Lindroth RL (2005) Induced resistance in the indeterminate growth of aspen (Populus tremuloides). Oecologia 145:298–306
Taylor LP, Jorgensen R (1992) Conditional male fertility in chalcone synthase-deficient petunia. Hered J 831:1–17
Tsai CJ, Harding SA, Tschaplinski TJ, Lindroth RL, Yuan YN (2006) Genome-wide analysis of the structural genes regulating defense phenylpropanoid metabolism in Populus. New Phytol 172:47–62
Warner SAJ, Scott R, Draper J (1993) Isolation of an asparagus intracellular PR gene (AoPR1) wound-responsive promoter by the inverse polymerase chain reaction and its characterization in transgenic tobacco. Plant J 3:191–201
Wasternack C, Parthier B (1997) Jasmonate-signalled plant gene expression. Trends Plant Sci 2:302–307
Winkel-Shirley B (2001) Flavonoid biosynthesis. A colorful model for genetics, biochemistry, cell biology, and biotechnology. Plant Physiol 126:485–493
Wu XF, Wang CL, Xie EB, Gao Y, Fan YL, Liu PQ, Zhao KJ (2009) Molecular cloning and characterization of the promoter for the multiple stress-inducible gene BjCHI1 from Brassica juncea. Planta 229:1231–1242
Yang J, Gu HY (2010) Duplication and divergent evolution of the CHS and CHS-like genes in the chalcone synthase (CHS) superfamily. Chin Sci Bull 51:505–509
Yi J, Derynck MR, Chen L, Dhaubhadel S (2010) Differential expression of CHS7 and CHS8 genes in soybean. Planta 231:741–753
Zhu Q, Dröge-Laser W, Dixon RA, Lamb C (1996) Transcriptional activation of plant defense genes. Curr Opin Genet Dev 6:624–630
Acknowledgments
This work was supported by the National Natural Science Foundation of China (3081576), the National Key Project for Research on Transgenic Plant (2011ZX08010-003), the Natural Science Foundation Project of CQ CSTC (CSTC, 2009BA1004, 2009BB0004) and the Fundamental Research Funds for the Central Universities (XDJK2009B018).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by P. Lakshmanan.
Y. Sun, Q. Tian contributed equally to this work.
Rights and permissions
About this article
Cite this article
Sun, Y., Tian, Q., Yuan, L. et al. Isolation and promoter analysis of a chalcone synthase gene PtrCHS4 from Populus trichocarpa . Plant Cell Rep 30, 1661–1671 (2011). https://doi.org/10.1007/s00299-011-1075-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-011-1075-1