Abstract
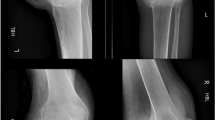
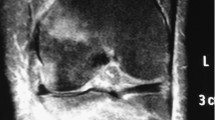
Many individuals with spinal cord injury (SCI) rely on wheelchairs as their primary mode of locomotion leading to reduced weight-bearing on the lower extremities, which contributes to severe bone loss and increased risk of fragility fractures. Engaging in a walking program may reverse this vicious cycle, as this promotes lower extremity weight-bearing and mobility, which may reduce bone loss and fragility fracture risk. However, fragility fracture risk associated with the use of wearable robotic exoskeletons (WREs) in individuals with SCI needs consideration. A 35-year-old man with chronic complete sensorimotor SCI (neurological level = T6) and low initial bone mineral density enrolled in a 6- to 8-week WRE-assisted walking program after successfully completing an initial clinical screening process and two familiarization sessions with the WRE. However, after the first training session with the WRE, he developed bilateral localized ankle edema. Training was suspended, and a CT-scan revealed bilateral calcaneal fractures, which healed with conservative treatment over a 12-week period. Opportunities for improving clinical screening and WRE design are explored. The relevance of developing clinical practice guidelines for safe initiation and progression of intensity during WRE-assisted walking programs is highlighted. This case of bilateral calcaneal fractures illustrates that aiming for “zero risk” during WRE-assisted walking programs may not be realistic. Although WREs are a relatively new technology, current evidence confirms their potential to greatly improve health and quality of life in individuals with chronic SCI. Hence, ensuring their safe use remains a key priority.

Similar content being viewed by others
References
Garland DE, Adkins RH, Scott M, Singh H, Massih M, Stewart C (2004) Bone loss at the os calcis compared with bone loss at the knee in individuals with spinal cord injury. The Journal of Spinal Cord Medicine 27(3):207–211. https://doi.org/10.1080/10790268.2004.11753749
Peppler WT, Kim WJ, Ethans K, Cowley KC (2017) Precision of dual-energy X-ray absorptiometry of the knee and heel: methodology and implications for research to reduce bone mineral loss after spinal cord injury. Spinal Cord 55(5):483–488. https://doi.org/10.1038/sc.2016.170
Edwards WB, Schnitzer TJJCOR (2015) Bone imaging and fracture risk after spinal cord injury. 13(5):310–317. https://doi.org/10.1007/s11914-015-0288-6
Bauman WA, Cardozo CP (2015) Osteoporosis in individuals with spinal cord injury. 7 (2):188–201. doi:doi:https://doi.org/10.1016/j.pmrj.2014.08.948
Giangregorio L, McCartney N (2006) Bone loss and muscle atrophy in spinal cord injury: epidemiology, fracture prediction, and rehabilitation strategies. The Journal of Spinal Cord Medicine 29(5):489–500. https://doi.org/10.1080/10790268.2006.11753898
Clark JM, Findlay DM (2017) Musculoskeletal health in the context of spinal cord injury. Current osteoporosis reports 15(5):433–442. https://doi.org/10.1007/s11914-017-0400-1
Karelis AD, Carvalho LP, Castillo MJ, Gagnon DH, Aubertin-Leheudre M (2017) Effect on body composition and bone mineral density of walking with a robotic exoskeleton in adults with chronic spinal cord injury. J Rehabil Med 49(1):84–87. https://doi.org/10.2340/16501977-2173
Gagnon DH, Vermette M, Duclos C, Aubertin-Leheudre M, Ahmed S, Kairy D (2017) Satisfaction and perceptions of long-term manual wheelchair users with a spinal cord injury upon completion of a locomotor training program with an overground robotic exoskeleton. Disability and rehabilitation Assistive technology:1–8. https://doi.org/10.1080/17483107.2017.1413145
Escalona MJ, Brosseau R, Vermette M, Comtois AS, Duclos C, Aubertin-Leheudre M, Gagnon DH (2018) Cardiorespiratory demand and rate of perceived exertion during overground walking with a robotic exoskeleton in long-term manual wheelchair users with chronic spinal cord injury: a cross-sectional study. Annals of physical and rehabilitation medicine 61(4):215–223. https://doi.org/10.1016/j.rehab.2017.12.008
Benson I, Hart K, Tussler D, van Middendorp JJ (2016) Lower-limb exoskeletons for individuals with chronic spinal cord injury: findings from a feasibility study. 30 (1):73-84. doi:https://doi.org/10.1177/0269215515575166
