Abstract
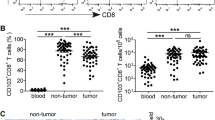
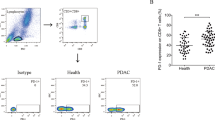
CD160 is an Ig-like glycoprotein expressed by the majority of circulating natural killer cells and γδ T cells. Whether CD160 could regulate CD8+ T-cell functions remains unknown. In this study, we investigated the effects of CD160 on CD8+ T cells in pancreatic cancer. First, we found that the frequency of PD-1+ cells was comparable between CD160+ and CD160−CD8+ T cells, with the former presenting significantly higher PD-1 expression level. In contrast, the frequency of TIM-3+ cells was higher among CD160+ cells but the expression level was comparable between CD160+ and CD160−CD8+ T cells. The IFN-γ and IL-2-expressing CD8+ T cells, directly ex vivo, were highly enriched in the CD160+ subset. However, when CD160+ and CD160−CD8+ T cells were stimulated, the proliferation levels of CD160+ and CD160− cells were initially comparable, but were significantly lower in CD160+CD8+ T cells than in CD160−CD8+ T cells later on. The IFN-γ and IL-2 transcription levels were initially higher in CD160+CD8+ T cells, but eventually reduced in CD160+CD8+ T cells compared to CD160−CD8+ T cells. Also, CD160+CD8+ T cells presented lower cytotoxic capacity than CD160−CD8+ T cells. Interestingly, we observed that tumor-infiltrating CD8+ T cells were significantly enriched with the CD160+ subset in pancreatic cancer patients. In addition, patients with higher frequencies of tumor CD160+CD8+ T cells presented lower survival. Overall, these data demonstrated that tumor-infiltrating CD8+ T cells were enriched with the CD160+ subset in pancreatic cancer, with active effector responses directly ex vivo but limited potential for further activation.







Similar content being viewed by others
Abbreviations
- BTLA:
-
B and T lymphocyte attenuator
- GMFI:
-
Geometric mead fluorescence intensity
- GZMB:
-
Granzyme B
- HVEM:
-
Herpes virus entry mediator
- PRF:
-
Perforin
- Rh:
-
Recombinant human
References
Maïza H, Leca G, Mansur IG, Schiavon V, Boumsell L, Bensussan A (1993) A novel 80-kD cell surface structure identifies human circulating lymphocytes with natural killer activity. J Exp Med 178(3):1121–1126
Anumanthan A, Boumsell L, Christ AD, Blumberg RS, Voss SD, Patel AT, Robertson MJ, Nadler LM, Freeman GJ (1998) Cloning of BY55, a novel Ig superfamily cloning of BY55, a novel ig superfamily member expressed on NK Cells, CTL, and intestinal intraepithelial lymphocytes. J Immunol 161(6):2780–2790
Cai G, Anumanthan A, Brown JA, Greenfield EA, Zhu B, Freeman GJ (2008) CD160 inhibits activation of human CD4+ T cells through interaction with herpesvirus entry mediator. Nat Immunol 9(2):176–185
Farren TW, Giustiniani J, Liu FT, Tsitsikas DA, Macey MG, Cavenagh JD, Oakervee HE, Taussig D, Newland AC, Calaminici M, Bensussan A, Jenner M, Gribben JG, Agrawal SG (2011) Differential and tumor-specific expression of CD160 in B-cell malignancies. Blood 118(8):2174–2183
Liu FT, Giustiniani J, Farren T, Jia L, Bensussan A, Gribben JG, Agrawal SG (2010) CD160 signaling mediates PI3K-dependent survival and growth signals in chronic lymphocytic leukemia. Blood 115(15):3079–3088
Barakonyi A, Rabot M, Marie-Cardine A, Aguerre-Girr M, Polgar B, Schiavon V, Bensussan A, Le Bouteiller P (2004) Cutting edge: engagement of CD160 by its HLA-C physiological ligand triggers a unique cytokine profile secretion in the cytotoxic peripheral blood NK cell subset. J Immunol 173(9):5349–5354
Maeda M, Carpenito C, Russell RC, Dasanjh J, Veinotte LL, Ohta H (2005) Murine CD160, Ig-like receptor on NK cells and NKT cells, recognizes classical and nonclassical MHC class I and regulates NK cell activation. J Immunol 175(7):4426–4432
Muscate F, Stetter N, Schramm C, Schulze zur Wiesch J, Bosurgi L, Jacobs T (2018) HVEM and CD160: regulators of immunopathology during malaria blood-stage. Front Immunol 9:2611
Le Bouteiller P, Barakonyi A, Giustiniani J, Lenfant F, Marie-Cardine A, Aguerre-Girr M, Rabot M, Hilgert I, Mami-Chouaib F, Tabiasco J, Boumsell L, Bensussan A (2002) Engagement of CD160 receptor by HLA-C is a triggering mechanism used by circulating natural killer cells to mediate cytotoxicity. Proc Natl Acad Sci 99(26):16963–16968