Bach Baunsgaard C, Vig Nissen U, Katrin Brust A, Frotzler A, Ribeill C, Kalke Y-B, León N, Gómez B, Samuelsson K, Antepohl W, Holmström U, Marklund N, Glott T, Opheim A, Benito J, Murillo N, Nachtegaal J, Faber W, Biering-Sørensen F (2018) Gait training after spinal cord injury: safety, feasibility and gait function following 8 weeks of training with the exoskeletons from Ekso Bionics. Spinal Cord 56(2):106–116. https://doi.org/10.1038/s41393-017-0013-7
Gagnon DH, Escalona MJ, Vermette M, Carvalho LP, Karelis AD, Duclos C, Aubertin-Leheudre M (2018) Locomotor training using an overground robotic exoskeleton in long-term manual wheelchair users with a chronic spinal cord injury living in the community: lessons learned from a feasibility study in terms of recruitment, attendance, learnability, performance and safety. Journal of neuroengineering and rehabilitation 15(1):12. https://doi.org/10.1186/s12984-018-0354-2
Pelletier CA, Miyatani M, Giangregorio L, Craven BC (2016) Sarcopenic obesity in adults with spinal cord injury: a cross-sectional study. Arch Phys Med Rehabil 97(11):1931–1937. https://doi.org/10.1016/j.apmr.2016.04.026
Dimai HP (2017) Use of dual-energy X-ray absorptiometry (DXA) for diagnosis and fracture risk assessment; WHO-criteria, T- and Z-score, and reference databases. Bone 104:39–43. https://doi.org/10.1016/j.bone.2016.12.016
Filippo TRM, De Carvalho MCL, Carvalho LB, de Souza DR, Imamura M, Battistella LR (2015) Proximal tibia fracture in a patient with incomplete spinal cord injury associated with robotic treadmill training. Spinal Cord 53(12):875–876. https://doi.org/10.1038/sc.2015.27
Cervinka T, Lynch CL, Giangregorio L, Adachi JD, Papaioannou A, Thabane L, Craven BC (2017) Agreement between fragility fracture risk assessment algorithms as applied to adults with chronic spinal cord injury. Spinal Cord 55:985–993. https://doi.org/10.1038/sc.2017.65
Asselin PK, Avedissian M, Knezevic S, Kornfeld S, Spungen AM (2016) Training persons with spinal cord injury to ambulate using a powered exoskeleton. Journal of visualized experiments : JoVE 112. https://doi.org/10.3791/54071
Esquenazi A, Talaty M, Packel A, Saulino M (2012) The ReWalk powered exoskeleton to restore ambulatory function to individuals with thoracic-level motor-complete spinal cord injury. Am J Phys Med Rehabil 91(11):911–921. https://doi.org/10.1097/phm.0b013e318269d9a3
Baim S, Leslie WD (2012) Assessment of fracture risk. Current osteoporosis reports 10(1):28–41. https://doi.org/10.1007/s11914-011-0093-9
Allon R, Levy Y, Lavi I, Kramer A, Barzilai M, Wollstein R (2018) How to best predict fragility fractures: an update and systematic review. The Israel Medical Association journal : IMAJ 20(12):773–779
Drey M, Henkel M, Petermeise S, Weiss S, Ferrari U, Rottenkolber M, Schmidmaier R (2018) Assessment of bone and muscle measurements by peripheral quantitative computed tomography in geriatric patients. Journal of clinical densitometry : the official journal of the International Society for Clinical Densitometry. https://doi.org/10.1016/j.jocd.2018.10.002
Hans D, Baim S (2017) Quantitative ultrasound (QUS) in the management of osteoporosis and assessment of fracture risk. J Clin Densitom 20(3):322–333. https://doi.org/10.1016/j.jocd.2017.06.018
Craven BC, Giangregorio LM, Alavinia SM, Blencowe LA, Desai N, Hitzig SL, Masani K, Popovic MR (2017) Evaluating the efficacy of functional electrical stimulation therapy assisted walking after chronic motor incomplete spinal cord injury: effects on bone biomarkers and bone strength. The Journal of Spinal Cord Medicine 40(6):748–758. https://doi.org/10.1080/10790268.2017.1368961
Edwards WB, Simonian N, Haider IT, Anschel AS, Chen D, Gordon KE, Gregory EK, Kim KH, Parachuri R, Troy KL, Schnitzer TJ (2018) Effects of teriparatide and vibration on bone mass and bone strength in people with bone loss and spinal cord injury: a randomized, controlled trial. Journal of bone and mineral research : the official journal of the American Society for Bone and Mineral Research 33(10):1729–1740. https://doi.org/10.1002/jbmr.3525
Smith AJJ, Lemaire ED, Nantel J (2018) Lower limb sagittal kinematic and kinetic modeling of very slow walking for gait trajectory scaling. PLoS One 13(9):e0203934–e0203934. https://doi.org/10.1371/journal.pone.0203934
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
None.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Bass, A., Morin, S., Vermette, M. et al. Incidental bilateral calcaneal fractures following overground walking with a wearable robotic exoskeleton in a wheelchair user with a chronic spinal cord injury: is zero risk possible?. Osteoporos Int 31, 1007–1011 (2020). https://doi.org/10.1007/s00198-020-05277-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-020-05277-4