Tu TC, Brown NK, Kim T-J, Wroblewska J, Yang X, Guo X (2015) CD160 is essential for NK-mediated IFN-γ production. J Exp Med 212(3):415–429
Nikolova M, Marie-Cardine A, Boumsell L, Bensussan A (2002) BY55/CD160 acts as a co-receptor in TCR signal transduction of a human circulating cytotoxic effector T lymphocyte subset lacking CD28 expression. Int Immunol 14(5):445–451
Viganò S, Banga R, Bellanger F, Pellaton C, Farina A, Comte D (2014) CD160-associated CD8 T-cell functional impairment is independent of PD-1 expression. PLoS Pathog 10(9):e1004380
Sedy JR, Bjordahl RL, Bekiaris V, Macauley MG, Ware BC, Norris PS, Lurain NS, Benedict CA, Ware CF (2013) CD160 Activation by herpesvirus entry mediator augments inflammatory cytokine production and cytolytic function by NK cells. J Immunol 191(2):828–836
Sun H, Xu J, Huang Q, Huang M, Li K, Qu K (2018) Reduced CD160 expression contributes to impaired NK cell function and poor clinical outcomes in patients with HCC. Cancer Res 79(7):1714–1721
Hall ML, Liu H, Malafa M, Centeno B, Hodul PJ, Pimiento J (2016) Expansion of tumor-infiltrating lymphocytes (TIL) from human pancreatic tumors. J Immunother Cancer 4:61
Monney L, Sabatos CA, Gaglia JL, Ryu A, Waldner H, Chernova T, Manning S, Greenfield EA, Coyle AJ, Sobel RA, Freeman GJ, Kuchroo VK (2002) Th1-specific cell surface protein Tim-3 regulates macrophage activation and severity of an autoimmune disease. Nature 415:536–541
Jones RB, Ndhlovu LC, Barbour JD, Sheth PM, Jha AR, Long BR (2008) Tim-3 expression defines a novel population of dysfunctional T cells with highly elevated frequencies in progressive HIV-1 infection. J Exp Med 205:2763–2779
Sakuishi K, Apetoh L, Sullivan JM, Blazar BR, Kuchroo VK, Anderson AC (2010) Targeting Tim-3 and PD-1 pathways to reverse T cell exhaustion and restore anti-tumor immunity. J Exp Med 207:2187–2194
Green DR, Droin N, Pinkoski M (2003) Activation-induced cell death in T cells. Immunol Rev 193:70–81
Cai G, Freeman GJ (2009) The CD160, BTLA, LIGHT/HVEM pathway: a bidirectional switch regulating T-cell activation. Immunol Rev 229(1):244–258
Tsujimura K, Obata Y, Matsudaira Y, Nishida K, Akatsuka Y, Ito Y, Demachi-Okamura A, Kuzushima K, Takahashi T (2006) Characterization of murine CD160+ CD8+ T lymphocytes. Immunol Lett 106(1):48–56
Sideras K, Biermann K, Yap K, Mancham S, Boor PPC, Hansen BE, Stoop HJA, Peppelenbosch MP, van Eijck CH, Sleijfer S, Kwekkeboom J, Bruno MJ (2017) Tumor cell expression of immune inhibitory molecules and tumor-infiltrating lymphocyte count predict cancer-specific survival in pancreatic and ampullary cancer. Int J Cancer 141(3):572–582
Han G, Chen G, Shen B, Li Y (2013) Tim-3: an activation marker and activation limiter of innate immune cells. Front Immunol 4:449
Simon S, Labarriere N (2017) PD-1 expression on tumor-specific T cells: friend or foe for immunotherapy? Oncoimmunology 7(1):e1364828
Funding
No relevant funding.
Author information
Authors and Affiliations
Contributions
SL, WZ, KL, and YW conceived and designed the study; SL and WZ conducted experiments; SL, WZ, and YW analyzed data; SL, WZ, and KL drafted the manuscript; YW supervised the study and reviewed the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical standards
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments. All protocols were reviewed and approved by the Ethics Committee of the First Hospital of Jilin University (160116).
Informed consent
All patients provided written informed consent at the time recruitment. All patients gave the consent to the use of data and materials for research and publication or consent to this particular study.
Cell line authentication
Capan-2 cells were purchased from the American Type Culture Collection.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Liu, S., Zhang, W., Liu, K. et al. CD160 expression on CD8+ T cells is associated with active effector responses but limited activation potential in pancreatic cancer. Cancer Immunol Immunother 69, 789–797 (2020). https://doi.org/10.1007/s00262-020-02500-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-020-02500-3